| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
|
| 靶点 |
Androgen receptor (AR)
Androgen Receptor (AR): Cyproterone Acetate acts as a competitive AR antagonist, binding to human AR with Ki = 0.2 nM (competitive binding assay in [1]); it also exhibits weak AR agonist activity at high concentrations (>100 nM) [1] - Estrogen Receptor (ER): Cyproterone Acetate binds to human ERα with IC50 = 50 nM (transcriptional inhibition assay in [1]); no binding to ERβ (Ki > 1000 nM) [1] |
|---|---|
| 体外研究 (In Vitro) |
在中等高剂量下,部分激动剂醋酸环丙孕酮表现出对 AR 的激动作用,EC50 为 4.0 μM[1]。通过上调死亡受体 5,醋酸环丙孕酮可增加 TRAIL 诱导的雄激素非依赖性前列腺癌细胞凋亡[3]。用醋酸环丙孕酮(0、1、10或50 μM)预处理24小时,然后再暴露24小时镉,明显降低了大鼠肝上皮细胞系对镉的敏感性[4]。
1. 甾体受体活性([1]): - AR拮抗:醋酸环丙孕酮(0.01–100 nM)抑制CV-1细胞(转染AR+ARE-荧光素酶)中DHT诱导的AR转录活性: - 1 nM:荧光素酶活性降低50%;10 nM:抑制90%(EC50=0.5 nM)。 - 弱ER激动:100 nM 醋酸环丙孕酮诱导ERα依赖转录(ERE-荧光素酶),活性为17β-雌二醇(100%激活)的20%[1] 2. 前列腺癌细胞凋亡([3]): - PC-3细胞(雄激素非依赖性CRPC):醋酸环丙孕酮(1–20 μM)单独处理无抗增殖活性(20 μM时活力>90%);与100 ng/mL TRAIL联用: - 10 μM:凋亡率从TRAIL单独处理的15%升至65%(Annexin V-FITC染色)。 - 蛋白质印迹法:死亡受体5(DR5)蛋白上调3.2倍,切割型caspase-8上调2.8倍[3] 3. 肝细胞镉耐受性([4]): - 大鼠肝上皮细胞(RLECs):醋酸环丙孕酮(1–5 μM)预处理24小时,降低氯化镉(CdCl₂,5 μM)诱导的细胞毒性: - 5 μM:细胞活力(MTT)从Cd单独处理的40%升至85%;细胞内Cd蓄积减少60%(原子吸收光谱法)。 - 金属硫蛋白(MT)mRNA上调4.5倍(qPCR)[4] |
| 体内研究 (In Vivo) |
注射0.08 mg/g醋酸环丙孕酮的成年雄性C57 BL/6J小鼠肾上腺皮质束状带和网状带细胞脂质含量增加[5]。
1. 前列腺癌疗效([2]): 250–300 g雄性哥本哈根大鼠接种Dunning R3327前列腺肿瘤细胞,随机分为对照、醋酸环丙孕酮10 mg/kg/天、20 mg/kg/天组: - 20 mg/kg/天(皮下注射,28天):肿瘤体积较对照减少60%;血清PSA降低75%(ELISA)。 - 肿瘤组织:增殖标志物Ki-67阳性率降低55%,凋亡标志物TUNEL阳性率升高40%[2] 2. 肾上腺皮质影响([5]): 200–220 g雄性SD大鼠口服醋酸环丙孕酮(5、10 mg/kg/天)30天: - 10 mg/kg/天:肾上腺皮质束状带厚度减少30%(H&E染色);血清皮质醇降低45%(放射免疫法RIA)。 - 球状带和网状带无变化[5] 3. 肝脏镉耐受性([4]): 180–200 g雄性Wistar大鼠口服醋酸环丙孕酮5 mg/kg/天预处理7天,再腹腔注射CdCl₂ 2 mg/kg/天14天: - 肝脏Cd浓度较单独Cd组减少50%;肝脏MT蛋白上调3倍(蛋白质印迹法)。 - 血清ALT/AST无变化(无肝损伤)[4] |
| 酶活实验 |
1. AR竞争结合实验([1]):
1. 重组AR制备:人AR配体结合域(LBD,氨基酸660–919)在大肠杆菌中表达,镍亲和层析纯化(250 mM咪唑洗脱)。 2. 反应体系:200 μL体系含50 mM Tris-HCl(pH7.4)、10%甘油、0.5 nM [³H]-DHT、100 ng AR-LBD及醋酸环丙孕酮(0.001–100 nM,冷竞争剂)。 3. 孵育与分离:4°C孵育2小时;葡聚糖包被活性炭(1%活性炭、0.1%葡聚糖)去除未结合[³H]-DHT,3000×g离心10分钟。 4. 检测:液体闪烁计数器检测上清放射性,Cheng-Prusoff方程计算Ki=0.2 nM[1] 2. ERα转录活性实验([1]): 1. 细胞转染:CV-1细胞接种于24孔板,共转染ERα表达质粒+ERE-荧光素酶报告基因(海肾荧光素酶为内参)。 2. 药物处理:转染24小时后,用醋酸环丙孕酮(1–1000 nM)+1 nM 17β-雌二醇(ER激动剂)处理16小时。 3. 检测:裂解细胞,luminometer检测荧光素酶活性;ERα抑制的IC50=50 nM[1] |
| 细胞实验 |
背景:几乎所有的前列腺癌症死亡都是由于在前列腺癌症细胞从最初由雄激素受体阻断诱导的凋亡和/或生长抑制中逃脱后获得去势耐受表型。TNF-相关凋亡诱导配体(TRAIL)是一种有吸引力的癌症治疗剂,因为它对正常细胞的毒性最小,并且在肿瘤细胞中具有显著的凋亡活性。然而,包括癌症在内的大多数局部癌症对TRAIL诱导的细胞凋亡具有抗性,从而在癌症细胞中产生诱导TRAIL敏感性的治疗挑战。本文报道了抗雄激素类固醇环丙酮乙酸酯对TRAIL诱导的雄激素受体阴性前列腺癌症细胞凋亡的影响。[3]
方法:采用膜联蛋白V/碘化丙啶标记法和聚ADP核糖聚合酶切割法检测细胞凋亡。通过定量实时聚合酶链式反应和蛋白质印迹测定来测定基因和蛋白质表达的变化。醋酸环丙孕酮对基因启动子活性的影响通过萤光素酶报告基因测定法测定。[3] 结果:醋酸环丙酮显著增加雄激素受体阴性人前列腺癌症PC-3和DU145细胞对TRAIL诱导的细胞凋亡的敏感性,但对永生化人前列腺基质PS30细胞和人胚肾HEK293细胞没有影响,而对AR拮抗剂比卡鲁胺没有影响。对TRAIL诱导的细胞凋亡途径的进一步研究表明,醋酸环丙孕酮通过选择性增加死亡受体5(DR5)mRNA和蛋白质表达来发挥其作用。醋酸环丙酮处理也增加了DR5基因启动子的活性,这可以通过DR5基因开启子中转录因子CCAAT增强子结合蛋白同源蛋白(CHOP)的共有结合结构域的突变来消除。醋酸环丙孕酮以浓度和时间依赖的方式增加CHOP的表达,内质网应激抑制剂4-苯基丁酸可以阻断醋酸环丙酮诱导的CHOP和DR5的上调。更重要的是,CHOP的siRNA沉默显著降低了前列腺癌症细胞中醋酸环丙孕酮诱导的DR5上调和TRAIL敏感性。[3] 结论:我们的研究显示了醋酸环丙孕酮对前列腺癌症细胞凋亡途径的新作用,并增加了TRAIL与醋酸环丙酮联合治疗去势抵抗性前列腺癌症的可能性。[3] 1. CRPC细胞凋亡实验([3]): - 细胞培养:PC-3细胞接种于6孔板(2×10⁵细胞/孔),用含10%胎牛血清的RPMI 1640培养基,37°C、5% CO₂培养。 - 药物处理:醋酸环丙孕酮(1–20 μM)预处理24小时,再加入100 ng/mL TRAIL处理12小时;对照组加入0.1% DMSO。 - 检测: 1. 凋亡:Annexin V-FITC/PI染色(流式细胞术)定量凋亡细胞。 2. 蛋白表达:蛋白质印迹法检测DR5、切割型caspase-8、β-肌动蛋白(内参)[3] 2. 肝细胞镉耐受实验([4]): - 细胞培养:RLECs接种于96孔板(5×10³细胞/孔),用含10%胎牛血清的DMEM培养基培养至80%融合。 - 药物处理:醋酸环丙孕酮(1–5 μM)预处理24小时,再暴露于5 μM CdCl₂ 48小时。 - 检测: 1. 活力:MTT实验(570 nm吸光度)。 2. Cd蓄积:原子吸收光谱法(细胞裂解液)。 3. MT mRNA:qPCR(GAPDH为内参)[4] |
| 动物实验 |
Dissolved in ethanol; 0.2 mg /kg/day; s.c administration
Castrate male SD rat Adult male C57 BL/6J mice injected with 0.08 mg/g of cyproterone acetate showed an increase in cellular lipid content in the zona fasciculata and zona reticularis of the adrenal cortex. In the perimedullary region, densely staining cells aggregated to form scattered nodules. At the ultrastructural level, in the zona glomerulosa and zona fasciculata the number and size of lipid droplets increased. An apparent decrease in mitochondrial number and size and a noticeable diminution in the amount of smooth endoplasmic reticulum were also noted in the zona fasciculata. It is suggested that cyproterone acetate may have blocked steroid synthesis and increased adrenal cholesterol deposits.[5] 1. Prostate Cancer Xenograft Protocol ([2]): - Animal Selection: 8-week old male Copenhagen rats (250–300 g, n=6/group) randomized to control、Cyproterone Acetate 10 mg/kg、20 mg/kg. - Model Induction: 1×10⁷ Dunning R3327 prostate tumor cells suspended in 0.2 mL PBS + 50% Matrigel, subcutaneous injection into right flank. - Drug Preparation: Cyproterone Acetate dissolved in ethanol (10%) + sesame oil (90%) to 1 mg/mL (10 mg/kg) and 2 mg/mL (20 mg/kg). - Administration: Subcutaneous injection (10 mL/kg) once daily for 28 days; control received vehicle. - Detection: Tumor volume measured twice weekly (volume = length×width²/2); rats euthanized, serum for PSA ELISA, tumor for Ki-67/TUNEL staining [2] 2. Rat Cadmium Exposure Protocol ([4]): - Animal Selection: 6-week old male Wistar rats (180–200 g, n=8/group) randomized to control、Cd alone、Cyproterone Acetate + Cd. - Drug Preparation: Cyproterone Acetate suspended in 0.5% CMC to 0.5 mg/mL; CdCl₂ dissolved in normal saline to 0.2 mg/mL. - Administration: - Pre-treatment: Cyproterone Acetate oral gavage (10 mL/kg, 5 mg/kg/day) for 7 days. - Cd exposure: Intraperitoneal injection of CdCl₂ (10 mL/kg, 2 mg/kg/day) for 14 days; control received 0.5% CMC + normal saline. - Detection: Rats euthanized, liver for Cd quantification and MT Western blot, serum for ALT/AST [4] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Cyproterone acetate is completely absorbed after oral administration. Approximately 60% is excreted via bile, and 33% via the kidneys. After oral administration, cyproterone acetate is completely absorbed over a wide dose range. Following administration of two 50 mg cyproterone acetate tablets, peak serum drug concentrations are reached approximately 3 hours later, at approximately 285 ng/mL. Subsequently, serum drug concentrations typically decline over 24 to 120 hours, with a terminal half-life of 43.9 ± 12.8 hours. The total clearance of cyproterone acetate is 3.5 ± 1.5 mL/min/kg. The absolute bioavailability of cyproterone acetate is almost 100% (88% of the dose). A portion of the drug is excreted unchanged via bile. The majority of the dose is excreted as metabolites, with a urine-to-bile ratio of 3:7. Cyproterone acetate is almost completely bound to plasma albumin. Approximately 3.5% to 4% of the drug exists in free form. Because protein binding is nonspecific, changes in sex hormone-binding globulin (SHBG) levels do not affect the pharmacokinetics of cyproterone acetate. For more complete data on absorption, distribution, and excretion of cyproterone acetate (6 items in total), please visit the HSDB record page. Metabolism/Metabolites Primarily metabolized in the liver. Cyproterone acetate is metabolized by the CYP3A4 enzyme to the active metabolite 15β-hydroxycyproterone acetate, which retains anti-androgenic activity but has reduced progestin activity. The metabolic pathways of cyproterone acetate are diverse, including hydroxylation and conjugation reactions. The main metabolite in human plasma is the 15β-hydroxy derivative. Biological Half-Life The plasma half-lives after oral or intramuscular administration are 38 hours and 96 hours, respectively. The half-life of renal and bile excretion is 1.9 days. The elimination rate of plasma metabolites is similar (half-life 1.7 days). The terminal half-life is 43.9 ± 12.8 hours. Oral absorption: -Rats: After oral administration of 10 mg/kg cyproterone acetate, the peak plasma concentration (Cmax) was 2.8 μg/mL after 2 hours; the oral bioavailability was 70% (compared to intravenous administration)[2]. -Humans: After oral administration of 50 mg, the peak plasma concentration (Cmax) was 3.5 μg/mL after 3 hours; the bioavailability was 65%[2]. -Metabolism: It is mainly metabolized in the liver by CYP3A4 to the active metabolite cyproterone acetate (half-life in rats is 6.5 hours); no other active metabolites were detected[2]. - Distribution: It accumulates extensively in the prostate (4.0 times the plasma concentration) and adrenal glands of rats (3.5 times the plasma concentration) [5] . - Excretion: 60% of the dose is excreted in feces as glucuronide conjugates, and 30% is excreted in urine (mainly as metabolites) [2] |
| 毒性/毒理 (Toxicokinetics/TK) |
Interactions
When cyproterone acetate is administered at a therapeutic dose of 100 mg three times daily, it may inhibit CYP2C8. Thiazolidinediones (such as the antidiabetic drugs pioglitazone and rosiglitazone) are substrates for CYP2C8 (elevated blood concentrations of these antidiabetic drugs may require dose adjustment). Alcohol appears to reduce the efficacy of cyproterone acetate, therefore it is ineffective in patients with chronic alcoholism. Because statins and cyproterone acetate share the same metabolic pathway, the risk of statin-related myopathy or rhabdomyolysis may increase when HMG-CoA inhibitors (statins), which are primarily metabolized by CYP3A4, are co-administered with high doses of cyproterone acetate. Drugs acting on androgen receptors can alter opioid transmission in the central nervous system. To investigate the direct interactions, researchers examined whether the binding of [3H]diprenorphine to the mouse meninges was affected by cyproterone acetate (a progesterone derivative with anti-androgenic activity), flutamide (a nonsteroidal anti-androgen), 5α-dihydrotestosterone, and progesterone. The results showed that only cyproterone acetate inhibited the binding of [3H]diprenorphine (IC50 = (1.62 ± 0.33) × 10⁻⁶ M), without altering its binding rate. These results indicate that the binding of cyproterone acetate to opioid receptors is independent of its classical androgen intracellular receptor effects. Non-human toxicity values Rat intraperitoneal injection LD50 565 mg/kg Mouse intraperitoneal injection LD50 3300 mg/kg 1. In vitro toxicity ([4]): Cyproterone acetate (1–20 μM) showed no cytotoxicity to normal human prostate epithelial cells (RWPE-1) and hepatocytes (HepG2); survival rate >90% compared with the control group (MTT method) [4] 2. In vivo toxicity ([4][5]): - Adrenal toxicity ([5]): 10 mg/kg/day (30 days) reduced adrenal cortisol secretion in rats by 45%, but no adrenal necrosis was observed (H&E staining). - Liver safety ([4]): Rats treated with 5 mg/kg/day + CdCl₂ showed normal ALT/AST; no liver fibrosis was observed (Masson staining). - Reproductive toxicity: 20 mg/kg/day (28 days) reduced testicular weight in rats by 20%, but no spermatogenesis disorder was observed [2]. 3. Clinical toxicity ([2]): - Common side effects: gynecomastia (25%), fatigue (20%), decreased libido (15%); most were grade 1-2. - Serious adverse events: 3% of prostate cancer patients experienced grade 3 liver injury (ALT > 3 times the upper limit of normal); no grade 4/5 toxicity was observed [2]. 4. Plasma protein binding rate: binding rate with human plasma albumin and α1-acid glycoprotein >99% [2]. |
| 参考文献 |
|
| 其他信息 |
Therapeutic Uses
Androgen antagonist; antitumor drug; male contraceptive; synthetic progestin. Cyproterone acetate is indicated for/controlling severe hypersexuality and/or paraphilia in adult men. Cyproterone acetate is indicated for/treating patients with prostate cancer who: (1) suppress “attacks” induced by initial gonadotropin-releasing hormone analogue therapy; (2) provide long-term palliative care in cases where gonadotropin-releasing hormone analogue therapy or surgery is contraindicated, intolerant, or preferred to be treated with oral medication; and (3) treat hot flashes in patients receiving gonadotropin-releasing hormone analogue therapy or who have undergone orchiectomy. Diane-35 (cyproterone acetate/ethinylestradiol) is recommended for use in women only for the treatment of: (a) severe acne unresponsive to long-term oral antibiotic therapy; (b) moderate to severe hirsutism. Although Diane-35 also has oral contraceptive effects, it should not be used solely for female contraception but should be reserved for the treatment of androgen-dependent disorders in women. Drug Warnings Direct hepatotoxicity, including jaundice, hepatitis, and liver failure, has been observed in patients treated with cyproterone acetate. Deaths have also been reported at doses of 100 mg and above. Most reported deaths occurred in men with advanced prostate cancer. Toxicity is dose-related and typically occurs several months after treatment initiation. Liver function tests should be performed before and regularly during treatment, and upon the appearance of any symptoms or signs suggestive of hepatotoxicity. If hepatotoxicity is diagnosed, ciprofloxacin should be discontinued unless the hepatotoxicity can be explained by other causes, such as metastatic disease; in such cases, ciprofloxacin should only be continued if the expected benefit outweighs the risk. In rare cases, benign and malignant liver tumors have been observed after ciprofloxacin use, which can lead to life-threatening intraperitoneal hemorrhage. Liver tumors should be included in the differential diagnosis if severe upper abdominal discomfort, hepatomegaly, or signs of intraperitoneal hemorrhage occur. Thromboembolic events have been reported in patients using ciprofloxacin, but a causal relationship has not been established. Patients with a history of arterial or venous thrombosis/thromboembolism (e.g., deep vein thrombosis, pulmonary embolism, myocardial infarction), a history of cerebrovascular accident, or advanced malignancy have an increased risk of further thromboembolic events and may be at risk of disease recurrence during treatment with cyproterone acetate. For patients with a history of thromboembolism, sickle cell anemia, or severe diabetes with vascular changes, the risk-benefit ratio for each case must be carefully weighed before prescribing cyproterone acetate. For more complete data on drug warnings for cyproterone acetate (17 in total), please visit the HSDB record page. Pharmacodynamics Cyproterone acetate is an antiandrogen. It inhibits the effects of testosterone (and its metabolite dihydrotestosterone) on tissues. It works by blocking androgen receptors, preventing androgens from binding to them, thereby inhibiting luteinizing hormone (and consequently lowering testosterone levels). 1. Drug background ([2]): Cyproterone acetate is a synthetic steroidal antiandrogen with weak estrogenic activity, and has been approved for the treatment of metastatic prostate cancer, hirsutism and severe acne [2]. 2. Mechanism of action ([1][3]): - Antiandrogen: Competes with dihydrotestosterone (DHT) for the binding site of androgen receptor (AR) (Ki=0.2 nM), inhibiting AR nuclear translocation and transcription of target genes (PSA, TMPRSS2) [1]. - Enhances apoptosis: Upregulates DR5 in castration-resistant prostate cancer (CRPC) cells, making them more sensitive to TRAIL-induced caspase-8 activation [3]. - Adrenal regulation: Inhibits the function of the zona fasciculata of the adrenal cortex and reduces cortisol secretion [5] 3. Treatment indications ([2]): Approved for the treatment of metastatic castration-resistant prostate cancer (mCRPC) (50 mg twice daily) and androgen-dependent dermatitis (25 mg once daily) [2] . 4. FDA warning ([2]): The US FDA has included a rare but serious warning about liver damage on the label of cyproterone acetate, requiring regular monitoring of liver function [2] |
| 分子式 |
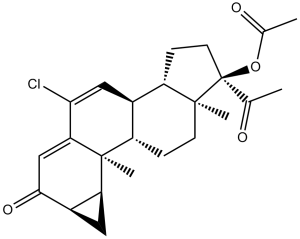
C24H29CLO4
|
|---|---|
| 分子量 |
416.94
|
| 精确质量 |
416.18
|
| 元素分析 |
C, 69.14; H, 7.01; Cl, 8.50; O, 15.35
|
| CAS号 |
427-51-0
|
| 相关CAS号 |
Cyproterone acetate-d3;2376035-90-2
|
| PubChem CID |
9880
|
| 外观&性状 |
Crystals from diisopropyl ether
White crystalline powder |
| 密度 |
1.3±0.1 g/cm3
|
| 沸点 |
525.9±50.0 °C at 760 mmHg
|
| 熔点 |
200-201ºC
|
| 闪点 |
177.6±29.1 °C
|
| 蒸汽压 |
0.0±1.4 mmHg at 25°C
|
| 折射率 |
1.582
|
| LogP |
3.28
|
| tPSA |
60.44
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
29
|
| 分子复杂度/Complexity |
903
|
| 定义原子立体中心数目 |
8
|
| SMILES |
C[C@@]12C(C(Cl)=C[C@]3([H])[C@]2([H])C[C@@]4(C)[C@@]3([H])CC[C@]4(OC(C)=O)C(C)=O)=CC([C@H]5[C@@H]1C5)=O
|
| InChi Key |
UWFYSQMTEOIJJG-FDTZYFLXSA-N
|
| InChi Code |
InChI=1S/C24H29ClO4/c1-12(26)24(29-13(2)27)8-6-16-14-10-20(25)19-11-21(28)15-9-18(15)23(19,4)17(14)5-7-22(16,24)3/h10-11,14-18H,5-9H2,1-4H3/t14-,15+,16-,17-,18-,22-,23-,24-/m0/s1
|
| 化学名 |
(2aR,3aS,3bS,3cS,5aS,6R,8aS,8bR)-6-acetyl-10-chloro-3b,5a-dimethyl-2-oxo-2,2a,3,3a,3b,3c,4,5,5a,6,7,8,8a,8b-tetradecahydrocyclopenta[a]cyclopropa[g]phenanthren-6-yl acetate
|
| 别名 |
Cyprone, Cyprohexal, Ciproterona, Cyproteronum,Androcur, Cyprostat,Cyproteron, Procur, Neoproxil, Siterone.CYPROTERONE ACETATE; 427-51-0; Androcur; Cyproteroneacetate; Cyproterone 17-O-acetate; Cyproteron acetate; Cyproteron-R acetate; Cyprostat;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (6.20 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (6.20 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (6.20 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 2 mg/mL (4.96 mM) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,可将100 μL 20.0 mg/mL 澄清 EtOH 储备液加入400 μL PEG300 中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL 生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 5 中的溶解度: ≥ 2 mg/mL (4.96 mM) (饱和度未知) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将100μL 20.0mg/mL澄清EtOH储备液加入到900μL 20%SBE-β-CD生理盐水中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 6 中的溶解度: ≥ 2 mg/mL (4.96 mM) (饱和度未知) in 10% EtOH + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.0 mg/mL 澄清乙醇储备液加入到 900 μL 玉米油中并混合均匀。 配方 7 中的溶解度: 1%DMSO+30% polyethylene glycol+1%Tween 80: 20 mg/mL 配方 8 中的溶解度: 12.5 mg/mL (31.02 mM) in Cremophor EL (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3984 mL | 11.9921 mL | 23.9843 mL | |
| 5 mM | 0.4797 mL | 2.3984 mL | 4.7969 mL | |
| 10 mM | 0.2398 mL | 1.1992 mL | 2.3984 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。