| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Potassium channel
Sulfonylurea receptor 2A/2B (SUR2A/SUR2B)-mediated ATP-sensitive potassium (KATP) channels[1] - cGMP-dependent protein kinase (PKG) [3] |
|---|---|
| 体外研究 (In Vitro) |
Nicorandil (SG-75) 激活由 Kir6.2 和磺酰脲受体 (SUR) 2A 或 2B 组成的 ATP 敏感 K+ 通道[1]。尼可地尔是一种用于治疗心绞痛的血管舒张药物。尼可地尔 (SG-75) 激活鸟苷酸环化酶,促进环 GMP (cGMP) 的合成。 cGMP 刺激蛋白激酶 G (PKG),后者磷酸化并抑制 GTPase RhoA,并降低 Rho 激酶活性。 Rho 激酶活性降低会促进肌球蛋白磷酸酶活性增加,从而降低平滑肌的钙敏感性。 PKG 还可以激活肌膜钙泵以去除活性钙。 PKG 对 K+ 通道起作用,增加 K+ 流出,产生的超极化会抑制电压门控钙通道。总体而言,这会导致平滑肌松弛和冠状血管舒张[2][3]。
在稳定表达SUR2A/Kir6.2或SUR2B/Kir6.2 KATP通道复合物的HEK293细胞中,尼可地尔(SG-75)(10-300 μM)以剂量依赖方式激活KATP通道。100 μM浓度时,SUR2A/Kir6.2介导的电流增加180%,SUR2B/Kir6.2介导的电流增加120%。该激活依赖SUR2A和SUR2B的核苷酸结合域,且SUR2A对药物更敏感[1] - 在分离的血管平滑肌细胞(VSMCs)中,尼可地尔(SG-75)(1-30 μM)通过cGMP-PKG信号通路抑制RhoA诱导的钙敏感性收缩。10 μM浓度时,收缩率抑制达55%,Western blot检测显示肌球蛋白轻链(MLC)磷酸化水平降低48%[3] |
| 体内研究 (In Vivo) |
尼可地尔(每天 2.5 毫克/公斤,口服)与氨氯地平(每天 5.0 毫克/公斤,口服)联合使用 3 天可显着防止这些改变,并使大鼠的酶活性恢复到接近正常水平。
在健康志愿者中,静脉注射尼可地尔(SG-75)(0.1-0.3 mg/kg)剂量依赖性扩张冠状动脉,增加冠脉血流量。0.3 mg/kg剂量时,冠脉血流量较基线增加70%,注射后5分钟血浆药物浓度达150 ng/mL并起效。其扩血管机制与硝酸酯类(释放NO)和激活KATP通道相关[2] |
| 酶活实验 |
尼可地尔激活由Kir6.2和磺酰脲受体(SUR) 2A或2B组成的atp敏感的K+通道。虽然SUR2A和SUR2B仅在其c端42个氨基酸(C42)上不同,并且具有相同的药物受体和核苷酸结合结构域(NBDs),但尼可地尔比SUR2A/Kir6.2通道更有效地激活SUR2B/Kir6.2通道。在这里,我们用膜片钳方法的内外结构分析了nbd在这些通道对尼可地尔的反应中的作用。NBD1 (K708A)和NBD2 (K1349A)的Walker A基序以及NBD2 (D1470N)的Walker B基序突变破坏了NBDs对核苷酸的结合和水解。实验用内源性ATP (1mm)进行。在SUR2A/Kir6.2通道中,K708A突变被消除,K1349A突变而非D1470N突变降低了对尼可地尔的敏感性。ADP (100 μM)显著增加了野生型通道对nicorandil的敏感性,而K1349A或D1470N突变则消除了这种敏感性。因此,SUR2A/Kir6.2通道对nicorandil的反应严重依赖于ATP- nbd1的相互作用,并由ATP或ADP与NBD2的相互作用促进。在SUR2B/Kir6.2通道中,K708A或K1349A突变部分抑制了对尼可地尔的反应,而双突变则消除了这种反应。D1470N突变也显著削弱了这种反应。ADP对通道无敏化作用。因此,NBD2水解ATP, NBD1和NBD2同样通过与ATP和ADP相互作用来促进反应,这解释了在ATP单独存在时,SUR2B/Kir6.2通道比SUR2A/Kir6.2通道对尼可地尔的敏感性更高。因此,C42调节两种nbd与细胞内核苷酸的相互作用。[1]
KATP通道激活检测:将编码SUR2A/Kir6.2或SUR2B/Kir6.2的质粒转染HEK293细胞,培养48小时后进行全细胞膜片钳记录。将梯度浓度(10-300 μM)的尼可地尔(SG-75)加入细胞外液,电压方案设定为钳制电位-70 mV,去极化至+40 mV(500 ms),复极化至-70 mV,将峰值电流幅度与对照组归一化,计算激活率[1] - PKG信号通路检测:用尼可地尔(SG-75)(1-30 μM)处理VSMCs 30分钟后裂解细胞,通过激酶检测试剂盒检测特异性PKG底物的磷酸化水平以评估PKG活性;采用pull-down实验检测RhoA活性,Western blot分析MLC磷酸化水平[3] |
| 细胞实验 |
KATP通道细胞实验:将转染后的HEK293细胞接种到盖玻片上,培养48小时。向记录槽中加入10 μM、50 μM、100 μM、300 μM的尼可地尔(SG-75),通过全细胞膜片钳记录,比较SUR2A/Kir6.2和SUR2B/Kir6.2通道的激活效率[1]
- 血管平滑肌细胞收缩实验:从大鼠主动脉分离VSMCs,接种到胶原蛋白包被的培养皿中。用尼可地尔(SG-75)(1 μM、10 μM、30 μM)预处理细胞1小时,再用RhoA激活剂刺激。通过相差显微镜观察并定量细胞收缩情况,收集细胞裂解液进行MLC磷酸化的Western blot分析[3] |
| 动物实验 |
2.5 mg/kg daily, p.o.
Rats In this study, researchers investigated the cardiovascular profile of nicorandil, an antianginal agent, in humans. Pharmacologically, nicorandil acts as both an adenosine triphosphate (ATP)-sensitive K+ (K(ATP)) channel opener and a nitrate. We examined which of these mechanistic components has a predominant vasodilatory effect at clinical doses. Fourteen patients underwent cardiac catheterization. The effects of the continuous intravenous infusion of nicorandil (12 mg/45 min) were examined in angiographically normal coronary arteries. Coronary vascular resistance was calculated from coronary artery diameter and coronary blood flow velocity measured using an intravascular Doppler catheter. We compared the hemodynamic responses to nicorandil with those to the intracoronary injection of nitroglycerin (250 microg) and papaverine (12 mg). The epicardial coronary arteries responded to nicorandil at the lowest plasma concentration examined (dilation of +14.0 +/- 3.3% at approximately 170 ng/ml), whereas dilation of the coronary resistance arteries (i.e., a decrease in coronary vascular resistance) took place only at higher concentrations (>200 ng/ml). Nitroglycerin caused no further changes in coronary artery diameter or coronary vascular resistance. Papaverine caused no further increase in coronary artery diameter, but markedly decreased coronary vascular resistance (1.6 +/- 0.3 to 0.4 +/- 0.1 mm Hg/ml/min; p < 0.05). Nicorandil significantly decreased pulmonary capillary wedge pressure (i.e., reduced cardiac preload) at a plasma level of >200 ng/ml, but did not change either systemic or pulmonary vascular resistance. Thus nicorandil preferentially dilated epicardial coronary arteries rather than coronary resistance arteries, and had a stronger effect on preload than on afterload. These changes in human coronary hemodynamics suggest that the nitrate actions of nicorandil as a coronary vasodilator predominate over those as a K(ATP) opener.[2] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Following oral administration, nicorandil is well absorbed from the gastrointestinal tract, with an oral bioavailability of 75%. Peak plasma concentration (Cmax) is reached within 30–60 minutes. The average Cmax is approximately 300 ng/ml. After twice-daily administration (10 or 20 mg), steady-state plasma concentrations of nicorandil are typically reached within 96–120 hours. The primary route of excretion is the kidneys; more than 60% of the administered dose is excreted in the urine 24 hours after administration. Only about 1% of nicorandil is excreted unchanged in the urine, with the remainder primarily as denitrification metabolites (9%) and their derivatives (e.g., nicotinic acid uric acid 6%, nicotinamide 1%, N-methylnicotinamide < 1%, nicotinic acid < 1%). Less than 2% of the administered dose is excreted via the biliary system. The apparent volume of distribution after oral (and intravenous) administration is approximately 1.0–1.4 L/kg body weight. The total clearance is approximately 1.15 L/min. Metabolism/Metabolites Niccolandil is primarily metabolized by the liver. The main biotransformation pathway of niccolandil is denitration, followed by nicotinamide metabolism. The main pharmacologically inactive denitration metabolite, 2-nicotinamide ethanol, is detectable in urine. Derivatives of denitration products obtained by nicotinamide metabolism include nicotinic acid, nicotinamide, N-methylnicotinamide, and nicotinic acid. Biological Half-Life The elimination half-life is approximately 1 hour. Plasma Concentration: In healthy individuals, intravenous injection of niccolandil (SG-75) (0.1–0.3 mg/kg) produces dose-dependent plasma concentrations, reaching a peak concentration of 150 ng/mL 5 minutes after injection of 0.3 mg/kg [2] |
| 毒性/毒理 (Toxicokinetics/TK) |
Protein Binding
Nicorandil binds to approximately 25% of human serum albumin and other plasma proteins. Human oral TDLo 195 mg/kg/1 year - Gastrointestinal: Other Changes Lancet., 352(1598), 1998 [PMID:9843111] Rat oral LD50 1220 mg/kg Yakkyoku. Pharmacy., 35(1627), 1984 Rat intraperitoneal LD50 1100 mg/kg Yakkyoku. Pharmacy., 35(1627), 1984 Rat subcutaneous LD50 1200 mg/kg Yakkyoku. Pharmacy, 35(1627), 1984 Rat intravenous LD50 502 mg/kg Sensory organs and special senses: tearing; eyes; behavior: changes in sleep duration (including changes in the righting reflex); lungs, pleural cavity, or respiration: Respiratory Stimulation Pharmacology and Therapeutics, 19(2561), 1991 |
| 参考文献 |
|
| 其他信息 |
Pharmacodynamics
Nicorandil is a potassium channel opener with nitrovasodilator (NO donor) action, thus dilating both arteries and veins. It sustainably dilates both resistance and conduction vessels, increasing coronary blood flow, but its effect on the coronary arteries does not involve coronary steal. Activation of potassium channels leads to smooth muscle cell hyperpolarization, subsequently causing arterial dilation and reduced afterload. Nicorandil reduces preload by increasing blood pooling in volume vessels through venous dilation. Overall, it improves blood flow and reduces infarct size by lowering end-diastolic pressure and reducing the extravascular components of vascular resistance. Open-label studies have shown that nicorandil treatment is effective for various types of angina. Nicotil (SG-75) is a dual-action drug that combines the properties of a nitrate-like (NO-releasing) and an ATP-sensitive potassium channel (KATP) opener [2] - Its clinical indications include the prevention and treatment of angina pectoris, exerting an antianginal effect by dilating coronary arteries and reducing myocardial oxygen consumption [2] - The drug activates KATP channels through the nucleotide-binding domains of SUR2A and SUR2B, with SUR2A-mediated channel activation being stronger [1] - In vascular smooth muscle, Nicotil (SG-75) promotes vasodilation by inhibiting RhoA-induced Ca²⁺ sensitization through the cGMP-PKG signaling pathway [3] |
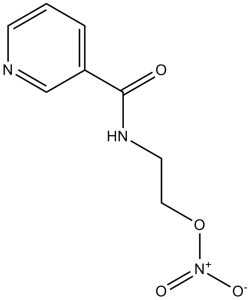
| 分子式 |
C8H9N3O4
|
|
|---|---|---|
| 分子量 |
211.17
|
|
| 精确质量 |
211.059
|
|
| 元素分析 |
C, 45.50; H, 4.30; N, 19.90; O, 30.30
|
|
| CAS号 |
65141-46-0
|
|
| 相关CAS号 |
Nicorandil-d4;1132681-23-2
|
|
| PubChem CID |
47528
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.3±0.1 g/cm3
|
|
| 沸点 |
456.7±25.0 °C at 760 mmHg
|
|
| 熔点 |
92ºC
|
|
| 闪点 |
230.0±23.2 °C
|
|
| 蒸汽压 |
0.0±1.1 mmHg at 25°C
|
|
| 折射率 |
1.548
|
|
| LogP |
0.72
|
|
| tPSA |
97.04
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
5
|
|
| 可旋转键数目(RBC) |
4
|
|
| 重原子数目 |
15
|
|
| 分子复杂度/Complexity |
228
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
LBHIOVVIQHSOQN-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C8H9N3O4/c12-8(7-2-1-3-9-6-7)10-4-5-15-11(13)14/h1-3,6H,4-5H2,(H,10,12)
|
|
| 化学名 |
2-(nicotinamido)ethyl nitrate
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (11.84 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (11.84 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7355 mL | 23.6776 mL | 47.3552 mL | |
| 5 mM | 0.9471 mL | 4.7355 mL | 9.4710 mL | |
| 10 mM | 0.4736 mL | 2.3678 mL | 4.7355 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。