| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
|
| 靶点 |
BTK/Bruton tyrosine kinase
(±)-Zanubrutinib (BGB-3111) targets Bruton's tyrosine kinase (BTK), a core component of the B-cell receptor (BCR) signaling pathway; it exhibits nanomolar-level inhibitory activity against BTK, [1] |
|---|---|
| 体外研究 (In Vitro) |
(±)-Zanubrutinib ((±)-BGB-3111) 在生化和细胞测定中均表现出纳摩尔级 Btk 抑制活性。 (±)-Zanubrutinib 抑制 BCR 聚集触发的 Btk 自磷酸化,阻断下游 PLC-γ2 信号传导,并有效抑制多种 MCL 和 DLBCL 细胞系的细胞增殖。当涉及包括 ITK 在内的一组激酶时,与 PCI-32765 相比,(±)-Zanubrutinib 表现出的脱靶活性要有限得多[1]。
1. 在生化和细胞实验中,(±)-Zanubrutinib对BTK表现出纳摩尔级的抑制效力,相较于伊布替尼,其对BTK的抑制具有高度选择性,对ITK等其他激酶的脱靶活性显著降低[1] 2. 在多种套细胞淋巴瘤(MCL)和弥漫大B细胞淋巴瘤(DLBCL)细胞系中,(±)-Zanubrutinib可抑制由BCR聚集触发的BTK自磷酸化,阻断下游PLC-γ2信号传导,并强效抑制肿瘤细胞增殖[1] 3. 伊布替尼会通过抑制ITK激酶活性拮抗利妥昔单抗诱导的抗体依赖性细胞介导的细胞毒性(ADCC),而(±)-Zanubrutinib对ITK的抑制作用极弱,其抑制利妥昔单抗诱导的ADCC的能力比伊布替尼弱至少10倍,具体表现为对利妥昔单抗诱导的NK细胞IFN-γ分泌及对MCL细胞的体外细胞毒性的抑制作用显著降低[1] |
| 体内研究 (In Vivo) |
(±)-Zanubrutinib 对注射到小鼠尾静脉并皮下或全身移植的 REC-1 MCL 异种移植物产生剂量依赖性抗肿瘤作用。在放置在皮下的异种移植物中。一项最初的 14 天大鼠毒性研究表明,(±)-Zanubrutinib 具有高度良好的耐受性,剂量高达 250 mg/kg/天也不会达到最大耐受剂量 (MTD)[1]。
1. 在小鼠BTK占据实验中,(±)-Zanubrutinib以剂量依赖性方式占据PBMC和脾脏等靶器官中的BTK,且在这些器官中的效力比伊布替尼高约3倍[1] 2. (±)-Zanubrutinib对REC-1 MCL异种移植小鼠(皮下和尾静脉注射的系统模型)均表现出剂量依赖性的抗肿瘤效果:在皮下移植模型中,(±)-Zanubrutinib以2.5 mg/kg每日两次(BID)给药,与伊布替尼50 mg/kg每日一次(QD,临床相关剂量)的抗肿瘤活性相当[1] 3. 在REC-1 MCL系统移植模型中,(±)-Zanubrutinib25 mg/kg BID给药组小鼠的中位生存期显著长于伊布替尼50 mg/kg QD及BID给药组[1] 4. 在ABC亚型DLBCL(TMD-8)皮下移植模型中,(±)-Zanubrutinib展现出比伊布替尼更优的抗肿瘤活性[1] |
| 酶活实验 |
在生化和细胞实验中,BGB-3111显示出纳米级的BTK抑制活性。在一些MCL和DLBCL细胞系中,BGB-3111抑制BCR聚集引发的BTK自磷酸化,阻断下游PLC-γ2信号传导,并有效抑制细胞增殖。与依鲁替尼相比,BGB-3111对一组激酶(包括ITK)显示出更有限的脱靶活性。虽然伊鲁替尼显著抑制利妥昔单抗诱导的NK细胞IFN-γ分泌和对套细胞淋巴瘤细胞的体外细胞毒性,但BGB-3111对利妥昔单抗诱导的ADCC的抑制作用至少弱于伊鲁替尼的10倍,这与其较弱的ITK抑制活性一致。[1]
|
| 细胞实验 |
1. BTK信号抑制实验:培养MCL和DLBCL细胞系,在(±)-Zanubrutinib不同浓度处理下诱导BCR聚集,裂解细胞后通过相关生化方法检测BTK自磷酸化水平及下游PLC-γ2信号通路激活情况,评估药物对BCR-BTK信号通路的抑制作用[1]
2. 细胞增殖实验:将MCL和DLBCL细胞系接种于培养板,加入(±)-Zanubrutinib处理,孵育一段时间后采用合适的细胞活力检测方法评估细胞增殖情况,明确药物的抗增殖效应[1] 3. ADCC抑制实验:将NK细胞与MCL靶细胞和利妥昔单抗共培养,同时加入(±)-Zanubrutinib或伊布替尼处理,检测NK细胞的IFN-γ分泌水平并评估其对MCL细胞的体外细胞毒性,对比两种药物对利妥昔单抗诱导的ADCC的影响[1] |
| 动物实验 |
在小鼠 BTK 受体占有率测定中,BGB-3111 治疗可导致剂量依赖性的 BTK 受体占有率,并且在包括外周血单核细胞 (PBMC) 和脾脏在内的靶器官中,其效力约为伊布替尼的 3 倍。BGB-3111 对皮下或尾静脉注射全身移植的 REC-1 MCL 异种移植瘤具有剂量依赖性的抗肿瘤作用。在皮下移植的异种移植瘤模型中,BGB-3111 2.5 mg/kg BID 的活性与伊布替尼 50 mg/kg QD(临床相关剂量)的活性相似。在全身移植模型中,BGB-3111 25 mg/kg BID 组的中位生存期显著长于伊布替尼 50 mg/kg QD 组和 BID 组。在ABC亚型DLBCL(TMD-8)皮下异种移植模型中,BGB-3111也表现出比伊布替尼更好的抗肿瘤活性。初步的14天大鼠毒性研究表明,BGB-3111耐受性良好,剂量高达250mg/kg/天时未达到最大耐受剂量(MTD)。[1]
1. 小鼠BTK占有率测定:向小鼠给予不同剂量的(±)-Zanubrutinib;在特定时间点收集外周血单核细胞(PBMC)和脾脏组织,并检测这些组织中BTK的占有率,以评估药物的靶点结合情况。[1] 2. REC-1 MCL皮下异种移植模型:将REC-1 MCL细胞皮下移植到小鼠体内;当肿瘤达到一定体积时,给予(±)-Zanubrutinib 2.5 mg/kg BID,并以ibrutinib 50 mg/kg QD作为对照;定期监测肿瘤生长情况以评估药物的抗肿瘤活性[1] 3. REC-1 MCL 系统性异种移植模型:将REC-1 MCL细胞经尾静脉注射到小鼠体内,建立系统性肿瘤模型;给予(±)-Zanubrutinib 25 mg/kg BID,并以ibrutinib 50 mg/kg QD/BID作为对照;记录小鼠的生存时间以比较不同治疗方案的抗肿瘤疗效[1] 4. TMD-8 DLBCL 皮下异种移植模型:将TMD-8 DLBCL细胞皮下移植到小鼠体内;分别给予(±)-Zanubrutinib和ibrutinib,并监测肿瘤生长情况以评价(±)-Zanubrutinib的抗肿瘤活性[1] 5. 大鼠毒性研究:连续14天给予大鼠(±)-Zanubrutinib,剂量最高达250 mg/kg/天;观察大鼠的一般状况,并评价最大耐受剂量(MTD)[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. 在一项为期 14 天的大鼠初步毒性研究中,(±)-Zanubrutinib 在高达 250 mg/kg/天的剂量下耐受性良好,且在研究期间未达到最大耐受剂量 (MTD) [1]
|
| 参考文献 | |
| 其他信息 |
另见:Zanubrutinib(注释已移至)。
1. BTK 是 BCR 通路的重要组成部分,也是 B 细胞恶性肿瘤的新型治疗靶点;第一代不可逆 BTK 抑制剂伊布替尼具有良好的临床活性,但可通过抑制 ITK 激酶活性拮抗利妥昔单抗诱导的 ADCC,从而限制其临床应用 [1] 2. (±)-Zanubrutinib 是一种新型高选择性第二代 BTK 抑制剂,目前正在进行血液肿瘤治疗的临床研究 [1] 3. (±)-Zanubrutinib 不影响利妥昔单抗诱导的 ADCC,并且在异种移植模型中具有比伊布替尼更好的抗肿瘤疗效,支持进一步开展该化合物作为单药或与抗 CD20 抗体联合用于血液肿瘤治疗的临床研究 [1] |
| 分子式 |
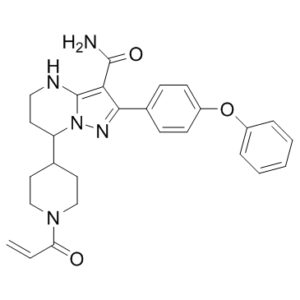
C27H29N5O3
|
|
|---|---|---|
| 分子量 |
471.56
|
|
| 精确质量 |
471.227
|
|
| 元素分析 |
C, 68.77; H, 6.20; N, 14.85; O, 10.18
|
|
| CAS号 |
1633350-06-7
|
|
| 相关CAS号 |
Zanubrutinib;1691249-45-2;(R)-Zanubrutinib;1691249-44-1;Zanubrutinib-d5; 1633350-06-7 (racemic); 1691249-45-2 (S-isomer); 1691249-44-1 (R-isomer)
|
|
| PubChem CID |
135905454
|
|
| 外观&性状 |
White to light yellow solid
|
|
| 密度 |
1.3±0.1 g/cm3
|
|
| 沸点 |
713.4±60.0 °C at 760 mmHg
|
|
| 闪点 |
385.2±32.9 °C
|
|
| 蒸汽压 |
0.0±2.3 mmHg at 25°C
|
|
| 折射率 |
1.680
|
|
| LogP |
3.64
|
|
| tPSA |
103
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
5
|
|
| 可旋转键数目(RBC) |
6
|
|
| 重原子数目 |
35
|
|
| 分子复杂度/Complexity |
756
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
O=C(C([H])=C([H])[H])N1C([H])([H])C([H])([H])C([H])(C([H])([H])C1([H])[H])C1([H])C([H])([H])C([H])([H])N([H])C2=C(C(N([H])[H])=O)C(C3C([H])=C([H])C(=C([H])C=3[H])OC3C([H])=C([H])C([H])=C([H])C=3[H])=NN12
|
|
| InChi Key |
RNOAOAWBMHREKO-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C27H29N5O3/c1-2-23(33)31-16-13-18(14-17-31)22-12-15-29-27-24(26(28)34)25(30-32(22)27)19-8-10-21(11-9-19)35-20-6-4-3-5-7-20/h2-11,18,22,29H,1,12-17H2,(H2,28,34)
|
|
| 化学名 |
2-(4-phenoxyphenyl)-7-(1-prop-2-enoylpiperidin-4-yl)-4,5,6,7-tetrahydropyrazolo[1,5-a]pyrimidine-3-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (4.41 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: 2.08 mg/mL (4.41 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (4.41 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1206 mL | 10.6031 mL | 21.2062 mL | |
| 5 mM | 0.4241 mL | 2.1206 mL | 4.2412 mL | |
| 10 mM | 0.2121 mL | 1.0603 mL | 2.1206 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。