| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
PLK1 (IC50 = 9 nM); PDGFR (IC50 = 18 nM); Bcr-Abl (IC50 = 32 nM); Flt1 (IC50 = 42 nM); Src (IC50 = 155 nM)
Polo-like Kinase 1 (PLK1) (Ki = 9 nM) [2] - Phosphoinositide 3-Kinase α (PI3Kα) (IC50 = 3.2 μM), PI3Kβ (IC50 = 4.5 μM) [2] - PI3Kγ (IC50 = 1.6 μM), PI3Kδ (IC50 = 2.6 μM) [1] |
|---|---|
| 体外研究 (In Vitro) |
Rigosertib 是 PLK1 的非 ATP 竞争性抑制剂,IC50 为 9 nM。 Rigosertib 还表现出对 PLK2、PDGFR、Flt1、BCR-ABL、Fyn、Src 和 CDK1 的抑制作用,IC50 为 18-260 nM。 Rigosertib 对 94 种不同的肿瘤细胞系显示出细胞杀伤活性,IC50 为 50-250 nM,包括 BT27、MCF-7、DU145、PC3、U87、A549、H187、RF1、HCT15、SW480 和 KB 细胞。而在正常细胞(例如 HFL、PrEC、HMEC 和 HUVEC)中,Rigosertib 的作用很小或没有作用,除非其浓度大于 5-10 μM。在 HeLa 细胞中,Rigosertib (100-250 nM) 会诱导纺锤体异常和细胞凋亡。 Rigosertib 还抑制多种耐药肿瘤细胞系,包括 MES-SA、MES-SA/DX5a、CEM 和 CEM/C2a,IC50 为 50-100 nM。在 DU145 细胞中,Rigosertib (0.25-5 μM) 可阻断 G2/M 期的细胞周期进展,导致含有 subG1 DNA 含量的细胞积累,并激活细胞凋亡途径。在 A549 细胞中,Rigosertib (50 nM-0.5 μM) 会导致活力丧失和 caspase 3/7 激活。在最近的一项研究中,Rigosertib 可诱导慢性淋巴细胞白血病 (CLL) 细胞凋亡,但对 T 细胞或正常 B 细胞没有毒性。 Rigosertib 还消除滤泡树突状细胞对 CLL 细胞的促生存作用,并减少 SDF-1 诱导的白血病细胞迁移。激酶测定:将重组 PLK1 (10 ng) 与不同浓度的 Rigosertib 在 15 µL 反应混合物(50 mM HEPES、10 mM MgCl2、1 mM EDTA、2 mM 二硫苏糖醇、0.01% NP-40 [pH 7.5])中孵育,室温下 30 分钟。激酶反应在 30 °C 下进行 20 分钟,体积为 20 µL(15 µL 酶 + 抑制剂、2 µL 1 mM ATP)、2 µL γ32P-ATP (40 µCi) 和 1 µL 重组 Cdc25C(100 ng)或酪蛋白(1 μg)底物。在 20 µL 2× Laemmli 缓冲液中煮沸 2 分钟终止反应。磷酸化底物通过 18% SDS-PAGE 分离。将凝胶干燥并暴露于 X 射线胶片 3-10 分钟。细胞测定:细胞(许多肿瘤细胞系,包括 BT20、MCF-7、DU145、PC3、U87、A549、H187、RF1、HCT15、HeLa 和 Raji 细胞)在补充有 10% 的 DMEM 或 RPMI 中生长胎牛血清和1单位/mL青霉素-链霉素溶液。将肿瘤细胞以 1 × 105 个细胞/mL/孔的密度接种到六孔培养皿中,24 小时后添加不同浓度的 Rigosertib。处理 96 小时后,从重复孔中测定细胞计数。活细胞总数通过台盼蓝排除法测定。
(E/Z)-Rigosertib sodium对28种人类癌细胞系(包括肺癌、结肠癌、乳腺癌和胰腺癌)的增殖具有抑制作用,IC50值范围为0.5-5.0 μM。它在HCT116和MCF-7细胞中诱导G2/M期细胞周期停滞,同时伴随Akt(Ser473)磷酸化水平降低和PARP切割增加。2 μM浓度处理48小时后,膜联蛋白V-FITC染色显示35-45%的细胞发生凋亡[1] - 在PLK1依赖型癌细胞系(HCT116、PC-3、A549)中,(E/Z)-Rigosertib sodium抑制细胞活力的IC50值分别为0.3 nM(PC-3)、0.8 μM(HCT116)和2.1 μM(A549)。蛋白质印迹分析显示PLK1底物(Cdc25C、周期蛋白B1)的磷酸化水平降低,α-微管蛋白染色显示纺锤体形成异常[2] - 在晚期实体瘤患者来源的肿瘤细胞中,(E/Z)-Rigosertib sodium以0.8-6.2 μM的IC50值抑制细胞增殖。在A549和HCT116细胞中,与紫杉醇联用时表现出协同抗增殖作用(协同指数=0.45-0.68)[3] |
| 体内研究 (In Vivo) |
在 Bel-7402、MCF-7 和 MIA-PaCa 细胞的小鼠异种移植模型中,Rigosertib (250 mg/kg) 显着抑制肿瘤生长。 Rigosertib (200 mg/kg) 在 BT20 细胞的小鼠异种移植模型中显示出对肿瘤生长的抑制作用。
在携带HCT116结肠癌异种移植物的裸鼠中,(E/Z)-Rigosertib sodium以100 mg/kg的剂量每周腹腔注射3次,连续4周,显著抑制肿瘤生长(肿瘤生长抑制率=65%;与溶媒组相比p < 0.001)。肿瘤组织中p-Akt和Ki-67表达降低[1] - 在PC-3前列腺癌异种移植物模型中,(E/Z)-Rigosertib sodium以80 mg/kg的剂量每周静脉注射2次,连续5周,实现72%的肿瘤生长抑制。与溶媒处理小鼠相比,肿瘤裂解物中PLK1激酶活性降低68%[2] - 在携带A549肺癌异种移植物的裸鼠中,口服(E/Z)-Rigosertib sodium 150 mg/kg,每日1次,连续3周,抑制肿瘤生长58%。与紫杉醇(10 mg/kg每周静脉注射1次)联用后,抑制率提升至83%,且未增加毒性[3] |
| 酶活实验 |
将重组 PLK1 (10 ng) 与不同浓度的 rigosertib 在 15 µL 反应混合物(50 mM HEPES、10 mM MgCl2、1 mM EDTA、2 mM 二硫苏糖醇)中在室温下孵育 30 分钟,0.01% NP-40 [pH 7.5])。 20 µL(15 µL 酶 + 抑制剂、2 µL 1 mM ATP)、2 µL γ 32 P-ATP (40 µCi) 和 1 µL 重组 Cdc25C (100 ng) 或酪蛋白 (1 μg)底物用于激酶反应,在 30°C 下进行 20 分钟。在 20 µL 2× Laemmli 缓冲液中煮沸 2 分钟,结束反应。 18% SDS-PAGE 用于分离磷酸化底物。干燥后,将凝胶暴露在 X 射线胶片下三到十分钟。
PI3K激酶实验:重组PI3K亚型(α、β、γ、δ)与磷脂酰肌醇-4,5-二磷酸(PIP2)和ATP共同孵育。将(E/Z)-Rigosertib sodium以系列浓度(0.1 μM至10 μM)加入,通过闪烁计数法检测磷脂酰肌醇-3,4,5-三磷酸(PIP3)的生成量,以测定激酶活性。反应在37°C下进行30分钟,从剂量-反应曲线计算IC50值[1] - PLK1激酶实验:重组PLK1催化结构域与荧光标记的肽底物(KKT(p)LRR)和ATP混合。(E/Z)-Rigosertib sodium的测试浓度范围为0.1 nM至1 μM,25°C孵育45分钟后,通过荧光共振能量转移(FRET)定量激酶活性,采用非线性回归分析确定Ki值[2] |
| 细胞实验 |
一天后,将不同浓度的 Rigosertib 添加到含有 1×10 5 细胞/mL/孔的已铺板肿瘤细胞的六孔培养皿中。处理96小时后,从重复的孔中获得细胞计数。通过使用台盼蓝排除法,可以找到活细胞的总数。
细胞增殖实验:癌细胞接种到96孔板(3×103个细胞/孔),过夜孵育。加入梯度浓度(0.01 μM至20 μM)的(E/Z)-Rigosertib sodium,细胞培养72小时。通过MTT法评估细胞活力,使用四参数逻辑回归推导IC50值[1] - 细胞周期和凋亡实验:HCT116细胞用(E/Z)-Rigosertib sodium(2 μM)处理24-48小时。细胞周期分析中,收集细胞、固定、碘化丙啶染色,通过流式细胞术分析。凋亡检测中,用膜联蛋白V-FITC和碘化丙啶染色细胞,随后进行流式细胞术定量。通过蛋白质印迹检测p-Akt、切割型PARP和PLK1底物蛋白[1] - 患者来源细胞实验:分离晚期癌症患者的原代肿瘤细胞并进行体外培养。细胞用(E/Z)-Rigosertib sodium(0.5-10 μM)单独处理或与紫杉醇(0.1 μM)联合处理。96小时后通过CCK-8法检测细胞活力,采用Chou-Talalay法计算协同指数[3] |
| 动物实验 |
Bel-7402、MCF-7 和 MIA-PaCa 细胞异种移植小鼠(雌性无胸腺,NCR-nu/nu)模型
250 mg/kg 腹腔注射 HCT116 结肠癌异种移植模型:将 HCT116 细胞(1×10⁷ 个细胞)皮下注射到 6-7 周龄雌性裸鼠右侧腹部。当肿瘤体积达到 120-150 mm³ 时,将小鼠随机分为治疗组(n=10)和载体对照组(n=10)。(E/Z)-Rigosertib 钠 溶于 DMSO,并用生理盐水稀释(1:9 v/v)至终浓度为 10 mg/mL。每周三次腹腔注射 100 mg/kg,持续 4 周。每周两次测量肿瘤体积和体重[1] - PC-3前列腺癌异种移植模型:将携带PC-3异种移植瘤(8×10⁶个细胞)的雄性裸鼠在肿瘤体积达到100-130 mm³时进行治疗。将(E/Z)-Rigosertib钠溶解于PEG400和生理盐水(2:8 v/v)混合液中,并以80 mg/kg的剂量经尾静脉注射,每周两次,持续5周。研究结束时,收集肿瘤组织进行PLK1活性检测[2] - A549肺癌异种移植模型:将携带A549异种移植瘤(5×10⁶个细胞)的裸鼠用(E/Z)-Rigosertib钠治疗,该药物配制成0.5%羧甲基纤维素钠(CMC-Na)口服混悬液,剂量为150 mg/kg,每日一次,持续3周。联合治疗组每周接受一次静脉注射紫杉醇(10 mg/kg)。研究期间持续监测肿瘤生长和动物存活情况[3] |
| 药代性质 (ADME/PK) |
小鼠单次口服100 mg/kg的(E/Z)-Rigosertib钠后,其口服生物利用度为35%[2]
- 小鼠静脉注射80 mg/kg后,其终末半衰期(t1/2)为4.2小时,分布容积(Vd)为1.8 L/kg,全身清除率(CL)为0.3 L/h/kg[2] - 通过平衡透析法测定,该药物在人血浆中的血浆蛋白结合率为92%。该药物主要在肝脏通过细胞色素P450酶代谢,约65%的剂量在72小时内经胆汁排泄[3] |
| 毒性/毒理 (Toxicokinetics/TK) |
在小鼠中,静脉注射(E/Z)-Rigosertib钠的最大耐受剂量(MTD)为120 mg/kg,未观察到死亡。高剂量治疗(200 mg/kg,腹腔注射)导致丙氨酸氨基转移酶(ALT)和天冬氨酸氨基转移酶(AST)轻度升高(较正常范围升高1.5倍),但未见组织病理学肝损伤[1]
- 小鼠长期口服给药(150 mg/kg,每日一次,持续4周)导致体重减轻<10%,未见明显的胃肠道毒性,且给药后12小时内血浆药物浓度稳定在体外IC50(1.2 μM)以上[3] - 在治疗剂量下,未观察到小鼠出现明显的血液学毒性(白细胞减少症、血小板减少症)或肾功能异常[2] |
| 参考文献 | |
| 其他信息 |
利戈塞替钠是利戈塞替的钠盐。它是一种抗癌药物,已被美国食品药品监督管理局 (FDA) 授予孤儿药资格,用于治疗骨髓增生异常综合征 (MDS) 患者。它具有微管去稳定剂、抗肿瘤剂、EC 2.7.11.21(polo激酶)抑制剂和细胞凋亡诱导剂的作用。它含有一个利戈塞替(1-)分子。
利戈塞替钠是利戈塞替的钠盐形式,利戈塞替是一种合成的苄基苯乙烯基砜类似物和Ras模拟物,具有潜在的抗肿瘤活性。给药后,利戈塞替靶向并结合许多Ras效应蛋白(包括Raf激酶和磷脂酰肌醇3-激酶 (PI3K))中的Ras结合域 (RBD)。这阻止了 Ras 与其靶标结合,并抑制了 Ras 介导的信号通路,包括 Ras/Raf/Erk、Ras/CRAF/polo 样激酶 1 (Plk1) 和 Ras/PI3K/Akt 信号通路。该药物可诱导多种易感肿瘤细胞的细胞周期阻滞和凋亡,并抑制其增殖。 另见:Rigosertib(注释已移至)。 (E/Z)-Rigosertib钠是一种新型多靶点激酶抑制剂,主要通过抑制PLK1和PI3K信号通路发挥抗肿瘤作用,对Ras突变型癌症的疗效更佳[1] - 该化合物具有喹唑啉核心结构,结构优化提高了其对PLK1的选择性和体内稳定性,优于早期类似物[2] - 临床前研究支持将(E/Z)-Rigosertib钠作为单药或联合疗法用于晚期实体瘤,目前已进入Ⅱ期临床试验[3] |
| 分子式 |
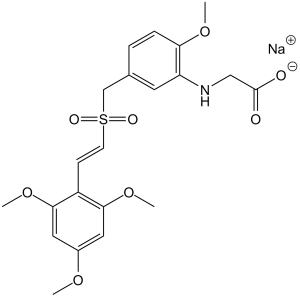
C21H24NNAO8S
|
|---|---|
| 分子量 |
473.47
|
| 精确质量 |
473.112
|
| 元素分析 |
C, 55.86; H, 5.58; N, 3.10; O, 28.35; S, 7.10
|
| CAS号 |
1225497-78-8
|
| 相关CAS号 |
Rigosertib sodium;592542-60-4;Rigosertib;592542-59-1
|
| PubChem CID |
23696523
|
| 外观&性状 |
white solid powder
|
| LogP |
2.622
|
| tPSA |
131.6
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
9
|
| 可旋转键数目(RBC) |
11
|
| 重原子数目 |
32
|
| 分子复杂度/Complexity |
684
|
| 定义原子立体中心数目 |
0
|
| SMILES |
S(/C(/[H])=C(\[H])/C1C(=C([H])C(=C([H])C=1OC([H])([H])[H])OC([H])([H])[H])OC([H])([H])[H])(C([H])([H])C1C([H])=C([H])C(=C(C=1[H])N([H])C([H])([H])C(=O)[O-])OC([H])([H])[H])(=O)=O.[Na+]
|
| InChi Key |
VLQLUZFVFXYXQE-USRGLUTNSA-M
|
| InChi Code |
InChI=1S/C21H25NO8S.Na/c1-27-15-10-19(29-3)16(20(11-15)30-4)7-8-31(25,26)13-14-5-6-18(28-2)17(9-14)22-12-21(23)24;/h5-11,22H,12-13H2,1-4H3,(H,23,24);/q;+1/p-1/b8-7+;
|
| 化学名 |
sodium;2-[2-methoxy-5-[[(E)-2-(2,4,6-trimethoxyphenyl)ethenyl]sulfonylmethyl]anilino]acetate
|
| 别名 |
ON-01910; Rigosertib sodium; ON01910; ON 01910; brand name: Estybon
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1121 mL | 10.5603 mL | 21.1207 mL | |
| 5 mM | 0.4224 mL | 2.1121 mL | 4.2241 mL | |
| 10 mM | 0.2112 mL | 1.0560 mL | 2.1121 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04177498 | Recruiting | Other: Quality-of-Life Assessment Drug: Rigosertib Sodium |
Recessive Dystrophic Epidermolysis Bullosa |
Thomas Jefferson University | August 24, 2021 | Early Phase 1 |
| NCT04263090 | Recruiting | Drug: Rigosertib Drug: Nivolumab |
Adenocarcinoma Stage IV |
Icahn School of Medicine at Mount Sinai |
June 29, 2020 | Phase 1 Phase 2 |
| NCT03786237 | Recruiting | Drug: Rigosertib Oral Capsules / Rigosertib Intravenous |
Epidermolysis Bullosa Dystrophica Squamous Cell Carcinoma |
Prof. Johann Bauer | April 12, 2021 | Phase 1 Phase 2 |
| NCT05764395 | Recruiting | Drug: Rigosertib Procedure: Biopsy |
Metastatic Melanoma Refractory Melanoma |
Vanderbilt-Ingram Cancer Center |
May 9, 2023 | Phase 2 |
 28(ON 01910.Na) selectively induces mitotic G2/M arrest and apoptosis in cancer cells.J Med Chem.2011 Sep 22;54(18):6254-76. |
|---|
DU145 and HFL-1 (normal human fibroblasts) cells were treated with increasing concentrations of28or DMSO (Vehicle) for 48 h.J Med Chem.2011 Sep 22;54(18):6254-76. |
Cellular viability together with the activity of caspases 3/7 were assayed concomitantly in A549 cells treated with28for 24 h (n=3).J Med Chem.2011 Sep 22;54(18):6254-76. |