| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
- Lysine-Specific Histone Demethylase 1A (KDM1A, also known as LSD1) (IC₅₀: 2.6 nM for recombinant human KDM1A; Ki: 1.8 nM for human KDM1A; no activity against KDM1B (LSD2) at concentrations up to 10 μM, showing >3800-fold selectivity for KDM1A over KDM1B) [3]
|
|---|---|
| 体外研究 (In Vitro) |
Iadademstat diHClide 是一种 KDM1A 抑制剂,可与黄素腺嘌呤核苷酸 (FAD) 辅因子不可逆地结合,使 KDM1A 失活。在混合谱系白血病 (MLL) 易位急性髓系白血病 (AML) 细胞系分化和集落形成的检测中,iadademstat 对 KDM1A 表现出强于 MAO 酶的选择性,对 KDM1B 表现出强选择性。无与伦比的亚纳摩尔生物活性。在 MLL-AF9 细胞中,iadademstat 以时间和剂量依赖性方式诱导 Cd11b 分化标记物。有趣的是,这发生在 H3K4me2 水平改变之前。 Iadademstat 还影响其他急性白血病 (AL) 细胞系,而 MLL 易位细胞是最脆弱的 [1]。
1. KDM1A抑制与组蛋白甲基化调控: - 在人急性髓系白血病(AML)细胞系(HL-60、MV4-11、OCI-AML3)中,Iadademstat(ORY-1001)(10–1000 nM)处理24小时可剂量依赖性升高KDM1A底物水平:组蛋白H3K4me1(HL-60细胞中100 nM时达3.5±0.4倍)和H3K4me2(HL-60细胞中100 nM时达2.8±0.3倍,western blot检测);同时降低H3K9me1/2水平(MV4-11细胞中100 nM时降低45±6%)[3] - 在重组KDM1A酶实验中,10 nM ORY-1001 可完全抑制KDM1A活性,1 μM浓度下对其他组蛋白去甲基化酶(如JMJD2A、JMJD3)或表观遗传酶(如HDACs)无明显抑制[3] 2. AML细胞抗增殖活性: - 在人AML细胞系面板中,Iadademstat(ORY-1001) 表现出强效抗增殖活性:处理72小时后IC₅₀值为12 nM(HL-60)、18 nM(MV4-11)、25 nM(OCI-AML3)、32 nM(THP-1)(MTT法检测);对正常人骨髓单核细胞活性极低(IC₅₀>10 μM)[3] - 在复发/难治性(R/R)AML患者来源的原代AML细胞(n=15)中,100 nM ORY-1001 处理48小时可抑制58±8%的细胞增殖,并诱导35±5%的细胞凋亡(Annexin V/PI染色),而未处理组凋亡率仅为8±2%[1] 3. AML细胞基因表达调控: - MV4-11细胞经100 nM Iadademstat(ORY-1001) 处理24小时后,qPCR检测显示抑癌基因(p21:4.2±0.5倍,p53:2.1±0.3倍)上调,癌基因(MYC:0.4±0.1倍,BCL-2:0.3±0.1倍)下调[3] |
| 体内研究 (In Vivo) |
1. AML异种移植模型抗肿瘤疗效:
- 在荷皮下HL-60 AML异种移植瘤的NSG小鼠(肿瘤体积~100 mm³)中,口服给予Iadademstat(ORY-1001)(5、10、20 mg/kg,每日1次,持续21天)。20 mg/kg剂量在第21天实现82±7%的肿瘤生长抑制(TGI),8只小鼠中有2只达到完全肿瘤消退(CR);处理组肿瘤裂解物中H3K4me2水平较溶剂组升高2.3±0.3倍[3]
- 在荷原位MV4-11 AML异种移植瘤的NSG小鼠(经尾静脉注射1×10⁶细胞建立模型)中,口服ORY-1001(15 mg/kg/天,持续28天)可将中位生存期从溶剂组的21天延长至38天,生存期获益达81%[3] Iadademstat 可延长播散性 T-ALL 模型的生存期,并减少小鼠和大鼠异种移植物中 AML 肿瘤的发展 [1]。 |
| 酶活实验 |
1. 重组人KDM1A活性实验:
- 将重组人KDM1A(181–836位氨基酸,与CoREST复合)与荧光肽底物(H3K4me2肽,序列ARTKQTARK(me2)STGGKAPRKQL)在assay缓冲液(50 mM Tris-HCl pH 8.0、100 mM NaCl、5 mM DTT、0.1 mg/mL BSA)中于37°C孵育。加入Iadademstat(ORY-1001)(0.1–1000 nM),通过添加2-酮戊二酸(终浓度100 μM)和Fe²⁺(终浓度10 μM)启动反应。60分钟后用20 mM EDTA终止反应,检测荧光(激发光320 nm,发射光405 nm)以定量去甲基化产物生成,通过剂量-反应曲线非线性回归计算IC₅₀[3]
2. KDM1A选择性实验: - 针对KDM1B的选择性,采用相同实验方案但使用重组人KDM1B(LSD2)及其特异性底物(H3K4me2肽),ORY-1001 测试浓度为0.1 nM–10 μM,10 μM以下无KDM1B抑制活性。针对其他表观遗传酶(JMJD2A、JMJD3、HDAC1–3),采用商品化酶活性试剂盒检测,1 μM浓度下无抑制活性[3] |
| 细胞实验 |
1. 抗增殖MTT实验:
- 人AML细胞系(HL-60、MV4-11、OCI-AML3)以5×10³细胞/孔接种于96孔板,用含10%胎牛血清的RPMI 1640培养基培养。加入Iadademstat(ORY-1001)(1 nM–10 μM),孵育72小时后每孔加入10 μL MTT试剂(5 mg/mL),继续孵育4小时;用100 μL DMSO终止反应,检测570 nm处吸光度,通过GraphPad Prism软件计算IC₅₀[3]
2. 组蛋白甲基化western blot实验: - HL-60细胞经ORY-1001(10–1000 nM)处理24小时后,用含蛋白酶和磷酸酶抑制剂的RIPA缓冲液裂解,通过核提取试剂盒制备核提取物;取20 μg核蛋白进行12% SDS-PAGE,转移至PVDF膜。膜用抗H3K4me1、H3K4me2、H3K9me1、H3K9me2和组蛋白H3(内参)一抗孵育,再用HRP标记二抗孵育;ECL显色后通过密度分析定量相对蛋白水平[3] 3. 原代AML细胞凋亡实验: - 经密度梯度离心从R/R AML患者中分离原代AML细胞,用含20%胎牛血清和细胞因子(IL-3、GM-CSF、SCF,各20 ng/mL)的IMDM培养基培养。细胞经100 nM Iadademstat(ORY-1001) 处理48小时后,用Annexin V-FITC和碘化丙啶(PI)室温染色15分钟,流式细胞术分析凋亡(Annexin V⁺/PI⁻为早期凋亡,Annexin V⁺/PI⁺为晚期凋亡)[1] |
| 动物实验 |
1. 皮下HL-60 AML异种移植模型:- 动物:雌性NSG小鼠(6-8周龄,每组n=8)。- 肿瘤诱导:将5×10⁶个HL-60细胞(重悬于1:1 PBS:Matrigel混合液中)皮下注射至小鼠右侧腹部。- 给药方案:当肿瘤体积达到约100 mm³时,将小鼠随机分为4组:溶剂组(0.5%甲基纤维素+0.2% Tween 80水溶液)和Iadademstat (ORY-1001)组,剂量分别为5、10和20 mg/kg。药物每日口服一次,连续给药21天。- 评价指标:每周两次使用游标卡尺测量肿瘤体积(V = 0.5 × 长 × 宽²);每周记录小鼠体重。研究结束时,收集肿瘤组织,裂解,并使用蛋白质印迹法检测H3K4me2水平[3]
2. 原位MV4-11 AML异种移植模型: - 动物:雌性NSG小鼠(6-8周龄,每组n=10)。 - 肿瘤诱导:经尾静脉注射1×10⁶个MV4-11细胞(荧光素酶标记)。注射后第7天通过生物发光成像(BLI)确认肿瘤移植。 - 给药方案:小鼠从注射后第7天开始,连续28天口服ORY-1001(15 mg/kg/天)或载体。 - 评估指标:每周进行BLI以监测肿瘤负荷;每日记录生存情况,直至所有载体组小鼠死亡。采用Kaplan-Meier法计算中位生存期和生存获益[3] |
| 药代性质 (ADME/PK) |
1. 人体药代动力学(I期研究):- 在接受口服Iadademstat (ORY-1001)治疗的复发/难治性急性髓系白血病(R/R AML)患者(n=55)中,每日一次,剂量为20-600 mg,药代动力学参数显示:- 达到最大血浆浓度的时间(Tmax):所有剂量均为1.5-2.5小时。- 最大血浆浓度(Cmax):28.3±5.2 ng/mL(20 mg),105.6±12.8 ng/mL(100 mg),320.4±35.7 ng/mL(400 mg)。 - 血浆浓度-时间曲线下面积(AUC₀-24h):85.6±10.3 ng·h/mL(20 mg),380.2±42.5 ng·h/mL(100 mg),1120.5±120.8 ng·h/mL(400 mg)(剂量至 400 mg 时呈剂量比例关系)。- 末端半衰期(t₁/₂):4.2±0.5 小时(各剂量组间一致)。 - 口服生物利用度:约35%(通过比较临床前研究中口服和静脉注射的AUC估算)[1]
2. 小鼠药代动力学:- 在雌性NSG小鼠中,口服ORY-1001(20 mg/kg)的结果为:Cmax = 450±50 ng/mL,Tmax = 1小时,AUC₀-24h = 1800±200 ng·h/mL,t₁/₂ = 3.8±0.4小时。静脉注射(5 mg/kg)的结果为:Cmax = 1200±150 ng/mL,AUC₀-24h = 1200±100 ng·h/mL,t₁/₂ = 2.1±0.3小时[3] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. 人体临床毒性(I期研究):- 在接受Iadademstat (ORY-1001)(20–600 mg/天)治疗的复发/难治性急性髓系白血病 (R/R AML) 患者中,治疗相关不良事件 (TRAE) 主要为 1–2 级:- 常见 TRAE(发生率 >20%):恶心 (42%)、疲乏 (38%)、腹泻 (31%)、呕吐 (28%) 和食欲下降 (22%)。- 3–4 级 TRAE(发生率 <10%):中性粒细胞减少症 (8%)、血小板减少症 (6%) 和丙氨酸氨基转移酶 (ALT) 升高 (5%)。 - 剂量限制性毒性 (DLT):在 600 mg/天剂量下观察到,6 例患者中有 2 例出现 4 级中性粒细胞减少症(持续时间 >7 天)[1]
- 血浆蛋白结合率:在人血浆中,ORY-1001 显示出高蛋白结合率 (>98%),通过平衡透析法测定[1] 2. 小鼠毒性:- 在一项为期 28 天的雌性 NSG 小鼠重复给药毒性研究中(口服剂量分别为 5、15 和 45 mg/kg/天),未观察到死亡。在 45 mg/kg/天剂量下,观察到轻度体重减轻 (<10%) 和血清 AST 短暂升高(高于正常值 1.5 倍),主要器官(肝脏、肾脏、骨髓)未见组织病理学改变[3] |
| 参考文献 | |
| 其他信息 |
1. 作用机制:- Iadademstat (ORY-1001) 是一种首创的共价 KDM1A 抑制剂,可与 KDM1A 的 FAD 辅因子不可逆地结合,从而阻断其去甲基酶活性。这导致 H3K4me1/2(激活组蛋白标记)的积累和 H3K9me1/2(抑制标记)的减少,从而改变肿瘤抑制基因(例如 p21)和癌基因(例如 MYC)的表达,进而抑制 AML 细胞增殖并诱导细胞凋亡 [3]
2. R/R AML 的临床疗效(I 期研究):- 在 55 例接受 Iadademstat (ORY-1001) 治疗的 R/R AML 患者中,总缓解率 (ORR) 为 22% (12/55),其中包括 5 例完全缓解 (CR, 9%) 和 7 例伴有不完全血液学恢复的完全缓解 (CRi, 13%)。中位缓解持续时间 (DOR) 为 5.8 个月(范围:2.1–12.3 个月)[1] 3. 治疗靶点原理:- KDM1A 在 AML 中过度表达,尤其是在 MLL 重排或 NPM1 突变的亚群中,它通过抑制分化和凋亡基因来促进白血病发生。ORY-1001 靶向这种依赖性,使其成为复发/难治性 AML 的一种有前景的药物[2][3] |
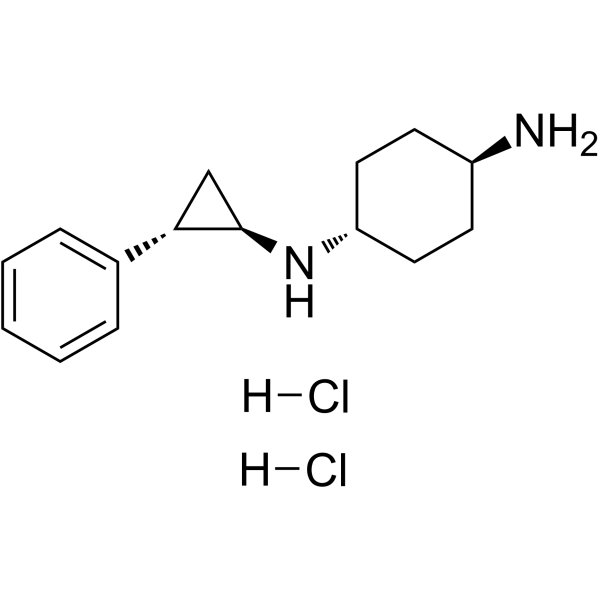
| 分子式 |
C15H23CLN2
|
|---|---|
| 分子量 |
266.809522867203
|
| 精确质量 |
302.131
|
| 元素分析 |
C, 59.41; H, 7.98; Cl, 23.38; N, 9.24
|
| CAS号 |
1431303-72-8
|
| 相关CAS号 |
1431303-72-8 (2HCl);1431304-21-0;1431303-71-7 (xHCl);1431326-61-2 (2HCl);
|
| PubChem CID |
71664305
|
| 外观&性状 |
White to off-white solid powder
|
| tPSA |
38
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
19
|
| 分子复杂度/Complexity |
239
|
| 定义原子立体中心数目 |
2
|
| SMILES |
C1CC(CCC1N)N[C@@H]2C[C@H]2C3=CC=CC=C3.Cl.Cl
|
| InChi Key |
UCINOBZMLCREGM-RNNUGBGQSA-N
|
| InChi Code |
InChI=1S/C15H22N2.2ClH/c16-12-6-8-13(9-7-12)17-15-10-14(15)11-4-2-1-3-5-11;;/h1-5,12-15,17H,6-10,16H2;2*1H/t12?,13?,14-,15+;;/m0../s1
|
| 化学名 |
4-N-[(1R,2S)-2-phenylcyclopropyl]cyclohexane-1,4-diamine;dihydrochloride
|
| 别名 |
1431326-61-2; ORY-1001; 1431303-72-8; ORY-1001(trans); Iadademstat dihydrochloride;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ~57.75 mg/mL (~190.42 mM)
DMSO : ~0.69 mg/mL (~2.28 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 100 mg/mL (329.74 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.7480 mL | 18.7399 mL | 37.4799 mL | |
| 5 mM | 0.7496 mL | 3.7480 mL | 7.4960 mL | |
| 10 mM | 0.3748 mL | 1.8740 mL | 3.7480 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。