| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Rho-associated protein kinas/ROCK; norepinephrine transporter/NET
Rho kinase (Ki = 0.2 nM); norepinephrine transporter [1] Rho kinase; norepinephrine transporter [2] Rho kinase; norepinephrine transporter [3] |
|---|---|
| 体外研究 (In Vitro) |
体外活性:先前的研究表明,在细胞水平上,netarsudil 已被证明能够诱导肌动蛋白应力纤维的损失、细胞形状的改变、粘着斑的损失以及 TM 细胞的细胞外基质组成的变化。 Netarsudil(以前称为 AR-13324)是 ROCK 抑制剂,Ki 为 0.2-10.3 nM。它还抑制去甲肾上腺素转运活性,从而减少房水的产生。细胞测定:先前的研究表明,在细胞水平上,netarsudil 已被证明能够诱导肌动蛋白应力纤维的损失、细胞形状的改变、粘着斑的损失以及 TM 细胞的细胞外基质组成的变化。
对去核小鼠眼球进行体外灌注实验,使用甲磺酸奈他舒地尔(100 nM)处理后,与溶媒(0.001% DMSO)组相比,房水流出系数显著增加。C57BL/6小鼠(n=8)中,药物处理组的流出系数平均增幅具有统计学意义(P=0.006);CD1小鼠(n=6)中,同样观察到显著的流出系数增加(P=0.025)。在药物或溶媒灌注45-60分钟后,通过9个连续的压力梯度测定流速(Q)与压力(P)的关系,进而计算流出系数 [2] 以恒定压力(15 mmHg)对去核人眼球进行体外灌注,使用0.3 μM 奈他舒地尔-M1(活性代谢产物)处理3小时后,与基线相比,房水流出系数(C)显著增加51%(P<0.01),与配对溶媒对照组相比显著增加102%(P<0.01)。同时,施莱姆管(SC)内壁(IW)和巩膜外静脉(ESVs)的有效滤过长度百分比(PEFL)显著增加(分别为P<0.05和P<0.01)。在药物处理组眼中,巩膜外静脉的PEFL显著高于内壁(P<0.01),且与流出系数的百分比变化呈正相关(R²=0.58,P=0.01)。此外,与对照组相比,巩膜外静脉的横截面积(P<0.01)和邻管结缔组织(JCT)厚度(P<0.05)均显著增加 [3] |
| 体内研究 (In Vivo) |
动物功效研究发现,奈塔舒地尔的局部治疗能够影响小鼠常规流出道的近端部分(小梁网和施累姆斯管)和远端部分(巩膜内血管)。
在荷兰带兔中,甲磺酸奈他舒地尔(酯类化合物60,母体Rho激酶抑制剂29的前药)给药后,实现了有效且持续24小时的眼内压(IOP)降低 [1] 向10周龄C57小鼠和6-14周龄CD1小鼠(每组5只)的右眼局部给予10 μl 0.04%甲磺酸奈他舒地尔,与安慰剂(CF324-01)处理组相比,显著降低了眼内压(IOP)(不同品系的P值分别为P<0.05或P<0.01) [2] 向活体小鼠(n=8)的对侧眼玻璃体内预加载100 nM甲磺酸奈他舒地尔,在人工将眼内压升高至40 mmHg后,药物处理组的眼内压恢复能力增强。表征压力衰减速率的常数α与溶媒(0.001% DMSO)处理组相比显著增加(P<0.01) [2] 对活体C57小鼠进行局部甲磺酸奈他舒地尔处理后,通过光学相干断层扫描(OCT)成像观察到,处理后45分钟小梁网(TM)增宽,施莱姆管(SC)横截面积显著增加。同时,流出血管的散斑方差强度增加,传统流出组织中的示踪剂沉积增强,眼内压降低 [2] 在眼内压升高的活体小鼠中,局部给予甲磺酸奈他舒地尔(10 μl 0.04%)后,当眼内压被控制在10、15和30 mmHg时,施莱姆管腔的横截面积增加(P<0.05或P<0.01)。OCT成像显示,C57和CD1小鼠(n=11)的施莱姆管面积相对于基线(处理前10 mmHg)发生显著变化 [2] 对C57和CD1小鼠进行局部甲磺酸奈他舒地尔处理后,通过OCT散斑方差图像分析发现,参与房水流出的巩膜血管的横截面积和散斑方差强度在处理后30-60分钟增加(P<0.05) [2] |
| 酶活实验 |
Netarsudil(以前称为 AR-13324)是一种 ROCK 抑制剂,Ki 为 0.2-10.3 nM。此外,它还能抑制去甲肾上腺素转运活性,从而减少房水的产生。
在PDB中总共发现了23个ROCK结构。最大和最小分辨率分别为3.4Å和2.93Å。选择7个ROCK-I和2个ROCK-II非冗余结构用于结合测定。在测试的46种化合物(20种异喹啉、15种氨基呋咱、6种苯二氮卓、4种吲唑和1种酰胺)中,与Y-27632相比,34种化合物的ROCK-1对接得分显著更高(p<0.0001)。所有ROCKi类的平均对接得分均高于Y-27632(p<0.0001)。ROCK-I的异喹啉、氨基呋咱和苯二氮卓类化合物呈现最高对接得分的频率更高;以及ROCK-II的异喹啉和酰胺(补充图S2A)。ROCK-I和II平均对接得分最高的前十种化合物如补充图S2B所示。异喹啉类药物占前十个最高对接得分内药物的70%,其中三种化合物的对接得分强于Ş12。除Y-27632外,ROCK抑制剂之间没有显著差异。有趣的是,计算机分子对接模拟显示,大多数评估的分子,特别是异喹啉、苯二氮卓和酰胺类分子,对ROCK-1和ROCK-2的结合强度高于Y-27632(补充图S2B)。进行了计算机分子对接模拟,将PDB中发现的AR-13324和Y-27632抑制剂的异构体与高分辨率ROCK蛋白偶联。所有测试的AR-13324分子对ROCK-1和-2的对接得分都高于Y-27632。此外,异喹啉、苯二氮卓和酰胺类的PDB分子也显示出比Y-27632异构体更高的平均对接得分(补充图S2B)[3]。 |
| 细胞实验 |
先前的研究表明,netarsudil 可能会导致 TM 细胞的细胞外基质组成发生改变,以及粘着斑、肌动蛋白应力纤维和细胞形状的丧失。
|
| 动物实验 |
局部用药
\n眼压升高小鼠 \n兔眼压降低持续时间评估:对荷兰带状兔进行适当剂量的局部滴眼,给予甲磺酸奈他舒地尔(酯60)。在给药后24小时内的不同时间点测量眼压,以评估降眼压作用的持续时间[1] \n小鼠眼压降低评估:将10周龄的C57小鼠和6-14周龄的CD1小鼠按年龄和性别分组(每组5只)。每个品系分为两组:一组右眼局部滴用10 μl 0.04%的甲磺酸奈他舒地尔,另一组滴用10 μl安慰剂(CF324-01)眼药水。给药前测量双眼眼压,并使用 Mann-Whitney U 检验比较各组间的眼压变化值 [2] \n小鼠眼压恢复评估:将载体(0.001% DMSO)或 100 nM 的甲磺酸奈他舒地尔预先装入灌注针,并插入活体小鼠对侧眼的眼内。双眼在 15 mmHg 眼压下维持 30 分钟,以使药物/载体进入眼内,然后将眼压人为升高至 40 mmHg 并维持 5 分钟。储液罐关闭,但保持与压力传感器的连通,以监测双眼眼压随时间的变化。采用学生t检验(n=8)计算并比较各组间的速率常数α[2] \n小鼠离体房水流出率测定:将C57BL/6(n=8)和CD1(n=6)小鼠的成对去核眼球通过微针灌注甲磺酸奈他舒地尔或载体(0.001% DMSO)45-60分钟。随后,将眼球暴露于9个连续的压力阶跃,并使用iPerfusion系统测量流速(Q)与压力(P)的关系,以计算房水流出率。采用配对加权t检验分析房水流出率的百分比变化[2] \n小鼠示踪剂沉积评估:将荧光微珠加载到含有或不含甲磺酸奈他舒地尔的微针中。将C57和CD1小鼠(每组n=5)的成对眼球的前房进行插管,并以0.167 μl/min的恒定流速灌注1小时。小鼠继续饲养1小时后处死,并将前节平铺固定,通过落射荧光显微镜进行观察。对常规房水流出区域的荧光强度、宽度和面积进行定量分析,并使用学生t检验进行比较[2]。 \n小鼠常规房水流出组织OCT成像:对活体C57小鼠进行局部应用甲磺酸奈他舒地尔或安慰剂治疗。在治疗前和治疗后45分钟采集虹膜角膜角200次B扫描的平均OCT图像。使用Schlemm II软件分割巩膜下层(SC),并使用Schlemm III软件分析散斑方差图像,以量化SC面积、巩膜血管的散斑方差强度和小梁网(TM)宽度(每组n=5)。采用学生t检验进行统计分析[2] \n小鼠眼压升高SC OCT成像:C57和CD1小鼠(n=11)分别接受局部应用甲磺酸奈他舒地尔或安慰剂治疗。在治疗前和治疗后30-60分钟,将玻璃针插入前房,依次控制眼压为10、15和30 mmHg。在相同位置采集虹膜角膜角的OCT图像,并使用Schlemm II软件量化SC横截面积,结果以基线值(治疗前10 mmHg)为基准。采用Mann-Whitney U检验进行统计比较[2] \n人眼离体灌注:将成对的人眼(n=5)在恒定压力(15 mmHg)下灌注0.3 μM甲磺酸奈他舒地尔或载体溶液3小时。在灌注固定前,向灌注液中添加荧光微球以追踪流出模式。采用全局和共聚焦成像计算小梁网 (TM)、小梁网囊 (ESV) 和脊髓 (SC) 内壁 (IW) 的灌注效率 (PEFL)。通过共聚焦显微镜、光学显微镜和电子显微镜研究形态学变化。测量流出率随时间的变化,并量化包括小梁网囊横截面积和小梁网囊壁厚度在内的参数,并在各组之间进行比较 [3] |
| 药代性质 (ADME/PK) |
吸收
在18名健康受试者中,连续8天每日一次(早晨双眼各滴一滴)局部滴用0.02%奈他舒地尔滴眼液后,奈他舒地尔及其活性代谢物AR-13503的全身暴露情况显示,第1天和第8天给药后,血浆中未检测到奈他舒地尔(定量下限[LLOQ]为0.100 ng/mL)。仅在第8天给药后8小时,一名受试者的血浆中检测到活性代谢物,浓度为0.11 ng/mL。 消除途径 使用人角膜组织、人血浆、人肝微粒体及其S9组分进行体外代谢的临床研究表明,奈他舒地尔的代谢是通过酯酶活性进行的。后续未检测到奈他舒地尔酯酶代谢产物 AR-13503 的代谢。事实上,在 3 小时的孵育过程中,未在人血浆中检测到酯酶代谢。 分布容积 由于奈他舒地尔及其活性代谢产物具有高度蛋白结合率,预计其分布容积较小。 清除率 奈他舒地尔的清除率受其局部给药和吸收后血浆浓度低以及在人血浆中蛋白结合率高的影响。 代谢/代谢物 局部眼用给药后,奈他舒地尔在眼内经酯酶代谢为活性代谢物奈他舒地尔-M1(或AR-13503)。 生物半衰期 奈他舒地尔与人角膜组织体外孵育的半衰期为175分钟。 奈他舒地尔甲磺酸盐(酯60)是一种前药,可提高其母体Rho激酶抑制剂(化合物29)的生物利用度[1]。 |
| 毒性/毒理 (Toxicokinetics/TK) |
妊娠期和哺乳期用药
◉ 哺乳期用药概述 目前尚无关于哺乳期使用奈他舒地尔的信息。由于奈他舒地尔滴眼后在母亲体内吸收不良,因此不太可能对母乳喂养的婴儿产生不良影响。在获得更多数据之前,哺乳期应谨慎使用奈他舒地尔,尤其是在哺乳新生儿或早产儿时。为减少滴眼后进入母乳的药物量,请用手指按压眼角附近的泪管至少1分钟,然后用吸水纸巾吸去多余的药液。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对哺乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 |
| 参考文献 | |
| 其他信息 |
另见:奈他舒地尔(具有活性成分);拉坦前列素;奈他舒地尔甲磺酸盐(成分)。
奈他舒地尔甲磺酸盐(曾用名 AR-13324)是一种 Rho 激酶和去甲肾上腺素转运体的双重抑制剂,用于治疗开角型青光眼和眼高压[1][2][3] 奈他舒地尔甲磺酸盐对 Rho 激酶的抑制作用可改善房水通过小梁网的流出,从而降低眼内压,这是抗青光眼治疗的关键策略[1] 在活体小鼠眼中,奈他舒地尔甲磺酸盐可影响传统房水流出道的近端(小梁网和施莱姆氏管)和远端(巩膜内血管),通过扩张小梁网和增加施莱姆氏管横截面积来增加流出组织的灌注,这与房水流出率增加和散斑方差强度增强有关。房水流出血管扩张,眼压降低[2] 在人眼中,netarsudil mesylate 的作用机制涉及 JCT 的急性扩张和 ESV 的扩张,导致房水流出通过更大的 IW 和 ESV 区域重新分布,从而增加房水流出率[3] 这是首篇利用实时成像 (OCT) 技术展示 netarsudil mesylate 对活体眼内常规房水流出组织实时药物作用的报告,为开发用于监测青光眼治疗的临床友好型 OCT 平台铺平了道路[2] Netarsudil mesylate 是从 α-芳基-β-氨基异喹啉类似物中发现的,这些类似物被发现是强效的 ROCK 抑制剂,能够抑制去甲肾上腺素转运体,并提供更持久的眼压降低作用[1] |
| 分子式 |
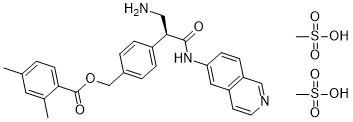
C30H35N3O9S2
|
|
|---|---|---|
| 分子量 |
645.74
|
|
| 精确质量 |
645.181
|
|
| 元素分析 |
C, 55.80; H, 5.46; N, 6.51; O, 22.30; S, 9.93
|
|
| CAS号 |
1422144-42-0
|
|
| 相关CAS号 |
Netarsudil hydrochloride;1253952-02-1;AR-13324 analog mesylate; 1422144-42-0; 1254032-66-0
|
|
| PubChem CID |
90410375
|
|
| 外观&性状 |
White to yellow solid powder
|
|
| tPSA |
220Ų
|
|
| 氢键供体(HBD)数目 |
4
|
|
| 氢键受体(HBA)数目 |
11
|
|
| 可旋转键数目(RBC) |
8
|
|
| 重原子数目 |
44
|
|
| 分子复杂度/Complexity |
770
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
S(C)(=O)(=O)O.S(C)(=O)(=O)O.O=C([C@@H](CN)C1C=CC(COC(C2C=CC(C)=CC=2C)=O)=CC=1)NC1C=CC2C=NC=CC=2C=1
|
|
| InChi Key |
QQDRLKRHJOAQDC-FBHGDYMESA-N
|
|
| InChi Code |
InChI=1S/C28H27N3O3.2CH4O3S/c1-18-3-10-25(19(2)13-18)28(33)34-17-20-4-6-21(7-5-20)26(15-29)27(32)31-24-9-8-23-16-30-12-11-22(23)14-24;2*1-5(2,3)4/h3-14,16,26H,15,17,29H2,1-2H3,(H,31,32);2*1H3,(H,2,3,4)/t26-;;/m1../s1
|
|
| 化学名 |
[4-[(2S)-3-amino-1-(isoquinolin-6-ylamino)-1-oxopropan-2-yl]phenyl]methyl 2,4-dimethylbenzoate;methanesulfonic acid
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (3.87 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (3.87 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (3.87 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5486 mL | 7.7431 mL | 15.4861 mL | |
| 5 mM | 0.3097 mL | 1.5486 mL | 3.0972 mL | |
| 10 mM | 0.1549 mL | 0.7743 mL | 1.5486 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Netarsudil lowered intraocular pressure (IOP) in both pigmented and nonpigmented mice.
Netarsudil mesylate enhanced IOP recovery in living mouse eyes.Eur J Pharmacol.2016 Sep 15;787:20-31. |
|---|
Netarsudil mesylate increased outflow facility in perfused mouse eyes ex vivo.Eur J Pharmacol.2016 Sep 15;787:20-31. |
Enhanced tracer deposition in outflow tissues of living mice subjected to netarsudil mesylate treatment.Eur J Pharmacol.2016 Sep 15;787:20-31. |
Netarsudil-induced changes in conventional outflow tissue morphology of living mice visualized by optical coherence tomography (OCT).Eur J Pharmacol.2016 Sep 15;787:20-31. |
|---|
Netarsudil increased cross-sectional area of Schlemms canal (SC) lumen in living mice with elevated intraocular pressure (IOP) visualized by optical coherence tomography (OCT).Eur J Pharmacol.2016 Sep 15;787:20-31. |
Netarsudil-induced changes in flow area and intensity in scleral vessels visualized on OCT speckle variance images.Eur J Pharmacol.2016 Sep 15;787:20-31. |