| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| Other Sizes |
|

| 靶点 |
EGFR tyrosine kinase
Gefitinib hydrochloride (Gefitinib) targets epidermal growth factor receptor (EGFR) tyrosine kinase (IC₅₀ = 0.033 μM for EGFR kinase activity; IC₅₀ = 0.054 μM for inhibiting EGF-stimulated tumor cell growth) [1] Gefitinib hydrochloride (Gefitinib) targets human epidermal growth factor receptor 2 (HER2) and inhibits HER2-driven signaling in HER2-overexpressing tumor cells [3] Gefitinib hydrochloride (Gefitinib) targets wild-type EGFR and mutant EGFRvIII: it inhibits wild-type EGFR-mediated phosphorylation, proliferation and anchorage-independent growth at low concentrations; for EGFRvIII-expressing cells, low concentrations (0.01-0.1 μM) increase EGFRvIII phosphotyrosine load and downstream ERK signaling, while high concentrations (1-2 μM) inhibit EGFRvIII-mediated activities [2] |
|---|---|
| 体外研究 (In Vitro) |
长期暴露于表达 EGFRvIII 的细胞后,吉非替尼 (0.01-0.1 mM) 会增加受体的磷酸酪氨酸负载,增加 ERK 信号传导,并刺激增殖和贴壁依赖性生长。这种效应可能是由 EGFRvIII 二聚化的诱导引起的。相反,吉非替尼 (1-2 mM) 均显着降低 EGFRvIII 磷酸酪氨酸负荷、EGFRvIII 介导的增殖和锚定非依赖性生长[1]。吉非替尼 (ZD1839) 的 IC50 为 20 nM,可防止这些 EGF 驱动的未转化细胞生长单层[2]。吉非替尼的 IC50 为 2 μM,可抑制 GLC82 和 CALU-3 细胞的生长[3]。
表皮生长因子受体(EGFR)在许多人类肿瘤中经常被扩增和/或突变,并且来自该受体的异常信号被认为是导致这些肿瘤中出现恶性表型的原因。吉非替尼是一种特异性结合和抑制EGFR酪氨酸激酶的小分子抑制剂,已被证明可以抑制一系列过表达EGFR的肿瘤细胞的生长、增殖、存活和侵袭。然而,吉非替尼的临床反应与EGFR水平和活性没有相关性,这表明下游信号传导和突变等其他分子机制在预测临床反应方面可能很重要。因此,我们研究了特异性EGFR抑制剂吉非替尼对表达天然存在的组成型活性EGFR变体EGFRvIII、低非转化水平EGFR和高转化水平EGFR的细胞的磷酸化水平、信号传导和生长的影响。结果显示,在表达EGFRvIII的细胞中,吉非替尼足以抑制EGFR磷酸化、EGFR介导的增殖和EGFR-介导的锚定非依赖性生长的水平不足以抑制这些特征。此外,数据表明,表达EGFRvIII的细胞长期暴露于低浓度吉非替尼(0.01-0.1微M)会导致受体磷酸酪氨酸负荷增加,ERK信号传导增加,并刺激增殖和锚定非依赖性生长,可能是通过诱导EGFRvⅢ二聚化。另一方面,较高浓度的吉非替尼(1-2微M)显著降低了EGFRvIII磷酸酪氨酸负荷、EGFRvⅢ介导的增殖和锚定非依赖性生长。需要进一步的研究来调查这些重要发现在临床环境中的意义。[2] 表皮生长因子受体(EGFR)在许多人类肿瘤中普遍过表达,为抗癌药物开发提供了新的靶点。ZD1839(“易瑞沙”)是一种对EGFR具有选择性的喹唑啉酪氨酸激酶抑制剂,在临床前研究和临床试验的早期阶段显示出良好的活性。然而,由于尚不清楚哪种肿瘤类型是该药物治疗的最佳靶点,因此已经研究了与ZD1839肿瘤敏感性相关的分子特征。在一组人类乳腺癌症和其他上皮肿瘤细胞系中,HER2-过表达肿瘤对ZD1839特别敏感。这些肿瘤细胞系的生长抑制与EGFR、HER2和HER3的去磷酸化有关,同时HER3与磷脂酰肌醇3-激酶的结合丧失,Akt活性下调。这些研究表明,HER2过表达的肿瘤特别容易受到HER家族酪氨酸激酶信号传导的抑制,并提出了治疗这些特别侵袭性肿瘤的新策略。[3] 1. 吉非替尼盐酸盐(Gefitinib hydrochloride)可强效抑制EGFR酪氨酸激酶活性(IC₅₀=0.033μM),并选择性抑制EGF刺激的肿瘤细胞增殖(IC₅₀=0.054μM);同时可阻断肿瘤细胞中EGF诱导的EGFR自磷酸化,证实其对EGFR信号通路的靶点特异性抑制作用[1] 2. 在表达野生型EGFR的细胞(NR6wtEGFR,低EGFR表达;NR6W,高EGFR表达)中,吉非替尼盐酸盐(Gefitinib hydrochloride)低浓度即可抑制EGF诱导的EGFR磷酸化(涉及Tyr845、Tyr992、Tyr1045、Tyr1068、Tyr1086、Tyr1148、Tyr1173位点)、下游信号分子(p-ERK、p-PLC-γ、p-AKT、p-STAT3)激活及细胞增殖;在表达EGFRvIII的NR6M细胞中,该化合物低浓度(0.01-0.1μM)会增加EGFRvIII磷酸化水平、增强ERK信号、促进细胞增殖及锚定非依赖性生长(可能通过诱导EGFRvIII二聚化),高浓度(1-2μM)则显著降低EGFRvIII磷酸化,抑制其介导的增殖和锚定非依赖性生长[2] 3. 吉非替尼盐酸盐(Gefitinib hydrochloride)可抑制HER2过表达的乳腺癌及上皮肿瘤细胞系生长,这种生长抑制与EGFR、HER2、HER3的去磷酸化、HER3与磷脂酰肌醇3-激酶(PI3K)结合减少及Akt活性下调相关[3] 4. 在LKB1野生型非小细胞肺癌(NSCLC)细胞系中,吉非替尼盐酸盐(Gefitinib hydrochloride)与二甲双胍联用呈现协同抗增殖和促凋亡作用:二甲双胍单独使用会通过增加C-RAF/B-RAF异源二聚化激活丝裂原活化蛋白激酶(MAPK),而吉非替尼盐酸盐(Gefitinib hydrochloride)可抑制EGFR磷酸化及下游信号,阻断二甲双胍诱导的MAPK激活,进而增强凋亡效应[4] 5. 吉非替尼盐酸盐(Gefitinib hydrochloride)可抑制辐射损伤大鼠来源的肺成纤维细胞增殖及EGFR表达,这一作用有助于减轻辐射诱导的肺纤维化;但同时会增加辐射大鼠肺内炎症细胞浸润及促炎细胞因子(IL-6、IL-1β)产生[5] |
| 体内研究 (In Vivo) |
当皮下培养作为肿瘤异种移植物的含有H1299或CALU-3 GEF-R细胞的裸鼠给予二甲双胍和吉非替尼(150 mg/kg,口服)时,肿瘤生长显着减少[3]。虽然吉非替尼治疗由于肺成纤维细胞增殖减少而减弱纤维化肺重塑,但它增加了受辐射大鼠的肺部炎症,包括炎症细胞浸润和促炎细胞因子表达[4]。
在对携带一系列人类肿瘤衍生异种移植物的小鼠的研究中,每天口服一次ZD1839以剂量依赖的方式抑制肿瘤生长。EGFR的表达水平并不能决定异种移植物肿瘤对ZD1839的敏感性。长期ZD1839(>3个月)治疗携带A431异种移植物的小鼠具有良好的耐受性,ZD1839完全抑制了肿瘤生长并诱导了已建立肿瘤的消退。ZD1839治疗期间没有出现耐药性肿瘤,但一些肿瘤在停药后复发。这些研究表明ZD1839在治疗许多人类肿瘤方面的潜在效用,并表明每天口服一次可能是一种合适的治疗方案。[1] 吉非替尼治疗在第15天和第57天增加了受照射大鼠肺部炎性细胞的浸润,这些细胞会产生更多的促炎细胞因子(IL-6、IL-1β),而吉非替宁治疗在第57天降低了受照射的大鼠肺部胶原蛋白含量,并降低了受照大鼠肺部成纤维细胞的增殖和EGFR表达。 结论:在受照射的大鼠中,吉非替尼治疗增强了肺部炎症,包括炎性细胞浸润和促炎细胞因子表达,而吉非替宁治疗由于抑制肺成纤维细胞增殖而减轻了纤维化肺重塑[5]。 1. 在接种多种人源肿瘤异种移植瘤(包括A431细胞)的裸鼠中,每日口服一次吉非替尼盐酸盐(Gefitinib hydrochloride)可呈剂量依赖性抑制肿瘤生长(测试剂量:3.125-200mg/kg);长期治疗(>3个月)耐受性良好,可完全抑制A431异种移植瘤生长并诱导已形成肿瘤消退;治疗期间未出现耐药肿瘤,但部分肿瘤在停药后复发[1] 2. 在NSCLC小鼠异种移植模型中,吉非替尼盐酸盐(Gefitinib hydrochloride)与二甲双胍联用相比单独使用任一药物,对肿瘤生长的抑制作用更强,证实该联用在LKB1野生型NSCLC中的体内协同抗肿瘤效应[4] 3. 在经胸腔单次20Gy X射线照射造成肺损伤的Wistar大鼠中,从照射后第1天至第14天口服给予吉非替尼盐酸盐(Gefitinib hydrochloride)(50mg/kg/天):第15天时,大鼠肺内炎症细胞浸润增加,促炎细胞因子(IL-6、IL-1β)水平升高;第57天时,肺胶原含量减少,肺成纤维细胞增殖受抑,纤维化肺重构得到缓解[5] |
| 酶活实验 |
1. EGFR酪氨酸激酶活性检测:将重组EGFR酪氨酸激酶结构域与系列浓度的吉非替尼盐酸盐(Gefitinib hydrochloride)在ATP及EGFR特异性肽底物存在下共同孵育。在适宜温度下孵育设定时间后,采用合适方法(如免疫印迹或激酶活性检测试剂盒)检测肽底物的磷酸化水平,计算抑制率并确定吉非替尼盐酸盐(Gefitinib hydrochloride)对EGFR激酶活性的IC₅₀值[1]
2. EGFR/EGFRvIII磷酸化检测:将表达野生型EGFR(NR6wtEGFR、NR6W)或突变型EGFRvIII(NR6M)的细胞用不同浓度的吉非替尼盐酸盐(Gefitinib hydrochloride)处理;对需EGF刺激的细胞,药物预处理后加入10nM EGF。制备全细胞裂解液,用磷酸化特异性抗体通过免疫印迹检测EGFR/EGFRvIII特定酪氨酸位点(Tyr845、Tyr992、Tyr1045、Tyr1068、Tyr1086、Tyr1148、Tyr1173)的磷酸化水平;检测总EGFR/EGFRvIII水平作为内参,对条带强度进行定量以评估化合物对受体磷酸化的影响[2] 3. HER家族磷酸化及PI3K结合检测:用吉非替尼盐酸盐(Gefitinib hydrochloride)处理HER2过表达肿瘤细胞,制备细胞裂解液。通过免疫印迹检测EGFR、HER2、HER3的磷酸化水平;采用免疫共沉淀(co-IP)检测HER3与PI3K的结合情况。分析磷酸化水平及蛋白-蛋白相互作用的变化,评估化合物对HER家族信号通路的抑制效果[3] |
| 细胞实验 |
目的:EGF受体(EGFR)酪氨酸激酶抑制剂(TKI)已被发现对肺癌癌症有效,但随着其使用的增加,对这些药物的临床耐药性已经发展。二甲双胍是一种广泛使用的抗糖尿病药物,在几种癌症模型中也表现出显著的生长抑制和促凋亡作用,无论是单独使用还是与化疗药物联合使用<小时>
实验设计:通过MTT、溴化物分析、流式细胞术、凤尾藻非依赖性生长、免疫共沉淀和蛋白质印迹分析,评估吉非替尼、选择性EGFR-TKI和二甲双胍对一组非小细胞肺癌(NSCLC)细胞系的影响<小时>
结果:二甲双胍与吉非替尼的联合用药在携带野生型LKB1基因的NSCLC细胞系中诱导了强烈的抗增殖和促凋亡作用。然而,用二甲双胍作为单一药物治疗,通过增加C-RAF/B-RAF异二聚作用诱导丝裂原活化蛋白激酶(MAPK)的活化和磷酸化。在二甲双胍治疗中加入吉非替尼对EGFR磷酸化和下游信号传导的抑制消除了这一现象,并在体外和体内诱导了强烈的凋亡作用<小时>
结论:二甲双胍和吉非替尼对LKB1野生型NSCLC细胞具有协同作用。然而,还需要进一步的研究来更好地研究二甲双胍作用对RAS/RAF/MAPK途径的影响,以及二甲双胍与分子靶向药物联合使用的最佳情况。[4]
1. 细胞增殖检测(EGFR依赖型细胞):将对EGF敏感的肿瘤细胞接种于96孔板,过夜培养。加入系列稀释的吉非替尼盐酸盐(Gefitinib hydrochloride),在标准培养条件下孵育72小时。采用比色试剂(如MTT或溴化物法)检测细胞活力,从剂量-反应曲线计算抑制细胞增殖的EC₅₀/IC₅₀值[1] 2. 锚定非依赖性生长检测:将NR6(对照)、NR6wtEGFR、NR6W(高EGFR)、NR6M(EGFRvIII)细胞用0-2μM 吉非替尼盐酸盐(Gefitinib hydrochloride)处理后接种于软琼脂中;NR6wtEGFR和NR6W细胞分别用10nM和0.1nM EGF刺激。培养18天后计数克隆,以未处理组克隆数为基准计算克隆数百分比,评估化合物对锚定非依赖性生长的影响[2] 3. 下游信号免疫印迹检测:将细胞(如NSCLC细胞、HER2过表达细胞)用吉非替尼盐酸盐(Gefitinib hydrochloride)处理(联合用药研究中需先预处理二甲双胍)。细胞裂解液经SDS-PAGE分离后转移至膜上,用针对磷酸化和总下游信号分子(ERK、AKT、PLC-γ、STAT3)的抗体进行检测。对条带强度定量,分析化合物对信号通路激活的影响[3,4] 4. 凋亡检测(联合治疗):将LKB1野生型NSCLC细胞分别用吉非替尼盐酸盐(Gefitinib hydrochloride)单独处理、二甲双胍单独处理或二者联合处理。通过流式细胞术(Annexin V/PI染色)或免疫印迹(检测剪切型caspase-3或PARP)检测凋亡;比较各组凋亡细胞百分比或凋亡标志物水平,评估协同促凋亡效应[4] 5. 肺成纤维细胞增殖检测:从辐射损伤大鼠中分离肺成纤维细胞,用吉非替尼盐酸盐(Gefitinib hydrochloride)处理。采用细胞计数试剂盒或BrdU掺入法检测细胞增殖;通过免疫印迹检测成纤维细胞中EGFR表达,评估化合物对成纤维细胞功能的影响[5] |
| 动物实验 |
溶于 0.5% 聚山梨醇酯溶液;100 mg/kg;灌胃给药。
将 LoVo 细胞皮内注射到裸鼠 (cba nu/nu) 体内。背景:吉非替尼是一种表皮生长因子受体 (EGFR) 酪氨酸激酶抑制剂,据报道与间质性肺疾病相关,其高发病率和高死亡率已成为人们高度关注的问题,尤其是在日本。本研究旨在探讨吉非替尼对实验模型中放射性肺疾病不同阶段的影响。方法:Wistar大鼠胸腔于第1天接受单次20 Gy X射线照射,并于第1至14天口服吉非替尼(50 mg/kg/天)。分别于第15天和第57天取大鼠肺脏,进行支气管肺泡灌洗(BAL)。[5] 1. 肿瘤异种移植模型(疗效研究):将人源肿瘤细胞(例如A431细胞、HER2过表达乳腺癌细胞、非小细胞肺癌细胞)皮下植入雌性裸鼠体内。当肿瘤平均体积达到约100-150 mm³时,将小鼠随机分为治疗组和对照组。将盐酸吉非替尼(吉非替尼)溶于合适的溶剂(例如水溶液或合适的有机溶剂混合物)中,每日一次灌胃给药,剂量为3.125-200 mg/kg。每周两次测量肿瘤体积和体重,持续2-4周(或长期研究持续3个月以上);肿瘤生长抑制率(TGI)相对于溶剂对照组计算[1] 2. 放射性肺损伤模型:雄性Wistar大鼠于第1天接受单次20 Gy X射线胸部照射。从第1天到第14天,每日口服给予盐酸吉非替尼(吉非替尼),剂量为50 mg/kg。分别于第15天和第57天处死大鼠;进行支气管肺泡灌洗 (BAL) 以收集炎症细胞,并采集肺组织进行组织病理学分析(炎症细胞浸润、胶原含量)和促炎细胞因子(IL-6、IL-1β)的检测 [5] 3. 联合治疗异种移植模型:将携带 LKB1 野生型 NSCLC 异种移植瘤的裸鼠随机分为四组:载体对照组、吉非替尼盐酸盐(吉非替尼)单药组(灌胃给药,剂量根据既往疗效研究确定)、二甲双胍单药组(口服给药)以及吉非替尼和二甲双胍联合用药组。药物给药持续一段时间;定期测量肿瘤负荷,并绘制肿瘤生长曲线以评估协同效应 [4] |
| 药代性质 (ADME/PK) |
吉非替尼盐酸盐(吉非替尼)在临床前研究中显示出良好的口服生物利用度,支持其每日一次口服给药方案;但相关文献[1]中未提供关于其半衰期、吸收、分布、代谢或排泄(例如清除率、分布容积)的详细数据。
|
| 毒性/毒理 (Toxicokinetics/TK) |
1. 在裸鼠中,长期(>3个月)使用有效抑制肿瘤生长的剂量,盐酸吉非替尼(吉非替尼)耐受性良好;未观察到明显的治疗相关毒性(例如,体重减轻、器官损伤)[1]
2. 在接受胸部照射后,用盐酸吉非替尼(吉非替尼)(50 mg/kg/天,口服,1-14天)治疗的Wistar大鼠中:它加剧了放射性肺炎(第15天肺部炎症细胞浸润和促炎细胞因子IL-6/IL-1β增加),但减轻了放射性肺纤维化(第57天肺胶原含量降低和肺成纤维细胞增殖受到抑制)[5] |
| 参考文献 |
|
| 其他信息 |
表皮生长因子受体 (EGFR) 因其在肿瘤生长、转移和血管生成以及肿瘤对化疗和放疗的耐药性中发挥重要作用,而成为抗癌治疗的一个极具潜力的靶点。我们开发了一种低分子量 EGFR 酪氨酸激酶抑制剂 (EGFR-TKI) ZD1839(Iressa(2))。ZD1839 是一种取代的苯胺基喹唑啉类化合物,是一种强效的 EGFR-TKI (IC50 = 0.033 μM),可选择性抑制 EGF 刺激的肿瘤细胞生长 (IC50 = 0.054 μM),并阻断 EGF 刺激的肿瘤细胞中 EGFR 的自身磷酸化。在携带多种人源肿瘤异种移植瘤的小鼠模型中,每日一次口服 ZD1839 可剂量依赖性地抑制肿瘤生长。 EGFR 的表达水平并不决定异种移植瘤对 ZD1839 的敏感性。对携带 A431 异种移植瘤的小鼠进行长期(>3 个月)ZD1839 治疗耐受性良好,ZD1839 完全抑制了肿瘤生长并诱导了已建立肿瘤的消退。在 ZD1839 治疗期间未出现耐药肿瘤,但部分肿瘤在停药后复发。这些研究表明 ZD1839 在治疗多种人类肿瘤方面具有潜在的应用价值,并提示每日一次口服给药可能是一种合适的治疗方案。[1]
1. 盐酸吉非替尼(吉非替尼) 是一种低分子量、口服有效的苯胺基喹唑啉衍生物,也是首个用于癌症治疗的选择性 EGFR 酪氨酸激酶抑制剂 (EGFR-TKI);它通过抑制 EGFR 介导的信号传导发挥抗肿瘤作用,而 EGFR 介导的信号传导对于肿瘤生长、转移、血管生成以及对化疗/放疗的耐药性至关重要[1] 2. 肿瘤细胞对盐酸吉非替尼(吉非替尼)的敏感性并非完全由 EGFR 表达水平决定; EGFR突变(例如EGFRvIII)和HER2过表达等因素也会影响疗效:HER2过表达的肿瘤尤其敏感,而EGFRvIII表达的细胞则表现出双相反应(低剂量刺激,高剂量抑制)[2,3] 3. 吉非替尼(吉非替尼)与二甲双胍的协同作用特异性地针对LKB1野生型非小细胞肺癌(NSCLC)细胞系,因为二甲双胍单独使用即可激活这些细胞中的MAPK通路,而吉非替尼(吉非替尼)则可阻断这种非靶向激活,从而增强抗肿瘤疗效[4] 4. 吉非替尼(吉非替尼)对放射性肺损伤具有双重作用:它会加剧炎症期(肺炎),但也能减轻……纤维化期,提示与胸部放射治疗联合使用时可能存在风险和益处[5] |
| 分子式 |
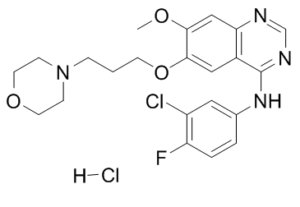
C22H24N4O3FCL.HCL
|
|---|---|
| 分子量 |
483.3633
|
| 精确质量 |
482.128
|
| CAS号 |
184475-55-6
|
| 相关CAS号 |
Gefitinib;184475-35-2
|
| PubChem CID |
19077490
|
| 外观&性状 |
Typically exists as white to off-white solids at room temperature
|
| 沸点 |
607.7ºC at 760 mmHg
|
| 闪点 |
321.3ºC
|
| 蒸汽压 |
4.9E-15mmHg at 25°C
|
| LogP |
5.088
|
| tPSA |
68.74
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
8
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
32
|
| 分子复杂度/Complexity |
545
|
| 定义原子立体中心数目 |
0
|
| SMILES |
ClC1=C(C=CC(NC2=NC=NC3=C2C=C(C(OC)=C3)OCCCN4CCOCC4)=C1)F.[H]Cl
|
| InChi Key |
QUINXWLATMJDQF-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C22H24ClFN4O3.ClH/c1-29-20-13-19-16(12-21(20)31-8-2-5-28-6-9-30-10-7-28)22(26-14-25-19)27-15-3-4-18(24)17(23)11-15;/h3-4,11-14H,2,5-10H2,1H3,(H,25,26,27);1H
|
| 化学名 |
N-(3-chloro-4-fluorophenyl)-7-methoxy-6-(3-morpholin-4-ylpropoxy)quinazolin-4-amine;hydrochloride
|
| 别名 |
ZD1839 HCl; ZD 1839 HCl; ZD-1839 HCl; Gefitinib HCl; trade name: Iressa.Gefitinib hydrochloride; 184475-55-6; gefitinib hcl; Gefitinib (hydrochloride); ZD-1839 hydrochloride; Gefitinib hydrochloride salt; N-(3-chloro-4-fluorophenyl)-7-methoxy-6-(3-morpholin-4-ylpropoxy)quinazolin-4-amine;hydrochloride; 4-Quinazolinamine,N-(3-chloro-4-fluorophenyl)-7-methoxy-6-[3-(4-morpholinyl)propoxy]-,monohydrochloride;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0689 mL | 10.3443 mL | 20.6885 mL | |
| 5 mM | 0.4138 mL | 2.0689 mL | 4.1377 mL | |
| 10 mM | 0.2069 mL | 1.0344 mL | 2.0689 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03292133 | Active Recruiting |
Drug: EGF816 Drug: Gefitinib |
Lung Cancer | Massachusetts General Hospital | October 31, 2017 | Phase 2 |
| NCT03122717 | Active Recruiting |
Drug: Gefitinib Drug: Osimertinib |
Non-Small Cell Lung Cancer | Dana-Farber Cancer Institute | May 9, 2017 | Phase 1 Phase 2 |
| NCT03758287 | Active Recruiting |
Drug: Gefitinib Drug: CT053PTSA |
Non-small Cell Lung Cancer | Sunshine Lake Pharma Co., Ltd. | November 2016 | Phase 1 Phase 2 |
| NCT03849768 | Active Recruiting |
Drug: Gefitinib Drug: HS-10296 |
Non Small Cell Lung Cancer | Jiangsu Hansoh Pharmaceutical Co., Ltd. |
February 1, 2019 | Phase 3 |
| NCT02856893 | Active Recruiting |
Drug: Gefitinib Drug: Osimertinib |
NSCLC | European Organisation for Research and Treatment of Cancer - EORTC |
October 10, 2017 | Phase 2 |
A, effect of metformin (MET) alone and in combination with gefitinib (GEF) on cell proliferation, on anchorage-independent growth ability of NSCLC cell lines, and on the induction of apoptosis in CALU-3, CALU-3 GEF-R, and H1299 cell lines.Clin Cancer Res.2013 Jul 1;19(13):3508-19. |
|---|
Effects on the downstream pathway by combined treatment of metformin and gefitinib. Western blotting of EGFR, MAPK, AKT p70S6K, and S6 activation following treatment with the indicated concentration of metformin and gefitinib in CALU-3 and CALU-3 GEF-R cell lines. β-Actin was included as a loading control.Clin Cancer Res.2013 Jul 1;19(13):3508-19. |
Effects of the combination treatment of metformin and gefitinib on NSCLC tumor xenografts.Clin Cancer Res.2013 Jul 1;19(13):3508-19. |