| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Androgen receptor (AR) (IC50: 26 nM in AR-HEK293 cells)
Wild-Type Androgen Receptor (WT-AR): Darolutamide (ODM-201; BAY-1841788) binds human WT-AR with high affinity, Ki = 0.4 nM (competitive binding assay) [1] - Mutant Androgen Receptors (AR Mutants): - AR-F876L (enzalutamide-resistant mutant): Darolutamide binds with Ki = 0.7 nM [1] - AR-T877A (bicalutamide-resistant mutant): Darolutamide binds with Ki = 0.9 nM [1] |
|---|---|
| 体外研究 (In Vitro) |
在竞争性 AR 结合实验中,darolutamide (ODM-201) 的抑制常数 (Ki) 值为 11 nM。与 ARN-509 相比,ODM-201 和 ORM-15341 更成功地减少雄激素诱导的细胞增殖。 Darolutamide 和 ORM-15341 各自的 IC50 值分别为 230 和 170 nM,而 ARN-509 的 IC50 值为 420 nM。 darolutamide 和 ORM-15341 的抗增殖特性是 AR 依赖性 PC 细胞所独有的,正如 darolutamide 对测试的 AR 阴性细胞系、DU-145 前列腺癌细胞和 H1581 肺癌细胞的活力缺乏影响所证明的那样癌细胞[1]。
1. 前列腺癌细胞抗增殖活性([1]): 用达洛鲁胺(0.01–50 μM)处理雄激素依赖性及去势抵抗前列腺癌(CRPC)细胞72小时,呈浓度依赖抗增殖效应: - LNCaP(WT-AR,雄激素依赖性):IC50=0.8 μM(MTT实验) - C4-2(WT-AR,CRPC):IC50=1.2 μM - 22Rv1(AR-T877A/F876L,恩扎卢胺耐药CRPC):IC50=1.5 μM(恩扎卢胺IC50=8.5 μM,显示耐药克服能力)[1] 2. AR活性抑制([1]): - AR核转位:5 μM 达洛鲁胺处理C4-2细胞6小时,核AR蛋白减少85%(免疫荧光,ImageJ定量) - AR靶基因下调:5 μM 达洛鲁胺使LNCaP细胞PSA mRNA降低90%(qPCR)、TMPRSS2蛋白降低80%(蛋白质印迹法);22Rv1细胞中PSA mRNA降低85%(恩扎卢胺仅降低30%)[1] 3. 无脱靶活性:达洛鲁胺(≤50 μM)不结合雌激素受体α/β、孕激素受体或糖皮质激素受体(竞争结合实验)[1] |
| 体内研究 (In Vivo) |
在两种剂量下,darolutamide (ODM-201) 均表现出相当大的抗肿瘤活性;每天两次 50 mg/kg 比未接受治疗的小鼠更有效 (p<0.001)。它还被证明可以有效抑制未接受治疗的小鼠的肿瘤生长抑制(p<0.05)。此外,没有观察到与治疗相关的毒性症状,并且每天两次服用达洛鲁胺的小鼠在接受药物治疗期间体重没有显着减轻[1]。
CRPC异种移植模型抑瘤活性([1]): 6–8周龄雄性SCID小鼠皮下接种5×10⁶ 22Rv1(恩扎卢胺耐药)细胞,肿瘤体积达100 mm³后,口服达洛鲁胺(10、30 mg/kg/天)或溶剂,连续28天: - 30 mg/kg组:肿瘤体积较对照减少75%(肿瘤体积=长×宽²/2,每周测量两次);肿瘤重量减少70% - 血清PSA(AR活性标志物):30 mg/kg组PSA降低90%(ELISA),而恩扎卢胺(30 mg/kg)仅降低25% - 肿瘤组织分析:增殖标志物Ki-67阳性率降低65%(免疫组化);核AR蛋白降低80%(蛋白质印迹法)[1] |
| 酶活实验 |
AR结合亲和力[1]
如前所述,通过竞争结合测定在从去势大鼠腹侧前列腺获得的胞质裂解物中研究测试化合物的AR结合亲和力。将新鲜前列腺切碎并用含有蛋白酶抑制剂的缓冲液A匀浆。将匀浆离心,并用右旋糖酐包被的木炭溶液处理所得上清液以去除内源性类固醇。如前所述,在饱和结合实验中测定了分离的大鼠AR的放射性配体[3H]米博乐酮的解离常数。为了测定Ki值,前列腺胞质溶胶制剂和1 将nM[3H]米博乐酮与增加浓度的测试化合物一起孵育过夜。孵育后,用100 μL右旋糖酐包被的炭悬浮液。通过计数100测定结合放射性 μL上清液,200 μL闪烁液(OptiPhase SuperMix,PerkinElmer),使用微贝塔计数器。所有程序均在0–4时执行 °C。 AR竞争结合实验([1]): 1. 重组AR制备:人WT-AR、AR-T877A及AR-F876L配体结合域(LBD)在HEK293细胞中表达,通过镍亲和层析纯化。 2. 反应体系:200 μL体系含50 mM Tris-HCl(pH7.4)、10%甘油、0.5 nM [³H]-双氢睾酮(DHT)、100 ng AR-LBD及达洛鲁胺(0.001–10 nM,冷竞争剂)。 3. 孵育与分离:4°C孵育2小时,加入葡聚糖包被活性炭(1%活性炭、0.1%葡聚糖),3000×g离心10分钟去除未结合的[³H]-DHT。 4. 检测与计算:液体闪烁计数器检测上清放射性,采用Cheng-Prusoff方程计算Ki值[1] |
| 细胞实验 |
ODM-201的拮抗作用[1]
在AR-HEK293细胞中测定抗雄激素对hAR的功能活性和效力。用测试化合物和0.45处理细胞 补充2的无类固醇测定培养基中的nM睾酮 nM GlutaMAX和25 mM HEPES。24之后 小时37 °C和5%CO2,裂解细胞,并根据制造商的说明使用Centro LB 960微板光度计使用萤光素酶测定系统测量萤光素酶活性 突变AR研究[1] 使用LipofectaminTM2000用雄激素应答性报告基因构建体(pGV5-basic GRE-hiv-luc)和编码AR突变体AR(F876L)、AR(T877A)或AR(W741L)的表达载体(pSG5-hAR-F876L、pSG5-hAR-T877A或pSG5-hAR-W741L)瞬时转染人U2-OS骨肉瘤细胞。突变体AR表达载体的构建如前所述进行。对于96孔板中的一个孔,190 ng的报告构建体DNA和10 ng受体构建体DNA在Opti-MEM®(Gibco)中稀释。在不存在或存在诱导次最大报告基因激活的参考激动剂的情况下,用增加浓度的测试化合物处理细胞(0.6 T877A和F876L情况下的nM睾酮,以及10 在W741L的情况下为nM DHT)在不含类固醇的测定培养基中培养24 小时。如上所述测量荧光素酶活性 AR核易位[1] 用AR抗体免疫标记的AR过表达HS-HEK293细胞用高含量筛选(HCS)读取器(Cellomics ArrayScan HCS VTI读取器,Thermo)或用共聚焦显微镜成像。将HS-HEK293细胞在不含类固醇的测定培养基中铺板在聚-D-赖氨酸包被的微孔板(BD)(HCS读取器)或盖玻片(共聚焦成像)上。培养48小时后,用0.3(HCS阅读器)或1 μM(共聚焦成像)测试化合物与0.3 nM睾酮5 小时。在用3.7%PFA固定后,用磷酸盐缓冲盐水(PBS)洗涤细胞,用0.1%Triton X-100(Sigma)渗透,并用PBS中的3%BSA处理以阻断非特异性染色。对于HCS读取器,用与Alexa Fluor®488(N20,Santa Cruz,稀释度1:50)缀合的多克隆AR抗体孵育细胞。洗涤细胞,用DAPI标记DNA(Sigma,1 μg/mL),并用NucTrans分析图像。V3测定算法(Thermo)。对于共聚焦成像,使用多克隆AR抗体作为一级抗体,使用Alexa Fluor®546抗兔IgG作为二级抗体。使用含有DAPI(Vector Laboratories)的Vectashield安装盖玻片。AR过表达的LN-AR-C细胞用3 μM试验化合物和0.3 nM睾酮4 小时,用AR抗体和第二抗体免疫标记并用HCS读取器成像 VCaP增殖测定[1] 用次最大浓度的米博乐酮(0.1 nM)和在补充有4 mM GlutaMAX。在与化合物孵育4天后,根据制造商的说明书,使用WST-1细胞增殖测定法(Roche)测量细胞活力。为了排除非AR介导的毒性,用增加浓度的ODM-201处理AR阴性PC细胞(DU-145)和肺癌癌症细胞(H1581),并如上所述测量细胞活力。 1. 前列腺癌细胞增殖实验([1]): - 细胞培养:LNCaP、C4-2、22Rv1细胞用含10%胎牛血清的RPMI 1640培养基培养,接种于96孔板(5×10³细胞/孔)。 - 药物处理:用达洛鲁胺(0.01–50 μM)处理72小时;设溶剂对照(0.1% DMSO)及恩扎卢胺(0.01–50 μM,阳性对照)。 - 检测:培养最后4小时加入MTT试剂(10 μL/孔),570 nm处测吸光度,通过GraphPad Prism软件计算IC50值[1] 2. AR靶基因与核转位实验([1]): - 核AR检测:C4-2细胞(2×10⁴细胞/盖玻片)用达洛鲁胺(5 μM)处理6小时,4%多聚甲醛固定,抗AR一抗及Alexa Fluor 488标记二抗染色,ImageJ定量核荧光强度。 - 靶基因分析:LNCaP/22Rv1细胞(2×10⁵细胞/6孔板)用达洛鲁胺(5 μM)处理24小时,提取总RNA,qPCR检测PSA/TMPRSS2 mRNA水平(GAPDH为内参);蛋白质印迹法检测TMPRSS2蛋白[1] |
| 动物实验 |
Dissolved in Macrocol + propylene glycol +5% glucose (50:30:20, v/v/v); 50 mg/kg; p.o. administration
BALB/c nude male mice bearing VCaP xenografts The castration-resistant VCaP xenograft experiment [1] BALB/c nude male mice (7 weeks old) were subcutaneously injected with 2 million VCaP cells in 100 μL of RPMI-1640 medium and Matrigel (BD) (1:1). Tumor growth was monitored twice weekly by caliper measurements. The volume of the tumor was calculated according to the formula W2 × L/2 (mm3), where W is the shorter and L the longer diameter of the tumor. When the average tumor volume reached ~200 mm3, mice were castrated or SHAM-operated under Avertin anesthesia. Oral treatments with two doses of Darolutamide (ODM-201) (50 mg/kg, qd or bid), enzalutamide (20 mg/kg, qd), or vehicle were initiated upon tumor regrowth (when average tumor volumes were ~400 mm3) and were continued for 37 days. For all in vivo studies, Macrocol® (Merck) + propylene glycol +5% glucose (50:30:20, v/v/v) was used as a vehicle. Pharmacokinetic studies in mice[1] In the PK studies analyzing the penetration of test compounds to the brain, nude male mice (BALB/c or Balb/cOlaHsd, 8-9 weeks of age) were orally dosed for 7 days with 25, 50, or 100 mg/kg of Darolutamide (ODM-201) twice daily (n = 5) or with 20 mg/kg enzalutamide once daily (n = 4), or with a single oral dose of ARN-509 (10 mg/kg) (n = 3). Control mice received vehicle. Blood samples were collected into K2EDTA tubes by cardiac puncture under CO2 anesthesia and plasma was separated by centrifugation. Brain samples (without olfactory bulbs and medulla oblongata) from each group and time point were pooled and homogenized before the analysis. Concentrations of Darolutamide (ODM-201) and ORM-15341 in mouse plasma and brain were determined by liquid chromatography-tandem mass spectrometry (LC-MS/MS) method at Charles River, UK, with the lower limit of quantification (LLOQ) being in plasma 1.00 ng/mL for both Darolutamide (ODM-201) and ORM-15341 and in brain 4.00 ng/g for Darolutamide (ODM-201) and 10.00 ng/g for ORM-15341. Enzalutamide and ARN-509 concentrations were determined by LC-MS/MS method at Orion Pharma (LLOQ for enzalutamide was 1.00 ng/mL in plasma and 5.00 ng/g in brain, and for ARN-509 0.250 ng/mL in plasma and 10.0 ng/g in brain). Plasma and brain concentration vs. time were evaluated by noncompartmental analysis using WinNonlin® Professional v. 5.2 software. Brain/plasma ratios were calculated based on AUC0–24 values for plasma and brain. 22Rv1 CRPC Xenograft Protocol ([1]): 1. Animal Selection: 6–8 weeks old male SCID mice (n=6/group) randomized to vehicle control、Darolutamide 10 mg/kg、Darolutamide 30 mg/kg、enzalutamide 30 mg/kg (positive control). 2. Model Induction: 5×10⁶ 22Rv1 cells were suspended in 0.2 mL PBS + 50% Matrigel and subcutaneously injected into the right flank of mice. 3. Drug Preparation: Darolutamide was suspended in 0.5% carboxymethylcellulose (CMC) + 0.1% Tween 80 to concentrations of 1 mg/mL (10 mg/kg) and 3 mg/mL (30 mg/kg). 4. Administration: Oral gavage (10 mL/kg body weight) once daily for 28 days; vehicle control received 0.5% CMC + 0.1% Tween 80. 5. Detection: Tumor volume was measured twice weekly; mice were euthanized on day 28, serum collected for PSA ELISA, tumor tissue for Ki-67 immunohistochemistry and nuclear AR Western blot [1] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Darolutamide is absorbed in the gastrointestinal tract. In the fasted state, peak concentrations are reached within 3-5 hours, and within 3-8 hours in the fed state. Median Tmax is between 3-6 hours.The average darolutamide steady-state peak plasma concentration after a 600 mg twice daily dose is approximately 4.79 mg/L. The Cmax is attained approximately 4 hours after administration of a single 600 mg oral dose. The AUC 0-12h is approximately 52.82 h•μg/mL. Effects of food The absolute bioavailability of darolutamide is approximately 30% after fasting and taking a single 300 mg dose. Steady-state concentrations are attained between 2 and 5 days after repeated administration with food. The bioavailability of darolutamide increases by 2.0 to 2.5 times when it is given with food. In a pharmacokinetic study, a radiolabeled dose of darolutamide in an oral solution showed that 63.4% of darolutamide-related material was excreted in the urine (7% of which was unchanged drug) and 32.4% in the feces (with 30% unchanged drug). After intravenous administration, the apparent volume of distribution of darolutamide is about 119L. The clearance of darolutamide after an intravenous dose is 116 mL/min (39.7%). Metabolism / Metabolites Darolutamide is mainly metabolized by the CYP3A4 hepatic microsomal enzyme in addition to UGT1A9 and UGT1A1. The main active metabolite keto-darolutamide in found in the plasma at 2 times the concentration of darolutamide. Biological Half-Life The half-life of darolutamide and its active metabolite, keto-darolutamide is about 20 hours. A phase 1 study determined a terminal half life ranging between 10-15 hours. Oral Absorption: Darolutamide had an oral bioavailability of ~85% in rats; oral administration of 30 mg/kg resulted in a peak plasma concentration (Cmax) of 4.2 μg/mL at 2 hours [1] - Distribution: Volume of distribution (Vd) was 18 L/kg in rats, indicating extensive tissue distribution; prostate tissue concentration was 5.2× plasma concentration [1] - Metabolism: Primarily metabolized in the liver via CYP3A4 to inactive metabolites (M1、M2); no active metabolites were detected in plasma [1] - Elimination: Plasma half-life (t1/2) was 8.5 hours in rats; 70% of the dose was excreted in feces, 20% in urine (mostly metabolites) [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
Hepatotoxicity
In prelicensure controlled trials with 1508 patients, serum AST elevations were more common with darolutamide than placebo therapy [23% vs 14%], but were rarely above 5 times the ULN [ Likelihood score: E (unlikely cause of clinically apparent liver injury). Protein Binding The plasma protein binding for darolutamide is 92% and 99.8% for keto-darolutamide, the active metabolite. They are mainly bound to albumin. 1. In Vitro Toxicity ([1]): Darolutamide (0.01–50 μM) showed no cytotoxicity to normal human prostate epithelial cells (RWPE-1) or hepatocytes (HepG2), with cell viability >90% vs. control (MTT assay) [1] 2. In Vivo Toxicity ([1]): - Rats treated with Darolutamide (30 mg/kg/day for 28 days) had no significant changes in body weight、ALT/AST (liver function) or BUN/creatinine (kidney function). - Liver and kidney histopathological analysis showed no inflammation, necrosis, or fibrosis [1] 3. Plasma Protein Binding: >99.5% bound to human plasma albumin and α1-acid glycoprotein [1] |
| 参考文献 | |
| 其他信息 |
Darolutamide is a nonsteroidal androgen receptor antagonist for the treatment of castrate-resistant, non-metastatic prostate cancer (nmCRPC). This condition occurs in the majority of patients with advanced prostate cancer who have been treated with androgen receptor antagonists. Though prior treatment for prostate cancer has been successful for these patients, the cancer eventually progresses to become resistant to existing therapies. This warrants further treatment. The goal of treatment with darolutamide is to delay the progression of prostate cancer to metastatic disease, increasing quality of life and life expectancy for those with advanced prostate cancer. Darolutamide was developed by Bayer HealthCare Pharmaceuticals Inc. and approved by the FDA on July 30th, 2019.
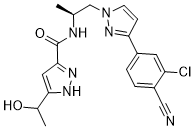
Darolutamide is a third generation, oral nonsteroidal antiandrogen used to treat nonmetastatic castration-resistant prostate cancer. Darolutamide is associated with a low rate of serum enzyme elevation during therapy, but has not been linked to cases of clinically apparent liver injury with jaundice. Darolutamide is a formulation containing an androgen receptor (AR) antagonist with potential antineoplastic activity. Darolutamide binds to ARs in target tissues; subsequently, inhibiting androgen-induced receptor activation and facilitating the formation of inactive complexes that cannot translocate to the nucleus. This prevents binding to and transcription of AR-responsive genes that regulate prostate cancer cell proliferation. This ultimately leads to an inhibition of growth in AR-expressing prostate cancer cells. Drug Indication Darolutamide is indicated for the treatment of adults with non-metastatic castration-resistant prostate cancer (nmCRPC) and metastatic hormone-sensitive prostate cancer (mHSPC) in combination with [docetaxel]. NUBEQA is indicated for the treatment of adult men with- non metastatic castration resistant prostate cancer (nmCRPC) who are at high risk of developing metastatic disease (see section 5. 1). - metastatic hormone sensitive prostate cancer (mHSPC) in combination with docetaxel and androgen deprivation therapy (see section 5. 1). Mechanism of Action The actions of androgens on androgen receptors (AR) potentiate the growth and survival of prostate cancer cells. Darolutamide competitively inhibits androgens from binding to their receptors, inhibiting AR nuclear translocation, as well as AR-mediated transcription. The end result of these processes is a decrease in prostate cancer cell proliferation and tumor size. Its main metabolite, keto-darolutamide, shows similar pharmacological activity to the parent drug, darolutamide. Darolutamide has been found to bind more tightly to the AR receptor than [apalutamide] and [enzalutamide], which are other androgen receptor antagonists. Darolutamide can act as a progesterone receptor (PR) antagonist in the laboratory setting with approximately 1% activity when compared to its actions at the androgen receptor. The clinical relevance is not known at this time. Pharmacodynamics Darolutamide, through its downstream effects on cancer cell growth, treats castrate-resistant prostate cancer. It inhibits cancer cell growth and markedly lowers prostate specific antigen (PSA) levels through potent androgen receptor antagonism. 1. Drug Background ([1]): Darolutamide (ODM-201; BAY-1841788) is a third-generation oral nonsteroidal antiandrogen (NSAA) developed to overcome resistance to first/second-generation AR inhibitors (e.g., bicalutamide、enzalutamide) in castration-resistant prostate cancer (CRPC) [1] 2. Mechanism of Action ([1]): - Step 1: Binds to WT and mutant AR (F876L、T877A) with high affinity (Ki <1 nM), competing with endogenous androgens (DHT) to block AR activation. - Step 2: Inhibits AR nuclear translocation (reducing nuclear AR by 80–85% in CRPC cells) and AR-DNA binding to androgen-responsive elements (AREs). - Step 3: Downregulates AR target genes (PSA、TMPRSS2) to inhibit CRPC cell proliferation and induce G1 cell cycle arrest [1] 3. Therapeutic Potential ([1]): Darolutamide shows potent activity against enzalutamide-resistant CRPC cells and xenografts, supporting its clinical development for treating metastatic castration-resistant prostate cancer (mCRPC) [1] |
| 分子式 |
C19H19CLN6O2
|
|
|---|---|---|
| 分子量 |
398.85
|
|
| 精确质量 |
398.125
|
|
| 元素分析 |
C, 57.22; H, 4.80; Cl, 8.89; N, 21.07; O, 8.02
|
|
| CAS号 |
1297538-32-9
|
|
| 相关CAS号 |
|
|
| PubChem CID |
67171867
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.4±0.1 g/cm3
|
|
| 沸点 |
719.5±60.0 °C at 760 mmHg
|
|
| 闪点 |
388.9±32.9 °C
|
|
| 蒸汽压 |
0.0±2.4 mmHg at 25°C
|
|
| 折射率 |
1.681
|
|
| LogP |
-0.04
|
|
| tPSA |
120Ų
|
|
| 氢键供体(HBD)数目 |
3
|
|
| 氢键受体(HBA)数目 |
5
|
|
| 可旋转键数目(RBC) |
6
|
|
| 重原子数目 |
28
|
|
| 分子复杂度/Complexity |
598
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
ClC1=C(C#N)C=CC(C2=NN(C[C@H](C)NC(C3=NNC(C(O)C)=C3)=O)C=C2)=C1
|
|
| InChi Key |
ANGUXJDGJCHGOG-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C25H29N5O/c1-19-8-9-22-23(27-19)6-3-7-24(22)29-16-14-28(15-17-29)12-10-20-4-2-5-21(18-20)30-13-11-26-25(30)31/h2-9,18H,10-17H2,1H3,(H,26,31)
|
|
| 化学名 |
N-((S)-1-(3-(3-chloro-4-cyanophenyl)-1H-pyrazol-1-yl)propan-2-yl)-5-(1-hydroxyethyl)-1H-pyrazole-3-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (5.21 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (5.21 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (5.21 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5072 mL | 12.5360 mL | 25.0721 mL | |
| 5 mM | 0.5014 mL | 2.5072 mL | 5.0144 mL | |
| 10 mM | 0.2507 mL | 1.2536 mL | 2.5072 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
|
|---|
|
|