| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 靶点 |
Farnesoid X receptor (FXR) agonist (EC50 = 193 nM in cell-based cotransfection assay; relative EC50 = 121 nM in co-activator recruitment assay) [1]
|
|---|---|
| 体外研究 (In Vitro) |
在基于细胞的共转染测试中,LY2562175 (EC50 = 193 nM) 刺激人 FXR 的转录激活。与 GW4064 相比,LY2562175 在促进从共激活剂 SRC-1 的核受体相互作用结构域募集肽方面表现出 93.5% 的功效,相对 EC50 为 121 nM [1]。
在使用转染了人FXR表达质粒和荧光素酶报告质粒的HEK293细胞进行的基于细胞的共转染实验中,LY2562175以几何平均EC50为193 nM的效力促进FXR转录激活。它作为部分激动剂,相对于完全激动剂GW4064表现出约41.3%的效能。[1] 在使用纯化的重组FXR配体结合域蛋白和共激活因子SRC-1的核受体相互作用结构域,并通过AlphaScreen技术测量的生化实验中,LY2562175以相对EC50为121 nM和相对于GW4064 93.5%的效能促进共激活因子招募。[1] LY2562175对FXR表现出选择性,因为在浓度高达10,000 nM时,它不会激活其他核受体,包括糖皮质激素受体(GR)、雄激素受体(AR)、盐皮质激素受体(MR)和孕激素受体(PR)。[1] |
| 体内研究 (In Vivo) |
当用 LY2562175 治疗时,血清甘油三酯和胆固醇以剂量依赖性方式降低。与用赋形剂治疗的动物相比,LY2562175 在 10 mg/kg 剂量下可将血清甘油三酯降低 76%,将胆固醇降低 80%。结果发现,血清甘油三酯的ED50为3.4mg/kg,血清胆固醇的ED50为2mg/kg。在禁食和非禁食状态下,雌性 ZDF 大鼠的 LY2562175 治疗导致血浆甘油三酯呈剂量依赖性降低。当LY2562175和BRL49653以固定剂量一起服用时,空腹和非空腹血浆甘油三酯的降低甚至更大。在该动物模型中,FPLC 脂蛋白分级分离显示 LY2562175 治疗导致 HDL-c 显着增加,vLDL-C 降低 [1]。
在喂食西方饮食(高脂/高胆固醇)的8周龄雄性低密度脂蛋白受体敲除(LDLR -/-)小鼠中,每日一次口服LY2562175,连续7天,可导致血清胆固醇和甘油三酯呈剂量依赖性下降。在10 mg/kg剂量下,与溶媒对照组相比,胆固醇降低80%,甘油三酯降低76%。降低血清胆固醇的ED50为2.0 mg/kg,降低甘油三酯的ED50为3.4 mg/kg。脂蛋白亚型分析显示VLDL和LDL胆固醇显著降低。[1] 在雌性Zucker糖尿病肥胖(ZDF)大鼠中,用LY2562175处理9天导致空腹和非空腹状态下的血浆甘油三酯呈剂量依赖性降低。FPLC分析显示VLDL-胆固醇降低和HDL-胆固醇大幅升高。在30 mg/kg剂量下,HDL水平相对于溶媒对照组几乎翻倍。与固定低剂量的罗格列酮(0.3 mg/kg)联用可进一步降低甘油三酯。[1] |
| 酶活实验 |
FXR共激活因子招募实验(AlphaScreen): 该实验测量了LY2562175诱导FXR配体结合域(LBD)构象变化并招募转录共激活因子的能力。将纯化的重组人FXR LBD蛋白与共激活因子蛋白SRC-1的核受体相互作用结构域共同孵育。使用AlphaScreen技术定量蛋白质之间的相互作用,该相互作用指示激动剂诱导的招募。相对于完全激动剂GW4064,确定了LY2562175的效力(相对EC50)和效能。[1]
|
| 细胞实验 |
FXR转录激活实验(基于细胞的共转染): 将HEK293细胞与表达人FXR的质粒以及含有两个拷贝的FXR反应元件(IR-1)驱动荧光素酶表达的报告质粒共转染。转染后,用LY2562175处理细胞。通过测量荧光素酶活性来确定FXR激动剂活性,该活性反映了FXR介导的转录激活水平。从剂量反应曲线计算EC50和相对效能(与GW4064相比)。[1]
核受体选择性筛选: 使用一组核受体共转录激活实验评估LY2562175的选择性。在基于细胞的系统中测试该化合物激活糖皮质激素受体(GR)、雄激素受体(AR)、盐皮质激素受体(MR)和孕激素受体(PR)的能力。通过适当的报告基因实验测量激活情况,在高浓度下缺乏活性表明对FXR的选择性。[1] |
| 动物实验 |
LDLR -/- 小鼠疗效研究:将 8 周龄雄性 LDL 受体敲除小鼠饲喂高脂/高胆固醇饮食(TD88137,含 0.15% 胆固醇和 42% 脂肪)两周。然后将小鼠随机分为 6 组(n=6),连续 7 天,每天灌胃一次,分别给予赋形剂(5% Solutol、5% 乙醇、1% 羧甲基纤维素)或 LY2562175(钠盐制剂),剂量分别为 1、3、10 和 30 mg/kg,灌胃体积为 5 mL/kg。第 7 天,在麻醉状态下通过心脏穿刺采集血液,用于血清脂质分析。 [1]
ZDF 大鼠疗效研究:雌性 Zucker 糖尿病肥胖 (ZDF) 大鼠接受不同剂量的 LY2562175、罗格列酮 (0.3 mg/kg) 或二者联合治疗,疗程 9 天。所提供的文本中未详细说明具体的赋形剂和给药方案(频率、途径)。采集血液样本,采用 FPLC 分析血浆甘油三酯和脂蛋白谱。[1] 临床前药代动力学研究:在 Sprague Dawley 大鼠、比格犬和食蟹猴中进行了药代动力学研究。静脉注射 (IV) 时,将 LY2562175 配制成 5% Solutol/5% 乙醇的生理盐水溶液,大鼠和犬的给药剂量为 1 mg/kg,猴的给药剂量为 0.91 mg/kg。口服给药时,将药物配制成含1.0%羧甲基纤维素、0.25%聚山梨醇酯80和0.05%消泡剂的纯净水溶液,剂量为3 mg/kg。在不同时间点采集血样,分离血浆,并采用液相色谱-串联质谱法(LC-MS/MS)进行分析。[1] |
| 药代性质 (ADME/PK) |
临床前药代动力学:在临床前动物模型中,静脉注射LY2562175后,其平均末端消除半衰期(T1/2)约为2至10小时。清除率较低至中等(低于各物种的肝血流量)。与啮齿类动物相比,非啮齿类动物的口服暴露量更高。口服给药后,各物种的达峰时间(Tmax)为1至3小时。大鼠、犬和猴的口服生物利用度分别为21±5%、82%和24±3%。[1]
血浆蛋白结合率:LY2562175在大鼠血浆中的游离分数(fu)为0.020,在人血浆中的游离分数为0.032。 [1] 人体药代动力学(单次递增剂量):在一项针对健康受试者的单次剂量递增研究(剂量范围为 5 至 600 mg)中,LY2562175 耐受性良好。给药后平均 2 至 3 小时达到血浆峰浓度。暴露量(AUC 和 Cmax)的增加幅度小于剂量比例。在 75 至 600 mg 的剂量范围内,平均消除半衰期为 16.9 至 24.2 小时。啮齿动物脂质调节 ED50 剂量下的 Cmax 为 4.62 ng/mL。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
在人体单次递增剂量研究中,LY2562175在5至600 mg的剂量范围内耐受性良好。[1]
血浆中LY2562175的游离分数(蛋白结合数据)见ADME部分。[1] |
| 参考文献 | |
| 其他信息 |
TERN-101 正在进行临床试验 NCT04328077(LIFT 研究:TERN-101 在非肝硬化性非酒精性脂肪性肝炎 (NASH) 患者中的安全性、耐受性、有效性和药代动力学研究)。
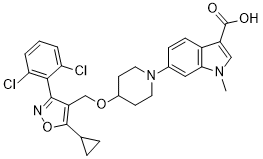
LY2562175(化学名称:6-(4-{[5-环丙基-3-(2,6-二氯苯基)异噁唑-4-基]甲氧基}哌啶-1-基)-1-甲基-1H-吲哚-3-羧酸) 是一种新型、强效且选择性的 FXR 激动剂,最初用于治疗血脂异常和动脉粥样硬化。[1] 在细胞试验中,它被描述为 FXR 的部分激动剂。 [1] 该化合物在血脂异常的临床前动物模型中能显著降低低密度脂蛋白胆固醇和极低密度脂蛋白胆固醇,同时升高高密度脂蛋白胆固醇。[1] |
| 分子式 |
C28H27CL2N3O4
|
|---|---|
| 分子量 |
540.437685251236
|
| 精确质量 |
539.137
|
| CAS号 |
1103500-20-4
|
| 相关CAS号 |
1103500-20-4 (free acid);LY2502175 sodium;
|
| PubChem CID |
25204767
|
| 外观&性状 |
White to light yellow solid powder
|
| LogP |
5.5
|
| tPSA |
80.7
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
6
|
| 可旋转键数目(RBC) |
7
|
| 重原子数目 |
37
|
| 分子复杂度/Complexity |
800
|
| 定义原子立体中心数目 |
0
|
| SMILES |
ClC1C=CC=C(C=1C1C(COC2CCN(C3=CC=C4C(C(=O)O)=CN(C)C4=C3)CC2)=C(C2CC2)ON=1)Cl
|
| InChi Key |
RPVDFHPBGBMWID-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C28H27Cl2N3O4/c1-32-14-20(28(34)35)19-8-7-17(13-24(19)32)33-11-9-18(10-12-33)36-15-21-26(31-37-27(21)16-5-6-16)25-22(29)3-2-4-23(25)30/h2-4,7-8,13-14,16,18H,5-6,9-12,15H2,1H3,(H,34,35)
|
| 化学名 |
6-(4-((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl)methoxy)piperidin-1-yl)-1-methyl-1H-indole-3-carboxylic acid
|
| 别名 |
LY 2562175; LY2562175; LY-2562175.
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~62.5 mg/mL (~115.65 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (3.85 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (3.85 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8503 mL | 9.2517 mL | 18.5034 mL | |
| 5 mM | 0.3701 mL | 1.8503 mL | 3.7007 mL | |
| 10 mM | 0.1850 mL | 0.9252 mL | 1.8503 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。