| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
β-catenin-responsive transcription (CRT)
The target of iCRT3 is the β-catenin/T-cell factor (TCF) complex, specifically inhibiting the interaction between β-catenin and TCF1; [2] The target of iCRT3 is the Wnt/β-catenin signaling pathway, acting as a small-molecule inhibitor of β-catenin-responsive transcription; [3] The target of iCRT3 is the Wnt/β-catenin signaling pathway; [1] |
|---|---|
| 体外研究 (In Vitro) |
iCRT3 抑制对 Wnt 和 β-catenin 敏感的转录。 iCRT3 显着降低了 TOP Flash 活动和 NTSR1 级别。 iCRT3可以显着抵消神经降压素(NTS)和Wnt3a的抗凋亡作用[1]。与 DMSO 对照相比,长期 iCRT3 维持的细胞表现出经典多能性的表达增加,但同时分化标记物和 T 细胞因子 (TCF) 靶基因的表达减少[2]。 iCRT3 治疗在 12.5、25、50 和 75 μM 剂量下,TNF-α 水平分别降低 14.7%、18.5%、44.9% 和 61.3%。与媒介物相比,iCRT3 疗法的 IκB 水平呈剂量依赖性上升[3]。
1. 对胶质母细胞瘤细胞中Wnt/β-catenin信号通路的抑制作用:iCRT3通过药理抑制Wnt/β-catenin通路,降低胶质母细胞瘤细胞中NTSR1的mRNA和蛋白表达水平[1] 2. 对小鼠胚胎干细胞(mESCs)中β-catenin/TCF1相互作用的抑制作用:iCRT3特异性抑制β-catenin与TCF1的相互作用,不影响β-catenin与TCF3的结合。这种抑制作用降低了TCF依赖性靶基因(如Cdx1、Axin2)的转录活性,延缓了mESCs的分化。经iCRT3处理14天后,mESCs的自我更新能力增强,维持较高的Nanog和Rex1表达水平,自发分化减少。在视黄酸(RA)诱导的分化模型中,iCRT3使mESCs能够抵抗分化,提高Nanog-GFP阳性细胞比例,增强集落形成效率。此外,iCRT3还可增加β-catenin/Oct4复合物的形成,该复合物与基础状态多能性相关[2] 3. 对脂多糖(LPS)刺激的RAW264.7巨噬细胞炎症反应的调控作用:用不同浓度的iCRT3预处理(50分钟)RAW264.7巨噬细胞后,加入LPS(1 ng/ml)刺激。iCRT3呈剂量依赖性抑制LPS诱导的Wnt/β-catenin通路激活(通过TOP-TK-Luc报告基因活性检测)和肿瘤坏死因子-α(TNF-α)的产生(通过酶联免疫吸附试验(ELISA)检测)。iCRT3还呈剂量依赖性抑制LPS诱导的IκB降解(通过蛋白质印迹法(Western blotting)分析),且不影响细胞活力(通过MTS实验检测,与未处理细胞相比活力维持在100%)[3] |
| 体内研究 (In Vivo) |
iCRT3 治疗可显着降低肿瘤生长速度。 iCRT3 的肿瘤抑制功能始终与增殖标志物 Ki67 指数的下降相关[1]。与载体组相比,10 mg/kg iCRT3 治疗组的 IL-6 水平降低了 82.9%。在假手术中,检测不到 IL-1β 水平;然而,在脓毒症小鼠中,当给予 5 和 10 mg/kg iCRT3 时,它们分别达到 371 pg/mL 和下降 30.2% 和 53.2%。用 5 和 10 mg/kg 剂量的 iCRT3 治疗的这些脓毒症小鼠的 AST 水平分别比用载体治疗的动物低 15.4% 和 44.2%。与媒介物组相比,用 10 mg/kg iCRT3 治疗后,肺形态得到改善,微观退化更少。将 iCRT3 治疗动物的肺组织与媒介物组进行比较时,凋亡细胞减少了 92.7%[3]。
1. 对胶质母细胞瘤模型肿瘤生长的抑制作用:iCRT3通过药理抑制Wnt/β-catenin信号通路,在体内抑制胶质母细胞瘤的肿瘤生长[1] 2. 对C57BL/6小鼠脓毒症诱导的炎症反应和器官损伤的缓解作用:对雄性C57BL/6小鼠进行盲肠结扎穿孔(CLP)诱导脓毒症,在CLP后5小时腹腔注射iCRT3(5 mg/kg或10 mg/kg体重)。CLP后20小时,iCRT3呈剂量依赖性降低血浆中促炎细胞因子(IL-6、TNF-α、IL-1β)和器官损伤标志物(AST、ALT、LDH)的水平。组织学分析显示,iCRT3(10 mg/kg)改善了肺组织完整性,降低了肺损伤评分,减少了肺胶原沉积(通过Masson三色染色),并抑制了肺细胞凋亡(通过TUNEL染色检测)。此外,iCRT3下调了肺组织中IL-6、TNF-α、IL-1β、中性粒细胞趋化因子(MIP-2、KC)的mRNA表达,降低了肺髓过氧化物酶(MPO)活性,从而减轻了中性粒细胞浸润[3] |
| 酶活实验 |
β-catenin-TCF报告活性测定[3]
RAW264.7细胞在转染前一天以1.24 × 105个细胞/ ml的密度接种。细胞与250 ng TOP-TK-Luc或TOP-TK-Luc和25 ng pRL-TK报告质粒短暂共转染,使用Lipofectamine 3000 Reagent,按照制造商的说明。转染24 h后,用iCRT3或对照物预处理细胞50 min,然后用LPS (1 ng/ml)刺激24 h。转染后48小时裂解细胞,根据制造商的说明,用双荧光素酶报告基因检测系统测定荧光素酶活性。TOP-TK-Luc包含最优位点,TOP-TK-Luc包含位于萤火虫荧光素酶报告基因上游的突变tcf结合位点。将TOP和FOP萤火虫荧光素酶活性归一化为来自共转染pRL-TK质粒的Renilla荧光素酶活性,作为转染效率的内部对照。所有实验都进行了至少两次的三次重复。 |
| 细胞实验 |
荧光素酶报告试验[1]
细胞在24孔板中以4 × 105个细胞/孔的大小进行镀膜,用Lipofectamine 2000 瞬时转染TopFlash(0.5µg)和Renilla报告基因(0.05µg)。A172或U87细胞中分别加入NTS、Wnt3a、SR48692和iCRT3处理24 h。收集细胞,转染2天后测定荧光素酶活性。采用双荧光素酶报告基因检测系统测定荧光素酶活性。 细胞增殖和细胞凋亡试验[1] 将细胞接种到96孔板中,每孔密度为5 × 103个细胞,在指定处理的培养基中再孵育48小时。根据制造商的说明,分别使用Cell Counting kit-8和Caspase-Glo 3/7检测试剂盒进行细胞活力和细胞凋亡检测。 对于长期培养,在DMSO或iCRT3的干条件下(血清加LIF),将细胞以有限的稀释度在6孔或96孔板中进行多次传代(14 d),每天更换培养基。每代进行AP染色以监测相对多能性水平。使用的小分子包括10µM iCRT3和1µM XAV939,用DMSO稀释。L- wnt3a和对照L细胞是R.T. Moon赠送的。[2] 1. 胶质母细胞瘤细胞实验:用iCRT3(浓度未明确)处理胶质母细胞瘤细胞以抑制Wnt/β-catenin通路。处理后提取细胞总RNA和蛋白,通过实时定量聚合酶链反应(qPCR)检测NTSR1的mRNA表达水平,通过Western blotting分析NTSR1的蛋白表达水平[1] 2. 小鼠胚胎干细胞(mESCs)实验: - β-catenin/TCF转录活性实验:将mESCs(NG4-TOPluc)转染TOPFlash报告基因质粒和内参pRL-TK质粒。转染后,用iCRT3(浓度未明确)处理细胞,并在含血清和白血病抑制因子(S+LIF)或含血清和RA(S+RA)的培养基中培养,检测荧光素酶活性以评估对β-catenin/TCF依赖性转录活性的抑制作用[2] - 流式细胞术分析:在S+LIF培养基中用iCRT3处理TNGA(Nanog报告基因)和Rex1-GFP mESCs 14天,通过流式细胞术检测Nanog-GFP和Rex1-GFP的表达水平,评估自我更新能力。在分化抵抗实验中,无LIF条件下用iCRT3处理mESCs 6天或RA诱导分化时处理48小时,通过流式细胞术分析Nanog-GFP阳性细胞比例[2] - qPCR分析:收集经iCRT3处理四代或RA诱导分化过程中的mESCs,提取总RNA并逆转录为cDNA,以肌动蛋白(actin)为内参,通过qPCR检测多能性标志物(Nanog、Oct4、Sox2)、分化标志物及TCF靶基因(Cdx1、Axin2)的mRNA表达水平[2] - 集落形成效率(CFE)实验:将mESCs用RA联合iCRT3或二甲基亚砜(DMSO)预处理48小时后,以有限稀释法接种到不含抑制剂的S+LIF培养基中,再培养48小时,量化集落数量、集落面积和碱性磷酸酶(AP)活性以评估集落形成效率[2] - 免疫共沉淀(CoIP)实验:制备RA诱导分化过程中经iCRT3处理的mESCs总细胞裂解液,用抗β-catenin、TCF1和TCF3的抗体进行CoIP实验,检测β-catenin与TCF1/TCF3的相互作用,通过Western blotting进行检测和密度分析[2] 3. RAW264.7巨噬细胞实验: - 报告基因实验:将RAW264.7细胞共转染β-catenin/TCF应答报告基因TOP-TK-Luc(或对照FOP-TK-Luc)和内参pRL-TK。转染后,用不同浓度的iCRT3预处理细胞50分钟,再用LPS(1 ng/ml)刺激24小时,检测荧光素酶活性以评估Wnt/β-catenin通路激活情况[3] - TNF-α ELISA实验:用不同浓度的iCRT3预处理RAW264.7细胞50分钟,再用LPS(1 ng/ml)刺激4小时,收集细胞上清液,通过ELISA检测TNF-α水平[3] - MTS细胞活力实验:用不同浓度的iCRT3处理RAW264.7细胞,通过MTS实验检测细胞活力,以未处理细胞的活力为100%[3] - IκB Western blotting实验:用不同浓度的iCRT3预处理RAW264.7细胞50分钟,再用LPS(1 ng/ml)刺激15分钟,制备总细胞裂解液,用抗IκB和actin的抗体进行Western blotting,检测IκB降解情况[3] |
| 动物实验 |
溶于 5% DMSO 生理盐水中;5 和 10 mg/kg;腹腔注射。
使用 C57BL/6 小鼠 A172 细胞建立皮下异种移植瘤模型,并测定 SR48692 和 iCRT3 的抗肿瘤作用。将 2 × 10⁶ 个 A172 细胞皮下接种于 NOD-SCID BALB/c 小鼠右背部。每 4 天记录一次原发肿瘤的生长情况。当肿瘤生长至约 200 mm³ 时,每三周腹腔注射一次 SR48692 (10 mg/kg) 和 iCRT3 (5 mg/kg)(溶于 PBS)。对照组小鼠注射含 5% (v/v) DMSO 的空白 PBS。肿瘤体积采用以下公式计算:体积 = 肿瘤长度 × 宽度²/2。药物治疗 24 天后处死小鼠。肿瘤切除后进行石蜡包埋,并采用免疫组织化学方法分析Ki67染色。[1] 小鼠随机分为三组:假手术组(n = 5只小鼠)、载体组和治疗组(每组n = 8只小鼠)。iCRT3用细胞培养级100% DMSO配制成50 mg/ml的储备液。iCRT3用含5% DMSO的无菌生理盐水稀释,配制成5 mg/kg和10 mg/kg体重(BW)的iCRT3浓度溶液。盲肠结扎穿孔术(CLP)后5小时,使用25G × 7/8″皮下注射针,腹腔注射200 μl含5% DMSO的生理盐水(载体)或5 mg/kg或10 mg/kg体重的iCRT3溶液。进行动物实验的研究人员对治疗分配情况不知情,以消除任何偏倚。[3] 1. 胶质母细胞瘤体内实验:动物模型建立(例如,细胞接种方法、细胞数量)、iCRT3剂量、给药途径和频率等具体细节未明确说明。采用iCRT3对Wnt/β-catenin通路进行药理学抑制,以观察其对肿瘤生长的抑制作用[1] 2. C57BL/6小鼠脓毒症模型实验: - 动物准备:选用雄性C57BL/6小鼠作为实验动物,随机分为假手术组、CLP+载体组和CLP+iCRT3治疗组(5 mg/kg和10 mg/kg体重,每组5-8只小鼠)[3] - 脓毒症模型建立:CLP组小鼠行盲肠结扎穿刺术以诱导脓毒症,假手术组仅行开腹手术,不行盲肠结扎穿刺术[3] - 给药:CLP术后5 h,各治疗组小鼠腹腔注射iCRT3。将 iCRT3 溶解于 5% DMSO 生理盐水中,剂量按规定配制。对照组小鼠注射等体积的 5% DMSO 生理盐水 [3] - 样本采集与检测:盲肠结扎穿孔术 (CLP) 后 20 小时,采集小鼠血液样本,检测血浆细胞因子水平和器官损伤标志物。取肺组织进行组织学分析(苏木精-伊红染色、马松三色染色)、TUNEL 染色、细胞因子和趋化因子 mRNA 表达的 qPCR 分析以及髓过氧化物酶 (MPO) 活性检测 [3] |
| 参考文献 |
|
| 其他信息 |
背景/目的:神经降压素(NTS)是一种肠道激素,它通过与主要受体NTSR1结合,在癌症进展中发挥重要作用。保守的Wnt/β-catenin信号通路通过激活β-catenin/T细胞因子(TCF)复合物并随后调控一系列靶基因,从而调节细胞增殖和分化。本研究旨在揭示NTS/NTSR1信号通路与Wnt/β-catenin信号通路之间的潜在联系。方法:我们采用基因沉默、药理学抑制和功能获得性实验以及生物信息学分析等方法,揭示NTS/NTSR1信号通路与Wnt/β-catenin信号通路之间的联系。本研究采用两种抑制剂在体内评估靶向NTS/NTSR1信号通路或Wnt/β-catenin通路的效率。结果显示,NTS/NTSR1可诱导丝裂原活化蛋白激酶(MAPK)和NF-κB通路的激活,进而促进Wnt蛋白(包括Wnt1、Wnt3a和Wnt5a)的表达。同时,在胶质母细胞瘤细胞中,Wnt通路激活剂Wnt3a可上调NTSR1的mRNA和蛋白表达水平,而Wnt抑制剂iCRT3则可下调NTSR1的mRNA和蛋白表达水平。此外,NTS/NTSR1 或 Wnt/β-catenin 信号通路的药理学抑制可抑制体外和体内的肿瘤生长。结论:这些结果揭示了胶质母细胞瘤细胞中 NTS/NTSR1 和 Wnt/β-catenin 信号通路之间存在正反馈环路,这可能对肿瘤发展至关重要,并为胶质母细胞瘤的治疗提供潜在靶点。[1]
小鼠胚胎干细胞 (mESC) 的自我更新或分化为各种细胞谱系的能力受信号通路和由 Nanog、Oct4 和 Sox2 组成的核心多能性转录网络 (PTN) 的调控。Wnt/β-catenin 通路通过缓解 T 细胞因子 TCF3 介导的 PTN 抑制来促进多能性。然而,β-catenin 作为转录激活因子与 TCF1 协同作用如何影响 mESC 的命运仍不清楚。本文研究表明,TCF1介导的转录在分化的小鼠胚胎干细胞(mESCs)中上调,而β-catenin/TCF1相互作用的化学抑制可改善长期自我更新能力并增强功能性多能性。TCF1基因缺失通过延迟多能性退出抑制分化,并赋予mESCs一种与高Nanog表达的自我更新mESCs极为相似的转录谱。综上所述,我们的数据表明,β-catenin在调控mESCs中的功能具有高度的上下文特异性,其与TCF1的相互作用促进分化,这进一步凸显了理解其单个蛋白质-蛋白质相互作用如何驱动干细胞命运的必要性。[2] Wnt/β-catenin通路参与调控多种感染性和炎症性疾病中的炎症反应。脓毒症是一种由感染引起的炎症反应失调导致的危及生命的疾病,目前尚无有效的治疗方法。近期研究发现,脓毒症中Wnt/β-catenin信号通路活性升高。然而,其在脓毒症相关炎症反应中的作用机制仍有待探索。本研究表明,抑制Wnt/β-catenin信号通路可减轻炎症并缓解脓毒症引起的器官损伤。我们利用体外LPS刺激的RAW264.7巨噬细胞,证实β-catenin反应性转录的小分子抑制剂iCRT3可显著降低LPS诱导的Wnt/β-catenin活性,并以剂量依赖的方式抑制TNF-α的产生和IκB的降解。腹腔注射iCRT3至盲肠结扎穿刺诱导脓毒症的C57BL/6小鼠后,可剂量依赖性地降低血浆中促炎细胞因子和器官损伤标志物的水平。iCRT3治疗可改善肺组织的完整性,并减少肺胶原沉积和细胞凋亡。此外,iCRT3 治疗还能降低脓毒症小鼠肺部细胞因子、中性粒细胞趋化因子以及髓过氧化物酶 (MPO) 的表达。基于这些发现,我们得出结论,靶向 Wnt/β-catenin 通路可能为脓毒症的治疗提供一种潜在的治疗方法。[3] 1. 背景:神经降压素 (NTS) 与其主要受体 NTSR1 结合,促进癌症进展。Wnt/β-catenin 通路通过激活 β-catenin/TCF 复合物来调节细胞增殖和分化。在胶质母细胞瘤细胞中,NTS/NTSR1 和 Wnt/β-catenin 信号通路之间存在正反馈环路,该环路参与肿瘤的发生发展。[1] 2. 作用机制:iCRT3 抑制 Wnt/β-catenin 信号通路,从而降低 NTSR1 的表达并抑制胶质母细胞瘤的生长。[1] 3.背景:Wnt/β-catenin信号通路调控小鼠胚胎干细胞(mESCs)的自我更新和分化。β-catenin可缓解TCF3介导的多能性转录网络(PTN)抑制,从而促进多能性;同时,β-catenin与TCF1的相互作用可促进mESC分化[2] 4. 作用机制:iCRT3特异性抑制β-catenin与TCF1的相互作用,降低分化促进靶基因的转录活性。它增强mESC的自我更新,延缓分化,并提高功能性多能性,且不影响β-catenin与TCF3的相互作用[2] 5. 背景:Wnt/β-catenin信号通路参与调控感染性和炎症性疾病中的炎症反应。脓毒症是由感染引起的炎症反应失调所致,在脓毒症中已检测到Wnt/β-catenin信号通路激活[3] 6.作用机制:iCRT3阻断Wnt/β-catenin信号通路,从而抑制NF-κB通路,减少促炎细胞因子和趋化因子的产生,减轻肺胶原沉积、细胞凋亡和中性粒细胞浸润,最终减轻脓毒症引起的炎症反应和器官损伤[3] 7. 治疗潜力:iCRT3通过靶向Wnt/β-catenin通路,具有治疗胶质母细胞瘤的潜在治疗价值[1] 8. 治疗潜力:iCRT3为研究β-catenin/TCF1相互作用在mESC命运调控中的作用提供了一种工具,并可能在干细胞研究中具有潜在的应用价值[2] 9.治疗潜力:利用iCRT3靶向Wnt/β-catenin通路可能是一种治疗脓毒症的潜在治疗方法[3] |
| 分子式 |
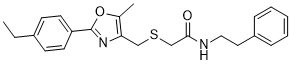
C23H26N2O2S
|
|
|---|---|---|
| 分子量 |
394.53
|
|
| 精确质量 |
394.171
|
|
| 元素分析 |
C, 70.02; H, 6.64; N, 7.10; O, 8.11; S, 8.13
|
|
| CAS号 |
901751-47-1
|
|
| 相关CAS号 |
|
|
| PubChem CID |
6622273
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
5.195
|
|
| tPSA |
80.43
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
4
|
|
| 可旋转键数目(RBC) |
9
|
|
| 重原子数目 |
28
|
|
| 分子复杂度/Complexity |
462
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
QTDYVSIBWGVBKU-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C23H26N2O2S/c1-3-18-9-11-20(12-10-18)23-25-21(17(2)27-23)15-28-16-22(26)24-14-13-19-7-5-4-6-8-19/h4-12H,3,13-16H2,1-2H3,(H,24,26)
|
|
| 化学名 |
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (6.34 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (6.34 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5347 mL | 12.6733 mL | 25.3466 mL | |
| 5 mM | 0.5069 mL | 2.5347 mL | 5.0693 mL | |
| 10 mM | 0.2535 mL | 1.2673 mL | 2.5347 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Proc Natl Acad Sci U S A.2011 Apr 12;108(15):5954-63. |
|---|
Proc Natl Acad Sci U S A.2011 Apr 12;108(15):5954-63. |
Proc Natl Acad Sci U S A.2011 Apr 12;108(15):5954-63. |
Effect of iCRT3 administration on systemic cytokine levels after CLP.Sci Rep.2017 Aug 23;7(1):9235. |
|---|
Effect of iCRT3 treatment on the expression of cytokines in the lungs after CLP.Sci Rep.2017 Aug 23;7(1):9235. |
Effect of iCRT3 treatment on the neutrophil infiltration in the lungs after CLP.Sci Rep.2017 Aug 23;7(1):9235. |