| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 5g |
|
||
| Other Sizes |
|
| 靶点 |
Hydroquinidine is a class Ia antiarrhythmic drug. The study mentions it inhibits the transient outward potassium current (Ito) of the cardiac action potential, which is implicated in Brugada syndrome. [2]
|
|---|---|
| 体内研究 (In Vivo) |
这项临床研究证明了长期氢奎尼丁治疗对短QT综合征(SQTS)患者的体内(人体)疗效。在17名接受治疗的患者中,氢奎尼丁(平均剂量584 ± 53 毫克/天)显著延长了QTc间期,从基线331 ± 3毫秒延长至391 ± 5毫秒(平均延长60 ± 6毫秒;p < 0.001)。
在对15名长期治疗患者(平均6 ± 1年)的匹配周期分析中,发生危及生命的心律失常事件(LAE:心脏骤停或心源性猝死)的患者比例从治疗前相等时期的40%降至治疗期间的0%(p = 0.03)。每名患者的LAE数量从0.73 ± 0.30次降至0次(p = 0.026)。 在另一项针对16名既往心脏骤停存活的SQTS患者的分析中,LAE的年发生率从未治疗时的12%(82人年发生10次事件)降至接受氢奎尼丁治疗期间的0%(44人年发生0次事件)(p = 0.028)。[1] 这项临床研究(人体)评估了氢奎尼丁在Brugada综合征(BrS)患者中的疗效。 在接受电生理(EP)指导治疗的无症状且可诱发室性心动过速/心室颤动(VT/VF)的BrS患者(n=29)中,氢奎尼丁(平均剂量 609 ± 89 毫克/天)在22名患者(76%)中预防了VT/VF的诱发。[2] 在接受长期氢奎尼丁治疗(平均随访 17 ± 13 个月)的21名无症状、可诱发患者中,17名(81%)保持无症状。两名患者在随访期间出现晕厥;一例与QTc间期显著延长(530 ms)和记录到的非持续性VT发作有关,另一例原因不明但可能与不依从用药有关。另外两名患者因晚期胃肠道不耐受而停用氢奎尼丁。[2] 在另一组10名无症状、可诱发、因持续可诱发或早期不耐受而接受植入式心律转复除颤器(ICD)治疗(而非长期氢奎尼丁)的患者中,平均随访13 ± 8个月期间发生了一次适当的ICD电击。[2] 在四名有多次适当ICD电击史的症状性BrS患者中,长期氢奎尼丁治疗在平均14 ± 8个月的随访期内,所有病例均预防了电击复发和VT/VF发作。其中一名患者记录到室性心律失常连发显著减少。[2] |
| 药代性质 (ADME/PK) |
该研究监测了氢化奎尼丁的血浆浓度。接受治疗的患者的平均血浆浓度为 2.7 ± 3.3 µmol/L。通常的治疗浓度范围(通过荧光法测定)为 3 至 6 µmol/L。
根据血浆浓度和电生理检查结果调整剂量。标准剂量为缓释制剂,每次 300 mg,每日两次(600 mg/天)。如果室性心动过速/室颤仍可诱发且血浆浓度低于 3 µmol/L,则剂量增加至 900 mg/天。[2] |
| 毒性/毒理 (Toxicokinetics/TK) |
本研究中,氢化奎尼丁治疗总体耐受性良好。17例患者中有2例(12%)因胃肠道不耐受(腹泻)在一周内停止治疗。治疗期间未观察到致心律失常事件。研究指出,其耐受性优于既往一项关于布鲁加达综合征的研究,这可能是由于本研究的平均日剂量较低(584 ± 53 mg/天 vs. 738 ± 25 mg/天)。 [1]
试验期间,35例患者中有7例(20%)出现药物不耐受,主要表现为腹泻,其中2例患者因此停用氢奎尼丁。 1例患者在治疗一周后出现可逆性氢奎尼丁诱发性肝炎(转氨酶升高3倍),伴有恶心和乏力,因此停药。[2] 1例患者在开始服用氢奎尼丁一个月后出现晕厥,伴有QTc间期显著延长至530毫秒(基线值为425毫秒),并记录到单形性室性心动过速发作。这被认为是一种可能的致心律失常事件。仅在该患者中观察到QTc间期>500毫秒。 [2] 总体而言,35 名患者中有 5 名(14%)因心脏外副作用(胃肠道不耐受或肝炎)而停止服用氢化奎尼丁。[2] |
| 参考文献 | |
| 其他信息 |
氢化奎尼丁正在临床试验 NCT00927732(氢化奎尼丁与安慰剂治疗布鲁加达综合征患者的比较)中进行研究。
另见:氢化奎宁(注释已移至此处)。 氢化奎尼丁 是一种奎尼丁衍生物,目前用于治疗短QT综合征 (SQTS),这是一种罕见的遗传性心律失常疾病,其特征是心电图上QT间期异常缩短,并伴有较高的猝死风险。 该研究方案包括以3 mg/kg/天的剂量开始使用氢化奎尼丁,并逐渐增加剂量,直至QTc间期>360 ms。平均维持剂量为 584 ± 53 mg/天(约 8 mg/kg/天)。 该研究得出结论,长期使用氢化奎尼丁可有效延长 QT 间期,并预防高危短 QT 综合征 (SQTS) 患者发生危及生命的心律失常事件。研究表明,该药物可作为辅助治疗,以减少植入式心脏复律除颤器 (ICD) 的放电次数,或在 ICD 禁忌、不可用或患者拒绝使用时作为替代疗法。 作者倡导在全球范围内继续提供奎尼丁类药物,用于治疗危及生命的遗传性心律失常,例如 SQTS。[1] 本研究使用盐酸氢化奎尼丁(缓释制剂)治疗布鲁加达综合征 (BrS)。 [2] 布鲁格达综合征 (BrS) 的可能机制是抑制瞬时外向钾电流 (Ito),从而重新平衡心外膜动作电位 1 期的电流,并减少 2 期折返性心律失常的基质。[2] 治疗方案包括初始电生理检查。随后开始服用氢化奎尼丁,并在至少两周后重复进行电生理检查,以评估室性心动过速/室颤诱发性的抑制情况。如果患者无法诱发室性心动过速/室颤且耐受药物,则继续长期治疗。[2] 氢化奎尼丁治疗后观察到的心电图变化包括 QRS 波时限(延长 13 ± 12 ms)和 QTc 间期(延长 10 ± 7 ms)轻微但显著延长。在35例患者中,有12例(34%)观察到特征性的Brugada型ST段抬高减轻或恢复正常,但这与室性心动过速/室颤诱发性的预防无关。[2]该研究提示,对于部分可诱发心律失常的无症状BrS患者,氢化奎尼丁可能是一种替代ICD植入的预防性治疗方法,但强调需要密切监测QTc间期和血浆药物浓度,以评估患者的耐受性和依从性。[2] |
| 分子式 |
C20H26N2O2
|
|---|---|
| 分子量 |
326.4326
|
| 精确质量 |
326.199
|
| CAS号 |
1435-55-8
|
| 相关CAS号 |
1476-98-8 (mono-hydrochloride);1668-97-9 (mono-hydrochloride)
|
| PubChem CID |
91503
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.2±0.1 g/cm3
|
| 沸点 |
498.4±30.0 °C at 760 mmHg
|
| 熔点 |
169-170 ºC
|
| 闪点 |
255.2±24.6 °C
|
| 蒸汽压 |
0.0±1.3 mmHg at 25°C
|
| 折射率 |
1.626
|
| LogP |
3.77
|
| tPSA |
45.59
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
4
|
| 重原子数目 |
24
|
| 分子复杂度/Complexity |
432
|
| 定义原子立体中心数目 |
4
|
| SMILES |
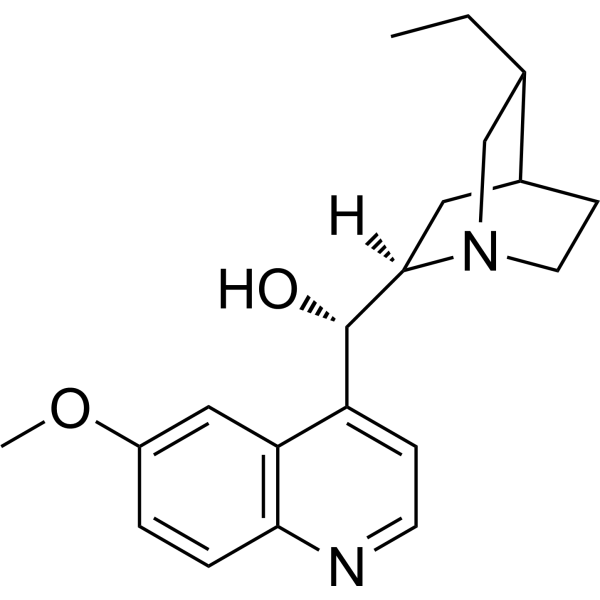
CC[C@H]1CN2CC[C@H]1C[C@@H]2[C@H](C3=C4C=C(C=CC4=NC=C3)OC)O
|
| InChi Key |
LJOQGZACKSYWCH-LHHVKLHASA-N
|
| InChi Code |
InChI=1S/C20H26N2O2/c1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18/h4-6,8,11,13-14,19-20,23H,3,7,9-10,12H2,1-2H3/t13-,14-,19+,20-/m0/s1
|
| 化学名 |
(S)-[(2R,4S,5R)-5-ethyl-1-azabicyclo[2.2.2]octan-2-yl]-(6-methoxyquinolin-4-yl)methanol
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: (1). 本产品在运输和储存过程中需避光。 (2). 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~20.83 mg/mL (~63.81 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (6.37 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (6.37 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (6.37 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0634 mL | 15.3172 mL | 30.6344 mL | |
| 5 mM | 0.6127 mL | 3.0634 mL | 6.1269 mL | |
| 10 mM | 0.3063 mL | 1.5317 mL | 3.0634 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。