| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5g |
|
||
| Other Sizes |
|
| 药代性质 (ADME/PK) |
吸收、分布和排泄
对12名受试者进行了对氨基水杨酸及其盐的生物利用度研究。比色法测定结果表明,钠盐、钾盐、钙盐和对氨基水杨酸的血药浓度峰值分别出现在0.5、0.75、1.5和3小时。 尿液排泄数据显示,尽管吸收率有所不同,但吸收基本完全。 氨基水杨酸易于从胃肠道吸收。单次口服4克游离酸后,1.5至2小时内血浆浓度可达约75微克/毫升。钠盐吸收更快。该药物似乎分布于全身体液中,并在胸膜液和干酪样组织中达到较高浓度。然而,脑脊液中的浓度较低,这可能是由于主动外排所致。超过80%的药物经尿液排出;其中超过50%以乙酰化化合物的形式排出。剩余部分的大部分由游离酸组成。 有关对氨基水杨酸(共8种代谢物)的更多吸收、分布和排泄(完整)数据,请访问HSDB记录页面。 代谢/代谢物 肝脏代谢。 乙酰化是许多药物(例如对氨基水杨酸)失活的主要途径。催化这些反应的酶,乙酰辅酶A:N-乙酰转移酶(EC 2.3.1.5),位于肝细胞溶胶中。 口服后,对氨基水杨酸被迅速吸收,并以未改变的对氨基水杨酸和乙酰形式经尿液排出。葡萄糖醛酸、甘氨酰和谷氨酰结合物。 在人体内产生5-氨基-2-羧基苯基-β-D-葡萄糖醛酸苷。在假单胞菌中产生4-氨基儿茶酚。在人体内产生4-氨基水杨酰谷氨酰胺和4-氨基水杨酰甘氨酸。/表格/ 在结核病患者接受异烟肼、噻唑烷酮、对氨基水杨酸和链霉素五种不同药物组合治疗前、治疗期间和停药后,采集其血液进行培养。检测DNA损伤的方法包括染色体畸变和姐妹染色单体交换(SCE)。共分析了179名受试者。这些药物联合使用时表现出协同、相加和拮抗作用,但单独使用时均未发现致染色体断裂作用。四种药物组合,即异烟肼联合噻唑酮、异烟肼联合对氨基水杨酸、异烟肼联合噻唑酮联合链霉素以及异烟肼联合对氨基水杨酸联合链霉素,均显著增加了染色体畸变的发生率,而异烟肼联合链霉素则未诱发染色体畸变。事实上,链霉素似乎降低了染色体畸变的发生率。仅有两名患者的姐妹染色单体交换(SCE)发生率增加:一名接受异烟肼联合噻唑酮治疗,另一名接受异烟肼联合对氨基水杨酸治疗。停药后染色体畸变的发生率下降;虽然略高于对照组,但差异无统计学意义。染色体畸变的恢复可能是由于受损细胞的清除或淋巴细胞中DNA的修复所致。尽管停药后药物引起的异常不会持续存在,但仍应谨慎使用这种染色体损伤药物组合,因为不能排除在治疗期间生殖细胞减数分裂过程中发生染色体损伤的可能性,而这种损伤可能会遗传给下一代。 有关对氨基水杨酸(共9种代谢物)的更多代谢/代谢物(完整)数据,请访问HSDB记录页面。 生物半衰期 该药物的半衰期约为1小时,单次常规剂量给药后4至5小时内血浆浓度可忽略不计。 |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
妊娠期和哺乳期用药
◉ 哺乳期用药概述 现有信息有限,表明母亲服用氨基水杨酸后,乳汁中药物浓度较低,预计不会对母乳喂养的婴儿造成任何不良反应,尤其是在婴儿2个月以上的情况下。如果在哺乳期使用此药,应监测纯母乳喂养的婴儿,注意罕见的黄疸、胃肠道紊乱、低钾血症、血小板减少症、溶血和低钾血症等情况。 ◉ 对母乳喂养婴儿的影响 氨基水杨酸作为多药联合治疗方案的一部分,用于治疗两名患有耐多药结核病的孕妇,治疗贯穿整个孕期和产后。她们的两个婴儿均接受了母乳喂养(未说明喂养程度和持续时间)。两名儿童分别在1.8岁和4.6岁时发育正常,其中一名儿童在1.8岁时出现轻微的语言发育迟缓,另一名儿童则生长发育不良,这可能是由于出生后感染结核病所致。 ◉ 对哺乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 蛋白质结合 50-60% 相互作用 ...对氨基水杨酸.../抑制氯霉素代谢/ 戊巴比妥/SRP:中枢神经系统抑制/在小鼠中经对氨基水杨酸预处理后增强。 INCR /SRP:中枢神经系统抑制/ 似乎是由于戊巴比妥从血清蛋白结合中释放出来,导致脑内浓度升高…… 与链霉素和异烟肼有相加和协同作用。 丙磺舒会降低该药物的肾脏排泄。 有关对氨基水杨酸(共19种)的更多相互作用(完整)数据,请访问HSDB记录页面。 |
| 其他信息 |
4-氨基水杨酸是一种氨基苯甲酸,是水杨酸在4位被氨基取代后形成的。它是一种抗结核药物。它属于氨基苯甲酸类化合物,也是酚类化合物。其功能与水杨酸相关。它是4-氨基水杨酸(1-)的共轭酸。
它是一种抗结核药物,常与异烟肼联合使用。该药物的钠盐比游离酸耐受性更好。 氨基水杨酸是具有抗结核活性的对氨基苯甲酸(PABA)类似物。氨基水杨酸通过与对氨基苯甲酸(PABA)竞争叶酸合成酶,从而抑制结核分枝杆菌的生长和繁殖,最终导致细胞死亡,发挥其抑菌作用。 氨基水杨酸钠是氨基水杨酸的钠盐形式,是具有抗结核活性的对氨基苯甲酸(PABA)类似物。氨基水杨酸钠通过与对氨基苯甲酸(PABA)竞争叶酸合成酶,从而抑制结核分枝杆菌的生长和繁殖,最终导致细胞死亡,发挥其抑菌作用。 氨基水杨酸钠是一种抗结核药物,常与异烟肼联合使用。该药物的钠盐比游离酸耐受性更好。 药物适应症 用于治疗结核病 当由于耐药性或耐受性原因无法制定有效的治疗方案时(参见第4.4节),Granupas适用于成人和28天及以上儿童的耐多药结核病联合治疗方案。应参考抗菌药物合理使用的官方指南。 作用机制 氨基水杨酸对结核分枝杆菌的抑菌作用主要通过两种机制实现。首先,氨基水杨酸抑制叶酸合成(与抗叶酸化合物无协同作用)。对氨基苯甲酸与蝶呤合成酶的结合是叶酸合成的第一步。氨基水杨酸与蝶呤合成酶的亲和力高于对氨基苯甲酸,能有效抑制叶酸的合成。由于细菌无法利用外源叶酸,细胞生长和繁殖速度减慢。其次,氨基水杨酸可能抑制细胞壁成分分枝杆菌素的合成,从而降低结核分枝杆菌对铁的吸收。 氨基水杨酸的抗菌活性具有高度特异性,除结核分枝杆菌以外的其他微生物不受影响。大多数非结核分枝杆菌不受该药物抑制。 氨基水杨酸是对氨基苯甲酸的结构类似物,其作用机制与磺胺类药物非常相似。由于磺胺类药物对结核分枝杆菌无效,而氨基水杨酸对磺胺类药物敏感的细菌也无效,因此,各种微生物中负责叶酸生物合成的酶可能具有很强的区分能力,能够区分各种类似物和真正的代谢物。 治疗用途 抗结核药物 实验用途:降脂药。6克,服用4周。结果表明,它可以降低升高的血清甘油三酯水平以及升高的血清胆固醇水平。 单独使用时,有时可以成功控制结核病……但会出现耐药性,而且毒性限制了剂量。因此,对氨基水杨酸几乎总是与其他1或2种抗结核药物联合使用。……对氨基水杨酸可增强其他药物的疗效,并延缓耐药性的出现。 氨基水杨酸……具有强效的降血脂作用,可降低胆固醇和甘油三酯水平。然而,由于胃肠道反应,其耐受性较差。 有关对氨基水杨酸(共15种)的更多治疗用途(完整)数据,请访问HSDB记录页面。 药物警告 任何情况下,如果溶液颜色比新配制的溶液颜色深,都不得使用。 ……应在给药后24小时内配制钙、钾、钠盐溶液。 对于某些似乎具有种族特异性的各种缺陷(例如红细胞葡萄糖-6-磷酸脱氢酶缺乏症),不同的药物会引起溶血。其中最重要的是呋喃妥因、氨基水杨酸…… 对于肾功能受损或其他血浆浓度控制机制障碍的患者,该药物可引起高钙血症。它还可能导致尿路结石。 /CA SALT/ 氨基水杨酸或其盐类最常见的不良反应是胃肠道紊乱,包括恶心、呕吐、腹痛、腹泻和厌食。氨基水杨酸罕见地引起消化性溃疡和胃出血。部分患者可通过与食物同服氨基水杨酸盐来减轻胃肠道不良反应;然而,症状可能严重到需要停药。服用氨基水杨酸或其盐类的患者偶尔也会出现维生素B12、叶酸、铁和脂质吸收不良,这可能是由于肠蠕动增强所致。制造商指出,对于服用氨基水杨酸超过1个月的患者,应考虑使用维生素B12进行维持治疗。 有关P-氨基水杨酸(共12条)的更多药物警告(完整)数据,请访问HSDB记录页面。 药效学 氨基水杨酸是一种抗分枝杆菌药物,通常与其他抗结核药物(最常用的是异烟肼)联合使用,用于治疗由敏感结核杆菌菌株引起的各种活动性结核病。氨基水杨酸临床药理学的两个主要考虑因素是:在酸性条件下会迅速产生有毒的非活性代谢物,以及游离药物的血清半衰期仅为1小时。氨基水杨酸对结核分枝杆菌具有抑菌作用(抑制细菌繁殖而不杀死细菌)。它还能抑制细菌对链霉素和异烟肼产生耐药性。 |
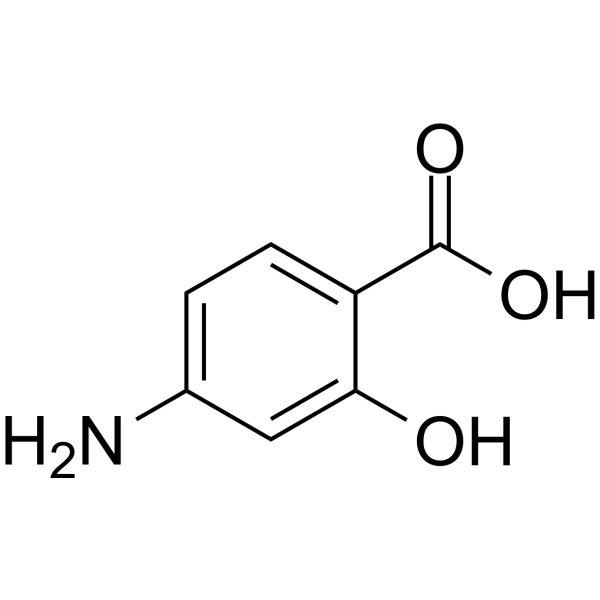
| 分子式 |
C7H7NO3
|
|---|---|
| 分子量 |
153.1354
|
| 精确质量 |
153.042
|
| CAS号 |
65-49-6
|
| 相关CAS号 |
Sodium 4-aminosalicylate dihydrate;6018-19-5;4-Aminosalicylic acid hemicalcium;133-15-3
|
| PubChem CID |
4649
|
| 外观&性状 |
MINUTE CRYSTALS FROM ALC
WHITE, OR NEARLY WHITE, BULKY POWDER NEEDLES, PLATES FROM ALC-ETHER A reddish-brown crystalline powder is obtained on recrystallization from ethanol-ether. |
| 密度 |
1.5±0.1 g/cm3
|
| 沸点 |
380.8±32.0 °C at 760 mmHg
|
| 熔点 |
135-145 °C(lit.)
|
| 闪点 |
184.1±25.1 °C
|
| 蒸汽压 |
0.0±0.9 mmHg at 25°C
|
| 折射率 |
1.691
|
| LogP |
1.14
|
| tPSA |
83.55
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
1
|
| 重原子数目 |
11
|
| 分子复杂度/Complexity |
160
|
| 定义原子立体中心数目 |
0
|
| SMILES |
O([H])C1C([H])=C(C([H])=C([H])C=1C(=O)O[H])N([H])[H]
|
| InChi Key |
WUBBRNOQWQTFEX-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C7H7NO3/c8-4-1-2-5(7(10)11)6(9)3-4/h1-3,9H,8H2,(H,10,11)
|
| 化学名 |
4-amino-2-hydroxybenzoic acid
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~100 mg/mL (~653.00 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (16.32 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (16.32 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (16.32 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.5300 mL | 32.6499 mL | 65.2997 mL | |
| 5 mM | 1.3060 mL | 6.5300 mL | 13.0599 mL | |
| 10 mM | 0.6530 mL | 3.2650 mL | 6.5300 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。