| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| Other Sizes |
|
| 靶点 |
RSV RdRp (IC50 = 1.1 µM); HCV RdRp (IC50 = 5 µM)
Viral RNA-dependent RNA polymerase (RdRp) of Ebola virus (EBOV) and related viruses. - EBOV RdRp inhibition: IC50 = 0.28 μM (enzymatic assay with purified EBOV polymerase complex) [1] |
|---|---|
| 体外研究 (In Vitro) |
- 抗EBOV活性:三磷酸代谢物GS-443902(源自GS-5734)在Huh-7细胞中抑制EBOV复制,EC50 = 0.086 μM(处理48小时后通过qRT-PCR定量病毒RNA)[1]
- 作用机制:GS-443902作为ATP类似物,与天然ATP竞争结合RdRp,导致病毒RNA合成延迟终止 [1] 在巨噬细胞、HMVEC 和 HeLa 细胞系中测量 GS-443902 三钠(GS-441524 三磷酸三钠;瑞德西韦代谢物三钠)的水平,Cmax 值分别为 300、110 和 90 pmol/百万细胞 [1] 。化合物 8a,GS-443902 Trisodium,是三磷酸 (TP) 的衍生物 [2]。 GS-443902 三钠(NTP;0.01、0.1、1、10、100 μM)与新生病毒 RNA 转录本结合,迫使它们过早终止并抑制 RSV RdRp 催化的 RNA 合成。在细胞内成功转化为 GS-443902 钠后,GS-5734 通过靶向其 RdRp 并减少病毒 RNA 合成来特异性抑制 EBOV 复制 [3]。 |
| 体内研究 (In Vivo) |
瑞德西韦(GS-5734;10 mg kg;IV)快速进入外周血单核细胞(PBMC),在恒河猴中,在给药 2 小时内显着且有效地转化为 GS-443902 三钠(瑞德西韦代谢三钠;NTP)。 GS-443902 三钠的半衰期为 14 小时,是 PBMC 中的主要代谢物,24 小时后达到抑制 50% 以上病毒所需的水平 [3]。
|
| 酶活实验 |
- EBOV RdRp抑制实验:纯化的EBOV RdRp复合物(L聚合酶-VP35辅助因子)与合成RNA模板引物、[α-32P]ATP、UTP、CTP、GTP及梯度浓度的GS-443902在30°C孵育60分钟。EDTA终止反应后,变性聚丙烯酰胺凝胶电泳(PAGE)分离RNA产物,磷屏成像定量并计算IC50 [1]
|
| 细胞实验 |
- EBOV抗病毒实验:Huh-7细胞感染EBOV(Mayinga株,MOI = 0.01)后,用前药GS-5734(在细胞内生成GS-443902)梯度浓度处理48小时。qRT-PCR定量上清液病毒RNA,ATP发光法检测细胞活性(GS-5734的CC50 > 10 μM)[1]
|
| 动物实验 |
西非最近爆发的埃博拉病毒疫情,其病例数、死亡人数、地理分布范围以及受影响国家数量均前所未有,凸显了开发安全、有效且易于获取的抗病毒药物以治疗和预防急性埃博拉病毒病(EVD)及其后遗症的迫切需求。目前尚无任何抗病毒疗法获得监管部门批准或证实具有临床疗效。本文报道了一种新型小分子GS-5734的发现,它是一种腺苷类似物的单磷酰胺前药,具有抗埃博拉病毒活性。细胞实验表明,GS-5734对多种埃博拉病毒变种和其他丝状病毒均具有抗病毒活性。在体外用GS-5734孵育的多种人类细胞类型中,药理活性核苷三磷酸(NTP)能够高效生成。在以呼吸道合胞病毒RNA聚合酶为替代物的引物延伸实验中,NTP可作为替代底物和RNA链终止剂。向非人灵长类动物静脉注射GS-5734后,外周血单核细胞中NTP水平持续升高(半衰期为14小时),并分布至包括睾丸、眼睛和大脑在内的病毒复制庇护所。在恒河猴埃博拉病毒病(EVD)模型中,每日一次静脉注射10 mg/kg的GS-5734,持续12天,可显著抑制埃博拉病毒(EBOV)的复制,并保护100%的EBOV感染动物免于致命性疾病,改善临床疾病症状和病理生理指标。即使在病毒暴露三天后才开始治疗,且在六只接受治疗的动物中有两只检测到全身性病毒RNA的情况下,GS-5734仍然有效。这些结果表明,小分子抗病毒化合物首次在非人灵长类动物中对EBOV具有实质性的暴露后保护作用。GS-5734在体外对其他致病性RNA病毒(包括丝状病毒、沙粒病毒和冠状病毒)具有广谱抗病毒活性,提示其具有更广泛的医学应用潜力。GS-5734易于大规模生产,目前正在进行药物安全性和药代动力学的临床研究。[2]
|
| 药代性质 (ADME/PK) |
代谢途径:GS-443902是前药GS-5734的细胞内磷酸化活性代谢物。它由GS-5734依次水解为丙氨酸代谢物(GS-704277)和核苷类似物(GS-441524),然后在靶细胞内经三重磷酸化形成[1]
- 细胞内持久性:在原代人巨噬细胞中,GS-443902的细胞内半衰期>20小时,表明前药给药后具有持久的抗病毒活性[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
体外细胞毒性:在浓度高达 10 μM 时,母体前药 GS-5734(生成细胞内 GS-443902)在 Huh-7 细胞中未观察到明显的细胞毒性(CC50 > 10 μM)[1]
- 线粒体毒性:浓度高达 100 μM 时,GS-443902 未抑制人线粒体 RNA 聚合酶 (POLRMT),表明其具有选择性抗病毒靶向作用,而非宿主聚合酶 [1] |
| 参考文献 |
|
| 其他信息 |
GS-443902 是核苷类似物 GS-441524 的药理活性三磷酸形式,GS-441524 是前药瑞德西韦 (GS-5734) 发挥抗病毒作用的关键成分 [1]。由于其保守的 RdRp 结构基序,GS-443902 对包括丝状病毒(埃博拉病毒)、冠状病毒(中东呼吸综合征冠状病毒、SARS-CoV-2)和副黏病毒在内的多种 RNA 病毒具有广谱活性 [1]。近期西非爆发的埃博拉病毒 (EBOV) 疫情是历史上规模最大的一次,病例超过 28,000 例,导致超过 11,000 人死亡,其中包括 500 多名医护人员。一项重点筛选和先导化合物优化工作确定了化合物 4b (GS-5734),其在巨噬细胞中对埃博拉病毒的 EC50 值为 86 nM,作为临床候选药物。结构活性关系研究表明,1'-CN基团和C连接的核碱基对于发挥最佳抗埃博拉病毒(EBOV)效力和选择性抑制宿主聚合酶至关重要。一种稳健的非对映选择性合成方法提供了足够量的化合物4b,使其能够在非人灵长类动物EBOV感染模型中进行临床前疗效评估。感染后第3-14天,每日一次静脉注射10 mg/kg的4b,可显著降低病毒血症和死亡率,使感染动物的存活率达到100% [Nature 2016, 531, 381-385]。一项II期临床试验(PREVAIL IV)目前正在招募受试者,并将评估化合物4b对EBOV幸存者在隔离场所病毒脱落的影响。[1]
|
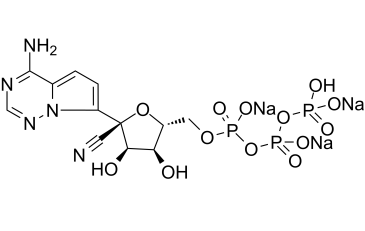
| 分子式 |
C12H12N5NA4O13P3
|
|---|---|
| 分子量 |
619.129748344421
|
| 精确质量 |
618.923
|
| CAS号 |
1355050-21-3
|
| 相关CAS号 |
GS-443902;1355149-45-9
|
| PubChem CID |
170907243
|
| 外观&性状 |
Typically exists as white to light yellow solids at room temperature
|
| tPSA |
301Ų
|
| 氢键供体(HBD)数目 |
7
|
| 氢键受体(HBA)数目 |
17
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
34
|
| 分子复杂度/Complexity |
941
|
| 定义原子立体中心数目 |
4
|
| SMILES |
C1=C2C(=NC=NN2C(=C1)[C@]3([C@@H]([C@@H]([C@H](O3)COP(=O)(O)OP(=O)(O)OP(=O)(O)O)O)O)C#N)N.[Na]
|
| InChi Key |
GONGCFUOBMNVIT-ZTYDICHKSA-J
|
| InChi Code |
InChI=1S/C12H16N5O13P3.4Na/c13-4-12(8-2-1-6-11(14)15-5-16-17(6)8)10(19)9(18)7(28-12)3-27-32(23,24)30-33(25,26)29-31(20,21)22;;;;/h1-2,5,7,9-10,18-19H,3H2,(H,23,24)(H,25,26)(H2,14,15,16)(H2,20,21,22);;;;/q;4*+1/p-4/t7-,9-,10-,12+;;;;/m1..../s1
|
| 化学名 |
tetrasodium;[[[(2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxyoxolan-2-yl]methoxy-oxidophosphoryl]oxy-oxidophosphoryl] phosphate
|
| 别名 |
GS-441524 sodium; 1355050-21-3; tetrasodium;[[[(2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxyoxolan-2-yl]methoxy-oxidophosphoryl]oxy-oxidophosphoryl] phosphate; GS-443902 Sodium; BCP29948; A937202; GS-441524; GS 441524; GS441524; GS 5734; GS-5734; GS5734
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: (1). 本产品在运输和储存过程中需避光。 (2). 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮。 (3). 该产品在溶液状态不稳定,请现配现用。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ~33.33 mg/mL (~55.82 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 50 mg/mL (83.73 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6152 mL | 8.0758 mL | 16.1517 mL | |
| 5 mM | 0.3230 mL | 1.6152 mL | 3.2303 mL | |
| 10 mM | 0.1615 mL | 0.8076 mL | 1.6152 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。