| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1g |
|
||
| 5g |
|
||
| 10g |
|
||
| 25g |
|
||
| 50g |
|
||
| Other Sizes |
|
| 靶点 |
Endogenous Metabolite
Uracil is a natural pyrimidine nucleobase found in RNA. In itself, it is not a drug with a specific protein target like an enzyme or receptor. Its derivatives and analogs are designed to target various biological processes. This review discusses many uracil derivatives with different targets, but specific target information (e.g., IC50, Ki) for the parent compound uracil is not provided. The derivatives mentioned target enzymes such as thymidine phosphorylase (TP), glycogen phosphorylase (GP), Plasmodium falciparum dUTPase (PfdUTPase), HIV-1 reverse transcriptase, and others. [1] Uracil itself does not directly bind to specific therapeutic targets in a drug-like manner; rather, it functions as a natural metabolite and a building block for RNA synthesis. However, uracil derivatives exert their effects through various mechanisms, including inhibition of thymidylate synthase, viral DNA polymerases, dipeptidyl peptidase-4 (DPP-4), and modulation of P2 receptors. In the clinic, uracil is utilized as a biochemical modulator in combination chemotherapy (e.g., UFT regimen: uracil + ftorafur), where it competitively inhibits dihydropyrimidine dehydrogenase (DPD), thereby prolonging the half-life and enhancing the bioavailability of 5-fluorouracil (5-FU). |
|---|---|
| 体外研究 (In Vitro) |
尿嘧啶本身作为单一药物时直接生物学活性有限。然而,其衍生物显示出显著的体外药理效应。例如,3-氧杂尿嘧啶(一种尿嘧啶类似物)在103 μM浓度下对多种人肿瘤细胞系显示出显著的细胞毒性活性,包括胰腺癌(RWP-2, MiaPaCa-2, PANC-1)、结肠癌(HT-29)、神经内分泌癌(COLO 320DM)和肺癌(SK-MES-1)细胞。此外,尿嘧啶类化合物对H1N1流感病毒(EC50低至0.3 μM)、柯萨奇病毒B4等病毒感染表现出体外抑制活性,并具有抗菌/抗真菌特性。尿嘧啶衍生物在细胞培养中还显示出对布氏锥虫和墨西哥利什曼原虫等病原体的抑制作用。
本文综述了近年来尿嘧啶衍生物的研究进展。本文还旨在讨论开发用于各种生物靶标的更强效、更特异的尿嘧啶类似物的潜在未来方向。尿嘧啶被认为是药物发现中的特权结构,具有广泛的生物活性和合成可及性。抗病毒和抗肿瘤是尿嘧啶类似物最广泛报道的两种活性,但它们也具有除草、杀虫和杀菌活性。它们的抗病毒潜力是基于对病毒复制途径中关键步骤的抑制,从而产生对HIV、乙型肝炎和丙型肝炎、疱疹病毒等的有效活性。尿嘧啶衍生物,如5-氟尿嘧啶或5-氯尿嘧啶是第一个产生的药理学活性衍生物。选择性差限制了其治疗应用,导致胃肠道或中枢神经毒性的高发生率。为了解决这些问题,已经对尿嘧啶结构进行了大量的修饰,从而开发出具有更好的药理学和药代动力学特性的衍生物,包括增加的生物活性、选择性、代谢稳定性、吸收和低毒性。作为生物活性剂的新型尿嘧啶和稠合尿嘧啶衍生物的研究涉及嘧啶环N(1)、N(3)、C(5)和C(6)位取代基的修饰。特别介绍了这类类似物的合成方法和生物学研究,如:5-氟尿嘧啶或5-氯尿嘧啶衍生物、替加富尔类似物、尿嘧啶的阿拉伯吡喃单核糖苷、尿嘧啶吡喃葡糖双核糖苷、脂质多霉素、caprazamycin或膜霉素、三丁基尿苷类似物、脲嘧啶的硝基或氰基衍生物、尿嘧啶喹唑啉酮、尿嘧啶吲哚或尿嘧啶-靛蓝偶联物、桥接多亚甲基链中含有一个或两个尿嘧啶单元和氮原子的嘧啶类化合物等。本文还讨论了具有与其他杂环环化的尿嘧啶环的融合尿嘧啶的合成和生物学活性[1]。 本文是一篇综述文章,总结了众多尿嘧啶衍生物的生物学活性。综述重点在于其修饰类似物的活性。例如,5-氟尿嘧啶(5-FU)抑制RNA复制酶。许多合成的尿嘧啶衍生物在细胞实验中显示出抗癌、抗病毒、抗菌和抗寄生虫活性。[1] 提及了一项关于人胸腺嘧啶DNA糖基化酶(hTDG)从DNA中切除尿嘧啶及其卤代衍生物(5-氟尿嘧啶/FU、5-氯尿嘧啶/CIU、5-溴尿嘧啶/BrU)的研究。从GX底物中切除这些碱基的活性(kmax)显著依赖于5'侧翼碱基对。例如,与CpGCIU相比,从TpGCIU、GpGCIU和ApGCIU序列中切除CIU的kmax分别降低了6倍、11倍和82倍。从任何5'侧翼序列的G FU、GCIU和GBrU中切除的活性达到或超过了从CpG T序列中切除的活性。[1] |
| 体内研究 (In Vivo) |
小鼠体内研究表明,尿嘧啶衍生物具有中枢神经系统效应,低剂量尿嘧啶和胸腺嘧啶可增加自发活动,高剂量则降低自发活动。作为UFT(尿嘧啶+替加氟)的组分,该联合方案在癌症患者中显示出临床疗效。一项I期研究报告,在既往未接受过治疗的晚期结直肠癌患者中,接受UFT(350 mg/m²)联合亚叶酸治疗的患者有2/8达到部分缓解。有限的小鼠体内研究表明,3-氧杂尿嘧啶(3.23 mg/kg,腹腔注射,每日一次,持续两周)显示出抗病毒效果且无明显毒性。
文中提及了一些衍生物的体内数据,例如尿嘧啶衍生物(6-氨基-1-甲基-5-亚硝基尿嘧啶,MANU)的金(I)配合物在实验性神经胶质瘤动物模型中,治疗七天后将肿瘤生长抑制至约十分之一。[1] |
| 酶活实验 |
尿嘧啶作为底物参与DPD(二氢嘧啶脱氢酶)酶活性测定,以评估代谢能力。尿嘧啶负荷试验的标准方案:口服给予尿嘧啶500 mg/m²。在尿嘧啶给药后的多个时间点(例如t = 15、30、45、60、80、100、120、150、180和220-240分钟)采集血液样本(2-4 mL)于肝素锂管中。通过HPLC-UV定量检测血浆中尿嘧啶及其代谢物二氢尿嘧啶(DHU)的水平。在外周血单核细胞中测定DPD活性。计算特定时间点的U/DHU比值以评估全身DPD活性,比值升高提示DPD缺乏。
描述了用于评估尿嘧啶衍生物的改良胸苷磷酸化酶(TP)生物测定法。该测定使用在大肠杆菌中表达的重组大肠杆菌TP作为酶,胸苷作为底物。使用该测定评估合成化合物的抑制活性。例如,一个5-氯尿嘧啶连接的吡唑并[1,5-a][1,3,5]三嗪衍生物(化合物262,R = 4-F5S-苯基)的IC50为0.04 µM,在相同条件下比先导化合物7-脱氮黄嘌呤(7DX,IC50 = 32 µM)强约800倍。该化合物被发现是非竞争性抑制剂。[1] 提及了用于评估恶性疟原虫和硕大利什曼原虫dUTP酶以及人酶抑制剂的测定法。化合物针对重组酶进行测试以衡量选择性。例如,一个三苯甲基化的脱氧尿苷类似物(5'-三苯甲氨基-2',5'-二脱氧尿苷 124,X=OH)对恶性疟原虫dUTP酶的Ki为0.2 µM,与人酶相比选择性超过200倍。[1] 提及了评估尿嘧啶衍生物作为糖原磷酸化酶(GP)抑制剂的动力学实验。鉴定出的最佳抑制剂是1-(β-D-吡喃葡萄糖基)-5-乙炔基尿嘧啶(99),其Ki为4.7 µM。[1] |
| 细胞实验 |
评估尿嘧啶衍生物细胞毒性的典型细胞增殖抑制实验:将人肿瘤细胞系(例如HT-29结肠癌细胞)以约5 × 10³ - 1 × 10⁴个/孔的密度接种于96孔板的适宜培养基中。贴壁过夜后,用不同浓度的待测化合物(尿嘧啶或其衍生物)处理细胞。化合物溶于DMSO并用培养基稀释,浓度范围通常为0.1 - 200 μM。在37°C、5% CO₂条件下孵育48-72小时后,采用MTT法、XTT法或[³H]-胸腺嘧啶掺入法评估细胞活力。设置未处理对照组和溶剂对照组(DMSO)作为比较。
提及了磺酰罗丹明B(SRB)法用于评估细胞毒性活性。化合物针对人癌细胞系进行测试,如宫颈癌(HeLa)、口腔癌(KB)和乳腺癌(MCF-7)。例如,一个3'-叠氮-2',3'-双脱氧-5-氟尿苷的磷酰胺衍生物(25,R = CH2CH3)在所有测试的癌细胞中显示出最高的活性,远高于母体核苷。[1] 提及了MTT法用于对人癌细胞系(如HeLa、MCF-7、DU145)进行细胞毒性评估。例如,尿嘧啶-靛红结合物(107)经过评估,其中一些对DU145细胞的IC50值低至13.90 µM。[1] 描述了抗病毒活性测定。化合物针对病毒进行测试,如1型单纯疱疹病毒(HSV-1)、水痘-带状疱疹病毒(VZV)、人巨细胞病毒(HCMV)、登革热病毒(DENV)和黄热病毒(YFV),使用适当的细胞系(例如,CCL-81用于HSV,AD-169用于HCMV,MDCK用于流感)。例如,5-(噻吩-2-基)-2'-脱氧尿苷(40)对HSV-1表现出显著的活性,一些卤代类似物(41)与溴夫定(BVDU)效力相当。一个氟环丙基尿嘧啶核苷(55)显示出中度的抗HCMV活性(在AD-169株中为10.61 µg/mL)。[1] 对一些不饱和尿嘧啶核苷测量了在正常人肠细胞系(H4)和肿瘤细胞系(Caco-2、黑色素瘤、MCF-7)上的细胞毒性(CC50)。发现化合物65在MCF-7乳腺癌细胞系中有效。[1] 对于尿嘧啶衍生的嘧啶环芳烃,研究了其对革兰氏阴性菌(铜绿假单胞菌、大肠杆菌)、革兰氏阳性菌(金黄色葡萄球菌、枯草芽孢杆菌、粪肠球菌)、病原真菌(黑曲霉、须毛癣菌、烟曲霉)和酵母(白色念珠菌)的抗菌和抗真菌活性。活性随聚亚甲基链长度的增加而增加,并在引入正癸基取代基时显著增强。[1] |
| 动物实验 |
评估包括尿嘧啶在内的嘧啶类中枢神经系统效应的方案:成年雄性C-57小鼠通过腹腔注射或胃肠外途径给药。使用活动笼或类似监测系统在给药后特定时间段(如20-60分钟)测量自发活动。低剂量(如10-50 mg/kg)用于测试兴奋效应,高剂量(如50-200 mg/kg)用于测试抑制效应。结果表示为相对于基线活动的变化百分比,并计算兴奋和抑制的ED50值。
该综述描述了一项在实验性胶质瘤动物模型中,对尿嘧啶衍生物(6-氨基-1-甲基-5-亚硝基尿嘧啶,MANU)的金(I)配合物进行的体内研究。结果显示,治疗7天后,与载体对照组相比,该金化合物使肿瘤生长减少了约十分之一。[1] |
| 药代性质 (ADME/PK) |
在健康志愿者中,口服尿嘧啶(500 mg/m²)后的药代动力学参数如下:AUC尿嘧啶(T=180)= 865.1 ± 240.3 mg·min/L,Cmax尿嘧啶 = 14.4 ± 4.7 mg/L,AUC二氢尿嘧啶(T=220)= 439.3 ± 134.5 mg·min/L。在DPD活性正常的个体中,尿嘧啶/二氢尿嘧啶比值随时间降低,从100分钟时的约0.9降至180分钟时的约0.1。在DPD缺乏患者中,所有时间点的U/DHU比值均显著升高,表明尿嘧啶向二氢尿嘧啶的代谢转化减少。尿嘧啶吸收迅速,主要在肝脏和外周组织中经DPD代谢。
该综述指出,5-氟尿嘧啶(5-FU,一种尿嘧啶衍生物)选择性差、半衰期短、分布广泛,限制了其治疗应用。然而,该综述并未提供母体化合物尿嘧啶的具体ADME/PK参数(吸收、分布、代谢、排泄、半衰期、生物利用度)。[1] 对于一些作为恶性疟原虫dUTPase抑制剂开发的三苯甲基化尿嘧啶类似物,初步ADME研究表明,部分先导化合物具有药物活性。但该综述也未给出具体参数。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
尿嘧啶本身作为内源性代谢物,在生理浓度下被认为是安全的。然而,在联合化疗方案(UFT)的背景下,主要剂量限制性毒性为胃肠道反应,包括腹泻、恶心和呕吐。在既往接受过化疗治疗的患者中,UFT 350 mg/m²(尿嘧啶+替加氟)剂量下观察到剂量限制性毒性。轻度疲劳和一过性高胆红素血症也常见报告。在分子水平上,DPD缺乏患者暴露于5-FU或含尿嘧啶前药时,发生严重毒性的风险显著增高,因为遗传性缺乏导致毒性代谢物蓄积。作为化学纯品,尿嘧啶的RTECS编号为YQ8650000;常规操作注意事项包括避免接触强氧化剂。
该综述指出,5-氟尿嘧啶 (5-FU) 因其选择性差而具有胃肠道或中枢神经系统毒性等副作用。[1] 对于一系列尿嘧啶-苯并二氧杂环庚烷杂合物(环状和非环状 5-FU O,N-缩醛 150、151),所有化合物对 MCF-7 细胞的 IC50 值均在微摩尔范围内。其中一种化合物 (151, R1=NO2, R2=H) 的细胞毒性最强。另一种化合物 (151, R1=R2=H) 可诱导 MCF-7 细胞凋亡和 G0/G1 期细胞周期阻滞。[1] 在对 5-[1-(2-卤代乙基(或硝基)乙氧基-2-碘乙基)]-2'-脱氧尿苷 (46) 的研究中,所有化合物均表现出较低的宿主细胞毒性。 [1] 对于 MANU 的金(I)配合物,血清参数分析表明治疗后不良反应较少。[1] 5,6-取代的 1-[(2-羟基乙氧基)甲基]尿嘧啶的构效关系分析表明,细胞毒性取决于亲脂性和空间位阻参数。[1] |
| 参考文献 | |
| 其他信息 |
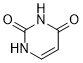
尿嘧啶是一种常见的天然嘧啶核碱基,其嘧啶环的2位和4位分别被两个氧代基团取代。它存在于RNA中,与腺嘌呤配对,并在DNA转录过程中取代胸腺嘧啶。尿嘧啶具有多种功能,包括作为前药、人体代谢物、大型蚤(Daphnia magna)代谢物、酿酒酵母(Saccharomyces cerevisiae)代谢物、大肠杆菌(Escherichia coli)代谢物、小鼠代谢物以及过敏原。它既是嘧啶核碱基,也是嘧啶酮。它是 (4S)-4-羟基-3,4-二氢嘧啶-2(1H)-酮的互变异构体。
尿嘧啶是大肠杆菌(K12 株、MG1655 株)中发现或产生的代谢产物。 据报道,尿嘧啶存在于哈米格拉·阿维拉尼亚菌、小球形拟球菌以及其他有相关数据的生物体中。 尿嘧啶是酿酒酵母中发现或产生的代谢产物。 它是核酸 RNA 中的四种核苷酸碱基之一。 另见:嘧啶(亚类)。 背景:尿嘧啶是一种常见的天然嘧啶衍生物,是 RNA 中的四种核碱基之一,它与腺嘌呤结合。在 DNA 中,它被胸腺嘧啶取代。它可以被认为是胸腺嘧啶的去甲基化形式。尿嘧啶发生酰胺-亚胺酸(内酰胺-内亚胺)互变异构,在 pH 7 时以内酰胺形式为主。尿嘧啶是一种弱酸。[1] 优势结构:尿嘧啶因其广泛的生物活性、易于合成以及在 N1、N3、C5 和 C6 位点修饰后可赋予药物样特性,被认为是药物发现中的优势结构。[1] 衍生物活性:尿嘧啶类似物最常被报道具有抗病毒和抗肿瘤活性,但也具有除草、杀虫和杀菌活性。[1] 机制解析(例如 5-氟尿嘧啶):5-氟尿嘧啶 (5-FU) 是一种抗代谢嘧啶类似物。由于其结构与尿嘧啶相似,但化学性质不同,它能抑制RNA复制酶,从而阻断RNA合成,抑制癌细胞生长。[1] 代谢转化:该综述描述了尿嘧啶衍生物(金担子菌素A中的β-羟基-N-甲基缬氨酸/βHOMeVal)在碱性条件下的特定化学转化,涉及逆醛醇缩合反应或脱水反应,但这并非尿嘧啶本身的通用代谢途径。[1] |
| 分子式 |
C4H4N2O2
|
|---|---|
| 分子量 |
112.0868
|
| 精确质量 |
112.027
|
| 元素分析 |
C, 42.86; H, 3.60; N, 24.99; O, 28.55
|
| CAS号 |
66-22-8
|
| 相关CAS号 |
66-22-8 (uracil); 3083-77-0 [1-beta-D-Arabinofuranosyluracil (Uracil 1-β-D-arabinofuranoside)]; 462-88-4 (Ureidopropionic acid); 504-07-4 (5,6-Dihydrouracil); 66-75-1 (Uramustine, Uracil mustard); 141-90-2 (2-Thiouracil); 58-96-8 (Uridin; β-Uridine)
|
| PubChem CID |
1174
|
| 外观&性状 |
Typically exists as white to light yellow solids at room temperature
|
| 密度 |
1.5±0.1 g/cm3
|
| 沸点 |
440.5±37.0 °C at 760 mmHg
|
| 熔点 |
330°C
|
| 闪点 |
220.2±26.5 °C
|
| 蒸汽压 |
0.0±1.1 mmHg at 25°C
|
| 折射率 |
1.640
|
| LogP |
-2.55
|
| tPSA |
65.72
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
8
|
| 分子复杂度/Complexity |
161
|
| 定义原子立体中心数目 |
0
|
| SMILES |
O=C1N([H])C([H])=C([H])C(N1[H])=O
|
| InChi Key |
ISAKRJDGNUQOIC-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C4H4N2O2/c7-3-1-2-5-4(8)6-3/h1-2H,(H2,5,6,7,8)
|
| 化学名 |
Pyrimidine-2,4(1H,3H)-dione
|
| 别名 |
2,4-Dioxopyrimidine; 2,4-Pyrimidinedione; Pirod; uracil; 66-22-8; 2,4-Dihydroxypyrimidine; 2,4(1H,3H)-Pyrimidinedione; pyrimidine-2,4(1H,3H)-dione; pyrimidine-2,4-diol; Pyrod; 2,4-Pyrimidinediol; Pyrod.
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ≥ 25 mg/mL (~223.04 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (22.30 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (22.30 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (22.30 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 8.9214 mL | 44.6070 mL | 89.2140 mL | |
| 5 mM | 1.7843 mL | 8.9214 mL | 17.8428 mL | |
| 10 mM | 0.8921 mL | 4.4607 mL | 8.9214 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。