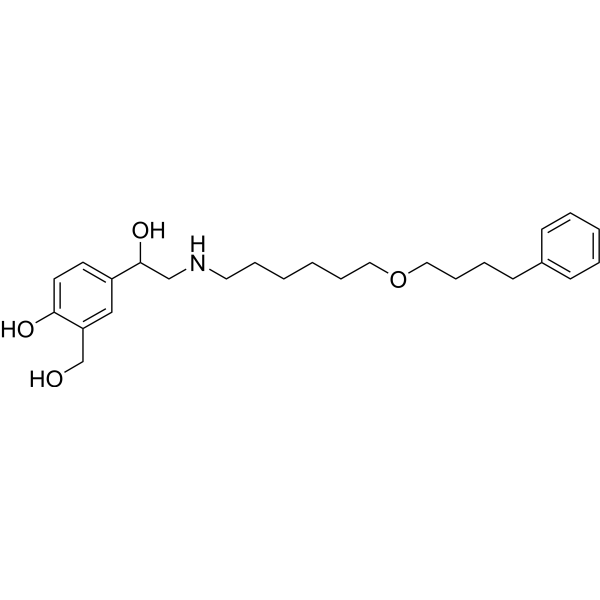
沙美特罗是一种有效的选择性 β2-肾上腺素受体激动剂 (EC50 = 5.3 nM);支气管扩张剂。与其他 β2 激动剂不同,它与 β2 受体的外位点结构域结合,起效缓慢,激活时间延长。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Salmeterol Effect on Exercise Performance
CTID: NCT06655012
Phase: N/A Status: Not yet recruiting
Date: 2024-10-23
Muscle Anabolic Response to Β2-adrenergic Stimulation with Increased Amino Acid Availability.
CTID: NCT06654986
Phase: N/A Status: Not yet recruiting
Date: 2024-10-23
Two Investigational Drugs in the Prevention of Airway Constriction Brought on by Exercise in Participants With Asthma (0476-911)
CTID: NCT00127166
Phase: Phase 3 Status: Completed
Date: 2024-05-10
ANTES B+ Clinical Trial
CTID: NCT06282861
Phase: Phase 4 Status: Recruiting
Date: 2024-03-05
A Multiple Dose Comparison of Tiotropium Inhalation Capsules and Salmeterol Inhalation Aerosol.
CTID: NCT00274560
Phase: Phase 3 Status: Completed
Date: 2023-12-01
View More
Tiotropium/Salmeterol Inhalation Powder in COPD
CTID: NCT00668772
Phase: Phase 3 Status: Terminated
Date: 2023-11-07
Tiotropium/Salmeterol Inhalation Powder (Spiriva Handihaler and Salmeterol Polyethylene (PE) Capsule) in Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT00662740
Phase: Phase 3 Status: Terminated
Date: 2023-08-23
Replication of the POET-COPD Trial in Healthcare Claims Data
CTID: NCT05083429
Phase: Status: Completed
Date: 2023-07-27
Single Blind Cross-over Dose Response Study in Subjects of Two Inhalers of Salmeterol and Fluticasone Propionate
CTID: NCT02232087
Phase: Phase 1 Status: Completed
Date: 2022-06-15
Uncontrolled Lower Respiratory Symptoms in the WTC Survivor Program
CTID: NCT02024204
Phase: N/A Status: Completed
Date: 2021-02-02
The ENERGITO® 2 Study Compares 2 Inhaled Medicines for Chronic Obstructive Pulmonary Disease (COPD). One Medicine is a Combination of Tiotropium and Olodaterol (Stiolto®) Taken Using the Respimat® Inhaler and the Other Medicine is a Combination of Fluticasone and Salmeterol Taken Using the Diskus
CTID: NCT03240575
Phase: Phase 4 Status: Completed
Date: 2020-04-16
Fluticasone and Salmeterol in Allergic Rhinitis
CTID: NCT01388595
Phase: Phase 4 Status: Completed
Date: 2019-10-16
Cardiovascular Function in COPD Patients
CTID: NCT03055988
Phase: Phase 4 Status: Completed
Date: 2019-08-16
Effects of Particle Size in Small Airways Dysfunction
CTID: NCT01892787
Phase: Phase 4 Status: Completed
Date: 2019-04-12
A Study To Investigate The Safety, Toleration And Efficacy of PF00610355 In Chronic Obstructive Pulmonary Disease (COPD) Patients.
CTID: NCT00808288
Phase: Phase 2 Status: Completed
Date: 2019-02-06
A Study to Compare the Efficacy of Fluticasone Furoate/Vilanterol Inhalation Powder With Usual Inhaled Corticosteroids (ICS)/Long Acting Beta Agonists (LABA) in Persistent Asthma
CTID: NCT02446418
Phase: Phase 3 Status: Completed
Date: 2019-01-14
Tiotropium Inhalation Capsules and Salmeterol Inhalation Aerosol on Muscular Efficiency and Resting Energy Expenditure (REE) in Patients With Stable Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT02172794
Phase: Phase 3 Status: Completed
Date: 2018-08-31
Validation of a New Shortness of Breath With Daily Activities Questionnaire in Patients With Chronic Obstructive Pulmonary Disease
CTID: NCT00984659
Phase: Phase 4 Status: Completed
Date: 2018-08-29
Childhood Asthma Research and Education (CARE) Network Trial - Best Add-On Therapy Giving Effective Response (BADGER)
CTID: NCT00395304
Phase: Phase 3 Status: Completed
Date: 2018-07-02
Blacks and Exacerbations on Long Acting Beta Agonists (LABA) vs. Tiotropium (BELT)
CTID: NCT01290874
Phase: Phase 3 Status: Completed
Date: 2018-03-30
Asthma Clinical Research Network (ACRN) Trial - Long-Acting Beta Agonist Response by Genotype (LARGE)
CTID: NCT00200967
Phase: Phase 3 Status: Completed
Date: 2018-01-23
The Effect of Salmeterol on Eosinophil (EOS) Function
CTID: NCT00214019
Phase: N/A Status: Completed
Date: 2017-11-21
GSK159802 In Healthy Male Subjects And Asthmatics
CTID: NCT00364273
Phase: Phase 1 Status: Completed
Date: 2017-09-29
Repeat Doses Of A New Medication (GW642444) In Asthmatic Patients
CTID: NCT00347139
Phase: Phase 2 Status: Completed
Date: 2017-09-14
How Different Beta-2 Receptor Genotypes Affect an Asthmatic's Response to Regular Salmeterol Treatment
CTID: NCT00595361
Phase: N/A Status: Completed
Date: 2017-06-08
The Effects of Different Long-acting Bronchodilator Medications on Asthma Patients With Different Genetic Variations
CTID: NCT00706446
Phase: N/A Status: Terminated
Date: 2017-05-31
Study to Evaluate Efficacy/Safety of Combination Budesonide/Indacaterol vs Fluticasone/Salmeterol in Patients With COPD
CTID: NCT02055352
Phase: Phase 4 Status: Completed
Date: 2017-04-18
Dose Finding Study of CHF 4226 for Treating Patients With COPD
CTID: NCT00605891
Phase: Phase 2 Status: Completed
Date: 2017-04-11
Pharmacological Properties of Salmeterol
CTID: NCT02558088
Phase: Phase 4 Status: Completed
Date: 2017-01-31
Advair® DISKUS® Versus Serevent® DISKUS® For Chronic Obstructive Pulmonary Disease Exacerbations
CTID: NCT00115492
Phase: Phase 4 Status: Completed
Date: 2017-01-20
Chronic Obstructive Pulmonary Disease Endpoints Study
CTID: NCT00358358
Phase: Phase 4 Status: Completed
Date: 2017-01-20
A 4-week Dose-Ranging, Dose-Interval, Efficacy, Safety and Tolerability Study of GSK961081 in Subjects With Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT01319019
Phase: Phase 2 Status: Completed
Date: 2016-12-01
ADVAIR DISKUS® (Fluticasone Propionate/Salmeterol) Inhaler Versus SEREVENT DISKUS® (Salmeterol) Inhlaer On Inflammatory Cells And Markers In Chronic Obstructive Pulmonary Disease. ADVAIR DISKUS® and SEREVENT DISKUS® Inhalers Are Trademarks of the GSK Group of Companies.
CTID: NCT00346749
Phase: Phase 4 Status: Terminated
Date: 2016-10-28
ADVAIR® DISKUS® Inhaler (Fluticasone Propionate/Salmeterol) Versus SEREVENT® DISKUS® Inhaler (Salmeterol) For The Treatment Of Chronic Obstructive Pulmonary Disease Exacerbations. ADVAIR® DISKUS® Inhaler and SEREVENT® DISKUS® Inhaler Are Trademarks of the GSK Group of Companies.
CTID: NCT00144911
Phase: Phase 4 Status: Completed
Date: 2016-10-28
Safety And Efficacy Of GSK233705 Plus Salmeterol Compared With 2 Active Comparators And Placebo In Subjects With Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT00422604
Phase: Phase 2 Status: Completed
Date: 2016-10-28
Study Investigating Repeat Doses Of A New Medication (GSK159797) In Asthmatic Patients
CTID: NCT00358488
Phase: Phase 2 Status: Completed
Date: 2016-10-28
Investigation Of A New Medication (GW642444) In Asthmatic Patients
CTID: NCT00354874
Phase: Phase 2 Status: Completed
Date: 2016-09-15
Childhood Asthma Research and Education (CARE) Network Trial - Montelukast or Azithromycin for Reduction of Inhaled Corticosteroids in Childhood Asthma (MARS)
CTID: NCT00471809
Phase: Phase 4 Status: Terminated
Date: 2016-07-29
Bioequivalence Study of Synflutide HFA Inhaler and Seretide Evohaler in Healthy Volunteers Without Charcoal Block
CTID: NCT02466347
Phase: Phase 1 Status: Completed
Date: 2016-07-26
Interactive Acute Smooth Muscle Effects of Salmeterol and Fluticasone in the Airway
CTID: NCT01231230
Phase: N/A Status: Completed
Date: 2016-06-30
Effects of Inhaled Corticosteroids on Sputum Bacterial Load in COPD
CTID: NCT01213693
Phase: N/A Status: Completed
Date: 2016-04-08
Characterization of Lung Function Profile of Inhaled Tiotropium + Olodaterol Fixed Dose Combination Compared to Fluticasone Propionate + Salmeterol Fixed Dose Combination in COPD Patients
CTID: NCT01969721
Phase: Phase 3 Status: Completed
Date: 2016-02-12
Effects of Anticholinergic or Long-Acting Beta 2 Agonist on FeNO and Pulmonary Function in SCI
CTID: NCT01355991
Phase: Phase 1 Status: Unknown status
Date: 2015-10-23
Nitric Oxyde Concentration in Chronic Obstructive Pulmonary Disease Patients - SANOB Study
CTID: NCT01853787
Phase: Phase 4 Status: Completed
Date: 2015-07-10
Bioequivalence Study of Synflutide HFA Inhaler and Seretide Evohaler in Healthy Volunteers With Charcoal Block
CTID: NCT02466503
Phase: Phase 1 Status: Completed
Date: 2015-06-09
Comparison of Indacaterol 150 mcg Once Daily (o.d.) With Salmeterol/Fluticasone Propionate 50 mcg/500 mcg Twice Daily (b.i.d.)
CTID: NCT01555138
Phase: Phase 4 Status: Completed
Date: 2015-04-28
Evaluation Inhaled Corticosteroids on Exhaled Nitric Oxide Gas Exchange
CTID: NCT00568347
Phase: Status: Completed
Date: 2015-03-17
Efficacy and Safety of QMF149 vs. Salmeterol Xinafoate/Fluticasone Propionate in Patients With Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT01636076
Phase: Phase 2 Status: Completed
Date: 2014-11-17
In Vivo Specificity of KUC 7483 CL Co-administered With Bisoprolol, Propranolol, and Acipimox in Healthy Male Subjects
CTID: NCT02256722
Phase: Phase 1 Status: Completed
Date: 2014-10-06
Pharmacokinetics of Salmeterol Via HandiHaler® in Healthy Male Volunteers
CTID: NCT02254187
Phase: Phase 1 Status: Completed
Date: 2014-10-01
Free Combinations of Tiotropium Inhalation Powder Capsule + Salmeterol Metered Dose Inhaler, Tiotropium Inhalation Powder Capsule and Salmeterol Metered Dose Inhaler in Patients With Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT02242253
Phase: Phase 2 Status: Completed
Date: 2014-09-17
Salmeterol Inhalation Powder Administered as the Xinafoate Salt From Hard Polyethylene Capsules Via the HandiHaler® 2, and Serevent® Diskus® in Patients With Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT02238106
Phase: Phase 2 Status: Completed
Date: 2014-09-12
Multiple Dose Comparison of Tiotropium Inhalation Capsules, Salmeterol Inhalation Aerosol and Placebo in Patients With Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT02172287
Phase: Phase 3 Status: Completed
Date: 2014-06-24
Efficacy and Safety Comparison of Steroid or Placebo in Combination With Salmeterol and Tiotropium in COPD
CTID: NCT00535366
Phase: Phase 2 Status: Completed
Date: 2014-05-30
Tiotropium and Salmeterol PK Study in COPD Patients
CTID: NCT00673478
Phase: Phase 3 Status: Completed
Date: 2014-05-16
Effects of Salmeterol on Autonomic Nervous System
CTID: NCT01536587
Phase: Phase 4 Status: Completed
Date: 2014-04-11
Tiotropium Once Daily 18 Mcg Versus Salmeterol Twice Daily 50 Mcg on Time to First Exacerbation in COPD Patients.
CTID: NCT00563381
Phase: Phase 4 Status: Completed
Date: 2013-12-24
A Comparison of the Effects of Tiotropium Inhalation qd and Salmeterol Inhalation Bid on Arterial Blood Gases.
CTID: NCT00274534
Phase: Phase 3 Status: Completed
Date: 2013-11-05
Efficacy of Inhaling Bronchodilator Medications in Chronic Obstructive Pulmonary Disease
CTID: NCT01391559
Phase: N/A Status: Completed
Date: 2013-08-07
Evaluate Onset of Effect in Patients With Chronic Obstructive Pulmonary Disease (COPD) Treated With Formoterol Turbuhaler®
CTID: NCT01048333
Phase: Phase 2 Status: Completed
Date: 2012-10-25
Sunovion Brovana Versus Serevent Inspiratory Capacity High Resolution Computed Tomography
CTID: NCT01361984
Phase: Phase 4 Status: Unknown status
Date: 2012-07-20
Safety of Exercise and High-dose Salbutamol in Patients With Chronic Obstructive Pulmonary Disease (COPD) Receiving Therapeutic Doses of Indacaterol (QAB 149) and Salmeterol
CTID: NCT00531050
Phase: Phase 2 Status: Completed
Date: 2012-05-23
The Effects of Montelukast on Smokers With Asthma
CTID: NCT00712335
Phase: Phase 4 Status: Completed
Date: 2012-05-16
Determine the Safety and Efficacy of (R,R)-Formoterol in the Treatment of Subjects With COPD
CTID: NCT00064402
Phase: Phase 3 Status: Completed
Date: 2012-02-22
To Evaluate the Long-term Safety of (R,R)-Formoterol in Subjects With COPD
CTID: NCT00064415
Phase: Phase 3 Status: Completed
Date: 2012-02-22
Management of Asthma in School-age Children on Therapy
CTID: NCT01526161
Phase: Phase 4 Status: Completed
Date: 2012-02-03
Safety of Indacaterol in Patients (≥ 12 Years) With Moderate to Severe Persistent Asthma
CTID: NCT00529529
Phase: Phase 3 Status: Completed
Date: 2011-08-29
Efficacy and Safety of Different Doses of Indacaterol in Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT01089127
Phase: Phase 3 Status: Completed
Date: 2011-08-19
Efficacy and Safety of Different Doses of Indacaterol
CTID: NCT01079130
Phase: Phase 3 Status: Completed
Date: 2011-08-19
Efficacy and Safety of Indacaterol in Patients With Chronic Obstructive Pulmonary Disease (COPD) Using Salmeterol as Active Control
CTID: NCT00567996
Phase: Phase 3 Status: Completed
Date: 2011-08-18
Safety and Efficacy of Indacaterol Once Daily Versus Salmeterol Twice Daily in Chronic Obstructive Pulmonary Disease (COPD)
CTID: NCT00821093
Phase: Phase 3 Status: Completed
Date: 2011-08-18
Dose Ranging Study for
Personalized pharmacological treatment of chronic obstructive pulmonary disease based on phenotyping: interventional study
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-08-08
Description of the ability to learn how to handle inhaler devices in COPD
CTID: null
Phase: Phase 4 Status: Completed
Date: 2016-05-19
An open label, prospective, randomized, parallel group, multicenter 4-week study to evaluate the efficacy and safety of salmeterol/ fluticasone propionate fixed dose combination following a switch from Viani ® Evohaler to Serroflo ® pressurized metered dose inhaler (pMDI) in subjects with stable persistent asthma
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-04-18
A 26-week, randomized, double blind, parallel-group multicenter study to assess the efficacy and safety of QVA149 (110/50 μg o.d.) vs tiotropium (18 μg o.d.) + salmeterol/fluticasone propionate FDC (50/500 μg b.i.d.) in patients with moderate to severe COPD
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-09-02
Pharmacokinetics of inhaled salmeterol administrated in healthy trained males
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-09-26
A prospective, multicenter, 12-week, randomized open-label study to evaluate the efficacy and safety of glycopyrronium (50 micrograms o.d.) or indacaterol maleate and glycopyrronium bromide fixed-dose combination (110/50 micrograms o.d.) regarding symptoms and health status in patients with moderate chronic obstructive pulmonary disease (COPD) switching from treatment with any standard COPD regimen
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-02-26
A RANDOMISED, DOUBLE-BLIND, DOUBLE-DUMMY, ACTIVE-CONTROLLED STUDY EVALUATING THE EFFICACY, SAFETY AND TOLERABILITY OF TWICE-DAILY ACLIDINIUM BROMIDE /FORMOTEROL FUMARATE COMPARED WITH TWICE-DAILY SALMETEROL/FLUTICASONE PROPIONATE FOR 24-WEEKS TREATMENT IN SYMPTOMATIC PATIENTS WITH CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD)
CTID: null
Phase: Phase 3 Status: Completed
Date: 2013-08-08
Randomised controlled single and chronic dosing crossover comparison of extra fine particle formoterol and coarse particle salmeterol in asthmatic patients with persistent small airways dysfunction
CTID: null
Phase: Phase 4 Status: Completed
Date: 2013-05-27
A 52-week treatment, multi-center, randomized, double-blind, double-dummy, parallel-group, active controlled study to compare the effect of QVA149 (indacaterol maleate / glycopyrronium bromide) with salmeterol/fluticasone on the rate of exacerbations in subjects with moderate to very severe COPD.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2013-02-27
Effects of bronchodilatation with salmeterol on the autonomic nervous system
CTID: null
Phase: Phase 4 Status: Completed
Date: 2012-03-29
A double-blind, double dummy randomised, parallel group, multicentre study to compare the efficacy and safety of Flutiform pMDI with fluticasone pMDI and with Seretide pMDI in paediatric subjects aged 5 to less than 12 years with moderate to severe persistent reversible asthma.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-08-30
A Phase III randomised, double-blind, placebo-controlled, parallel-group trial to evaluate efficacy and safety of tiotropium inhalation solution delivered via Respimat® inhaler (2.5 and 5 μg once daily) compared with placebo and salmeterol HFA MDI (50 μg twice daily) over 24 weeks in patients with moderate persistent asthma
CTID: null
Phase: Phase 3 Status: Completed
Date: 2010-09-20
A Phase III randomised, double-blind, placebo-controlled, parallel-group trial to evaluate efficacy and safety of tiotropium inhalation solution delivered via Respimat® inhaler (2.5 and 5 μg once daily) compared with placebo and salmeterol HFA MDI (50 μg twice daily) over 24 weeks in patients with moderate persistent asthma
CTID: null
Phase: Phase 3 Status: Completed
Date: 2010-06-14
A PHASE 2B, PARALLEL, DOUBLE BLIND, DOUBLE DUMMY, ACTIVE
CTID: null
Phase: Phase 2 Status: Completed
Date: 2010-05-19
Estudio multicéntrico, aleatorizado, doble ciego, doble enmascarado, con grupos paralelos de 12 semanas de tratamiento para evaluar la superioridad de indacaterol (150 µg o.d.) administrado mediante SDDPI en pacientes con EPOC de moderada a grave, usando salmeterol (50 ?g b.i.d.) como comparador activo administrado mediante inhalador DISKUS
CTID: null
Phase: Phase 3 Status: Completed
Date: 2009-02-19
A phase III, randomized, double-blind, triple-dummy, placebo controlled, multicenter, 5- period, single-dose complete block crossover study to determine the onset of action of indacaterol (150 and 300 μg) in patients with moderate to severe COPD using salbutamol (200 μg) and salmeterol/fluticasone (50/500 μg) as active controls.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2008-04-09
A Phase III, randomized, double-blind, placebo controlled, multicenter, 3-period, 14 day crossover study to determine the 24-h lung function profile of indacaterol (300 μg o.d.) in patients with moderate to- severe COPD, using open-label salmeterol (50 μg b.i.d.) as active control
CTID: null
Phase: Phase 3 Status: Completed
Date: 2008-01-18
A phase III randomized, double-blind, double dummy, placebo controlled, multicenter, 4 treatments, 3 period incomplete block crossover study to assess the efficacy and safety of indacaterol 300 µg o.d. dosed in the evening in patients with moderate to severe chronic obstructive pulmonary disease (COPD), using salmeterol 50 µg b.i.d. as active control
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-12-18
Effect of inhalation of tiotropium once daily 18 mcg versus salmeterol
CTID: null
Phase: Phase 4 Status: Completed
Date: 2007-11-13
Beta Agonist Lung injury Trial - Prevention Study
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-10-31
An exploratory, multi-centre, double-blind, placebo-controlled crossover study, to investigate the bronchodilatory efficacy of a single dose of indacaterol in fixed combination with mometasone furoate delivered via a MDDPI (Twisthaler®) in adult patients with persistent asthma using open label Seretide® Accuhaler® (50/250 mcg b.i.d.) as an active control.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2007-10-25
THE EFFECT ON ALVEOLAR NITRIC OXIDE OF SALMETEROL, FLUTICASONE, AND IN COMBINATION, IN STABLE BRONCHIECTASIS.
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2007-09-10
Rationale for therapy with low dose steroids combined with long-acting beta2-agonists in patients with allergic asthma: redirecting innate immune responses by long-term treatment with high doses of inhaled steroids
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2007-09-04
A double-blind, randomised, cross-over, multi-centre study, to evaluate onset of effect in the morning in patients with severe Chronic Obstructive Pulmonary Disease (COPD) treated with budesonide/formoterol (Symbicort®Turbuhaler®) 320/9 μg, compared with salmeterol/fluticasone (Seretide® Diskus®) 50/500 μg, both given as one inhalation twice daily for one week each.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2007-08-24
A 26-week treatment, multi center, randomized, double blind, double dummy, placebo controlled, parallel group study to assess the efficacy and safety of indacaterol (150 μg o.d.) in patients with chronic obstructive pulmonary disease, using salmeterol (50 μg b.i.d.) as an active control
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-07-20
A 26 week treatment, randomized, multi center, double blind, double dummy, parallel-group study to assess the safety of indacaterol (300 and 600 µg o.d.) in patients with moderate to severe persistent asthma, using salmeterol (50 µg b.i.d.) as an active control.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-07-12
A double-blind, randomized, cross-over, placebo-controlled, 2-part study to compare the effect of exercise and high-dose salbutamol on maximal heart-rate in patients with COPD following therapeutic doses of inhaled QAB149 and salmeterol
CTID: null
Phase: Phase 2 Status: Completed
Date: 2007-07-11
A study to assess the pharmacokinetics of single escalating doses of inhaled GSK961081 DPI (a dual pharmacophore) in healthy subjects (Part 1) and a randomised, double-blind, double dummy, crossover (incomplete block) study to assess the safety, tolerability, pharmacodynamics (pulmonary and systemic) and pharmacokinetics of 14 days dosing with inhaled GSK961081 DPI compared with placebo and tiotropium plus salmeterol in patients with COPD (Part 2).
CTID: null
Phase: Phase 1, Phase 2 Status: Completed
Date: 2007-06-11
A PHASE IIA RANDOMISED, DOUBLE-BLIND, DOUBLE-DUMMY, PLACEBO AND ACTIVE CONTROLLED 5-WAY CROSS-OVER TRIAL TO EXAMINE THE BRONCHODILATOR EFFECTS OF PF-610,355 AND TO TEST FOR SUPERIORITY VERSUS PLACEBO IN REVERSIBLE ASTHMATIC PATIENTS.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2007-06-05
A randomized, double-blind, double-dummy, two-way cross-over
CTID: null
Phase: Phase 4 Status: Completed
Date: 2007-04-25
A multi-centre, randomised, double-blind, double-dummy,
CTID: null
Phase: Phase 2 Status: Completed
Date: 2006-10-11
A Randomized, Controlled, 14-Treatment Day, Multicenter Study to Determine the Optimal Efficacious and Safe Dose of CHF 4226 in a Metered Dose Inhaler in Treating Patients With Chronic Obstuctive Pulmonary Disease
CTID: null
Phase: Phase 2 Status: Completed
Date: 2006-10-02
A 16-week randomised, placebo-controlled, double-blind, double-dummy, parallel-group study comparing the efficacy and safety of tiotropium inhalation solution delivered by the Respimat® inhaler (2 puffs of 2.5 micrograms once daily) with that of salmeterol from the hydrofluoroalkane metered dose inhaler (2 puffs of 25 micrograms twice daily) in moderate persistent asthma patients homozygous for B16-Arg/Arg
CTID: null
Phase: Phase 2 Status: Completed
Date: 2006-07-31
DOUBLE BLIND, DOUBLE DUMMY, RANDOMISED, PARALLEL GROUP, MONOCENTRIC CLINICAL TRIAL ON THE EFFECTS OF CHF 1535 MDI OR SERETIDE DPI ON LUNG HYPERINFLATION AND EXERCISE TOLERANCE IN PATIENTS WITH COPD A PILOT STUDY
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-05-08
A CUMULATIVE DOSE RESPONSE STUDY TO EVALUATE THE THERAPEUTIC EQUIVALENCE OF A NEW SALMETEROL INHALATION AEROSOL CONTAINING A REPLACEMENT HFA PROPELLANT IN A PRESSURISED METERED DOSE INHALER AND EXISTING SALMETEROL–CFC PRODUCT
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-01-25
RANDOMIZED, DOUBLE-BLIND, PLACEBO-CONTROLLED CLINICAL CROSSOVER TRIAL IN ADULT ASTHMATICS EVALUATING THE EFFECT OF CONCOMITANT TWO WEEKS TREATMENT WITH MONTELUKAST (SINGULAIR™) 10 MG ONCE DAILY OR MATCHING PLACEBO TO PREVENT THE DEVELOPMENT OF TOLERANCE TO BRONCHOPROTECTION AND BRONCHODILATION BY BETA-AGONISTS OCCURRING AFTER TWO WEEKS REGULAR TREATMENT WITH SALMETEROL (SEREVENT™) 50µG B.I.D.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2005-12-08
500µg roflumilast once daily in combination with 50µg salmeterol twice daily versus 50µg salmeterol twice daily alone over 52 weeks in patients with COPD.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2005-03-21
An exploratory, multi-center, randomized, open-label, single dose, crossover study to assess the safety and tolerability of 200µg of QAB149, delivered via a MDDPI, with or without the co-administration of water, with inhalation of 50µg of salmeterol, via a MDDPI, as an active comparator, in adult and adolescent patients with stable persistent asthma, or patients with COPD.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2005-02-25
A RANDOMISED, DOUBLE-BLIND, PLACEBO CONTROLLED STUDY TO EVALUATE THE EFFICACY AND SAFETY OF TACROLIMUS MDI AS ADD-ON THERAPY TO ICS AND LABA
CTID: null
Phase: Phase 2 Status: Completed
Date: 2004-11-01
A randomised, placebo-controlled, double-blind, double-dummy, crossover study to assess the onset of action of two inhalations of Symbicort 160/4.5μg compared with two inhalations of Seretide 25/250μg, two inhalations of Ventoline 100μg, and placebo, delivered by pressurised metered dose inhalers, in patients with chronic obstructive pulmonary disease (COPD).
CTID: null
Phase: Phase 3 Status: Completed
Date: 2004-10-08
A Multicentre, Randomised, Double-Blind, Parallel Group, 24 Week Study to Compare the Effect of the Salmeterol/Fluticasone Propionate Combination Product (SERETIDE) 50/250mcg with Salmeterol 50mcg Both Delivered Twice Daily via the DISKUS/ACCUHALER Inhaler on Lung Function and Dyspnoea in Subjects With Chronic Obstructive Pulmonary Disease (COPD).
CTID: null
Phase: Phase 3 Status: Completed
Date: 2004-06-30
A randomised, double-blind, placebo- and active-controlled, parallel-arm, multicentre study to assess efficacy, pharmacokinetics, safety and tolerability of multiple dose levels of abediterol administered once daily for four weeks, in patients with asthma symptomatic on inhaled corticosteroids
CTID: null
Phase: Phase 2 Status: Ongoing, Prematurely Ended, Completed
Date:
A PHASE IIa, RANDOMISED, DOUBLE-BLIND, DOUBLE-DUMMY, PLACEBO AND ACTIVE COMPARATOR CONTROLLED, 5-WAY CROSSOVER CLINICAL TRIAL TO ASSESS THE ACTIVITY, SAFETY, TOLERABILITY AND PHARMACOKINETICS OF SINGLE DOSES OF LAS 100977 ADMINISTERED BY INHALATION TO ASTHMA PATIENTS
CTID: null
Phase: Phase 2 Status: Completed
Date:
Open-label, single-center, randomized and parallel-group study to exmamine the effects of tulobuterol and salmeterol on the peripheral airway resistance in asthma.
CTID: UMIN000001177
Phase: Phase IV Status: Complete: follow-up complete
Date: 2008-06-06