| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
The primary target of Dichlorphenamide is the carbonic anhydrase (CA) enzyme family, with inhibitory activity against cytosolic CA II and membrane-bound CA IV—key isoforms involved in aqueous humor secretion in the eye. Its pharmacodynamic effects are mediated by CA inhibition [1]
|
|---|---|
| 体内研究 (In Vivo) |
双氯苯酰胺可局部降低雄性白化兔的眼压[1]。
1. 青光眼治疗中的降眼压效果:在一项开放性青光眼患者临床研究(n=24)中,口服Dichlorphenamide 25 mg,每日2次,连续2周。基线眼压为32-38 mmHg;治疗1周后,眼压降低25-30%(降至23-27 mmHg);治疗2周后,眼压稳定在22-26 mmHg(较基线降低28-32%)。2周观察期内未出现降眼压效果耐受现象 [1] 。 2. 难治性癫痫治疗中的抗癫痫效果:在一项36例难治性癫痫患者(对≥2种抗癫痫药无应答)的回顾性研究中,Dichlorphenamide作为辅助治疗药物使用,剂量范围为每日50-150 mg(分2-3次服用)。治疗3个月后: - 50%患者(18/36)癫痫发作频率降低≥50%; - 25%患者(9/36)癫痫发作频率降低30-50%; - 25%患者(9/36)癫痫发作频率无显著变化。 其中,全身强直-阵挛发作对药物应答最佳(此类患者中62%发作频率降低≥50%)[2] 。 |
| 动物实验 |
动物/疾病模型:雄性白化兔(约2.5 kg)
剂量:二氯苯胺钠50 μL 10%水溶液或2 mg/kg、6 mg/kg 给药途径:50 μL滴眼液或灌胃(口服)2 mg/kg或6 mg/kg,持续5小时。 实验结果:滴眼30分钟后眼压显著降低。口服1小时后眼压降低。与口服相比,滴眼后虹膜和睫状体中的药物浓度显著升高。滴眼后虹膜和睫状体中的药物浓度显著降低。 |
| 毒性/毒理 (Toxicokinetics/TK) |
蛋白质结合
55% 1. 青光眼患者的不良反应:口服二氯苯胺(25 mg,每日两次)在大多数青光眼患者中耐受性良好。轻微且短暂的不良反应包括: - 胃肠道症状:恶心(12.5%,3/24 例患者),口干(16.7%,4/24 例患者); - 肾脏症状:尿量增加(20.8%,5/24 例患者) . 2. 癫痫患者的不良反应:在辅助抗癫痫研究中,二氯苯胺(每日 50-150 mg)的不良反应包括: - 中枢神经系统 (CNS) 症状:头晕(27.8%,10/36 例患者),嗜睡(19.4%,7/36 例患者),头痛(13.9%,5/36 例患者); - 胃肠道症状:厌食(16.7%,6/36 例患者),恶心(11.1%,4/36 例患者); - 电解质紊乱(轻度低钾血症):8.3%(3/36 例患者),口服补钾后缓解[2] 。 |
| 参考文献 | |
| 其他信息 |
根据州或联邦政府的标签要求,二氯苯胺可能具有发育毒性。
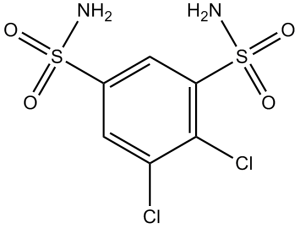
二氯苯胺是一种磺胺类药物,其化学名称为苯-1,3-二磺酰胺,其中4位和5位的氢原子被氯原子取代。它是一种口服碳酸酐酶抑制剂,可部分抑制眼内房水的分泌(流入),从而降低眼内压。它用于治疗青光眼。它是一种EC 4.2.1.1(碳酸酐酶)抑制剂,也是一种抗青光眼药物和眼科药物。它是一种磺胺类药物,也是一种二氯苯类药物。 一种用于治疗青光眼的碳酸酐酶抑制剂。 二氯苯胺是一种碳酸酐酶抑制剂。二氯苯那敏的作用机制是作为碳酸酐酶抑制剂。 一种用于治疗青光眼的碳酸酐酶抑制剂。 药物适应症 用于辅助治疗:慢性单纯性(开角型)青光眼、继发性青光眼,以及急性闭角型青光眼术前,以降低眼压,延缓手术。 作用机制 碳酸酐酶抑制剂通过部分抑制房水的分泌(流入)来降低眼压,但其作用机制尚未完全阐明。有证据表明,在碳酸酐酶的作用下,二氧化碳在睫状体中水合产生HCO₃⁻离子,HCO₃⁻离子扩散到后房。后房的Na⁺和HCO₃⁻离子浓度高于血浆,因此呈高渗状态。水随后通过渗透作用被吸引到后房,导致压力下降。 磷酸盐尿可能与肾脏产生的环磷酸腺苷(cAMP)的直接刺激有关。该药物的作用类似于甲状旁腺激素,可增强尿液中磷酸盐和环磷酸腺苷的排泄,这与它拮抗激素对骨骼的作用相反。/乙酰唑胺/ ……抑制……碳酸酐酶。……是非竞争性抑制。……该酶通常在组织中大量存在。肾脏中超过99%的酶活性必须被抑制,生理效应才会显现。酶是抑制剂结合的主要组织成分。乙酰唑胺引起的尿液变化可能归因于肾小管对(+)H分泌的抑制。现有证据表明,乙酰唑胺对近端小管的影响大于远端小管,对升支几乎没有影响。磷酸盐尿症……用作利尿作用定位的指标……/乙酰唑胺/ ……增加尿液中碳酸氢盐和固定阳离子(主要是钠)的排泄。结果,细胞外液中碳酸氢盐浓度降低,导致代谢性酸中毒。……肾脏对乙酰唑胺的反应显著降低……/但是/利尿反应增强。乙酰唑胺 该药物通过降低脑脊液分泌速率来降低眼内压。 治疗用途 碳酸酐酶抑制剂 ……用于治疗原发性青光眼、继发性青光眼急性期以及术前控制眼内压。……虽然它具有利尿作用,但不推荐用于此目的。 ……该药物已被发现可以抑制癫痫发作并降低脑脊液生成速率。乙酰唑胺 ……降低脑脊液生成速率;青光眼患者的眼内压相应降低。该药物的这种作用似乎与全身酸碱平衡无关。/乙酰唑胺/ 有关二氯苯那敏(共11种)的更多治疗用途(完整)数据,请访问HSDB记录页面。 药物警告 有研究表明,术后(虹膜切除术后)使用该药物可能会通过缩小术后引流滤泡的大小和延迟前房的重建,从而对滤过手术的结果产生不利影响。碳酸酐酶抑制剂 ……应谨慎用于阻塞性肺病患者,因为他们可能出现急性呼吸衰竭。碳酸酐酶抑制剂 利尿作用初期可能令人不适,但由于持续的代谢性酸中毒,在持续治疗过程中会逐渐消退。 /碳酸酐酶抑制剂/ ...应谨慎用于...患有与盐皮质激素活性增强相关的疾病(例如,原发性醛固酮增多症、库欣综合征)的患者以及正在接受排钾药物(例如,噻嗪类利尿剂、袢利尿剂、皮质类固醇)治疗的患者。 /碳酸酐酶抑制剂/ 有关二氯苯胺(共7条)的更多药物警告(完整)数据,请访问HSDB记录页面。 药效学 二氯苯胺是一种口服碳酸酐酶抑制剂,适用于以下疾病的辅助治疗:慢性单纯性(开角型)青光眼、继发性青光眼,以及急性闭角型青光眼术前治疗(需延迟手术以降低眼内压)。碳酸酐酶抑制剂通过部分抑制房水分泌(流入)来降低眼内压。 1. 化学分类和作用机制:二氯苯胺是一种磺胺类衍生的碳酸酐酶(CA)抑制剂。对于青光眼,它抑制眼睫状体中的CA II/IV,减少房水分泌,从而降低眼内压[1] 。其抗癫痫机制在[2]中尚未明确定义,但据推测可能涉及通过抑制碳酸酐酶(CA)来调节脑pH值或离子稳态[2]。 2. 临床适应症: - 已获准用于治疗开角型青光眼和急性闭角型青光眼(辅助治疗以降低眼压)[1]。 - 用作难治性癫痫的辅助治疗,特别是全身强直-阵挛性发作[2]。 3. 剂量特点: - 青光眼:口服剂量为每次25 mg,每日两次;对于眼压降低不充分的患者,剂量可调整为每次50 mg,每日两次[1]。 - 癫痫:辅助口服剂量为每日50-150 mg,分2-3次服用;根据癫痫发作反应和耐受性调整剂量[2] 。 4. 临床局限性:在癫痫患者中,25% 的患者无反应,轻微的中枢神经系统/胃肠道不良反应可能需要减少剂量[2] ;在青光眼患者中,尿量增加可能会影响患者的依从性[1] 。 |
| 分子式 |
C6H6CL2N2O4S2
|
|
|---|---|---|
| 分子量 |
305.16
|
|
| 精确质量 |
303.914
|
|
| CAS号 |
120-97-8
|
|
| 相关CAS号 |
Dichlorphenamide disodium;76382-13-3
|
|
| PubChem CID |
3038
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.8±0.1 g/cm3
|
|
| 沸点 |
590.5±60.0 °C at 760 mmHg
|
|
| 熔点 |
239-241ºC
|
|
| 闪点 |
310.9±32.9 °C
|
|
| 蒸汽压 |
0.0±1.7 mmHg at 25°C
|
|
| 折射率 |
1.635
|
|
| LogP |
0.93
|
|
| tPSA |
137.08
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
6
|
|
| 可旋转键数目(RBC) |
2
|
|
| 重原子数目 |
16
|
|
| 分子复杂度/Complexity |
452
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
GJQPMPFPNINLKP-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C6H6Cl2N2O4S2/c7-4-1-3(15(9,11)12)2-5(6(4)8)16(10,13)14/h1-2H,(H2,9,11,12)(H2,10,13,14)
|
|
| 化学名 |
4,5-dichlorobenzene-1,3-disulfonamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (8.19 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (8.19 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (8.19 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2770 mL | 16.3848 mL | 32.7697 mL | |
| 5 mM | 0.6554 mL | 3.2770 mL | 6.5539 mL | |
| 10 mM | 0.3277 mL | 1.6385 mL | 3.2770 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00494507 | Completed Has Results | Drug: Dichlorphenamide (double-blind) | Hyperkalemic Periodic Paralysis Hypokalemic Periodic Paralysis |
University of Rochester | June 2007 | Phase 3 |
| NCT00004802 | Completed | Drug: dichlorphenamide | Paralysis, Hyperkalemic Periodic Hypokalemic Periodic Paralysis |
National Center for Research Resources (NCRR) |
June 1992 | Phase 3 |