| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
|
| 靶点 |
β-lactam
Penicillin G binds to TEM-1 β-lactamase with a binding constant (Ka) of 1.41×10⁷ L/mol at 278 K, indicating strong affinity through hydrogen bonding and Van der Waals forces. The binding is spontaneous and exothermic.[1] |
|---|---|
| 体外研究 (In Vitro) |
青霉素G钠盐-TEM-1系统的紫外-可见(UV-Vis)吸收光谱与青霉素G钠盐和TEM-1 β-内酰胺酶的吸收光谱显着不同,表明两种酶之间形成了新的复合物。随着青霉素G钠盐浓度的增加,TEM-1β-内酰胺酶的紫外-可见吸收增加,并出现轻微的红移,表明青霉素G钠盐与TEM-1β-内酰胺酶之间的相互作用引起轻微的红移。 TEM-1 β-内酰胺酶的构象变化[1]。
Penicillin G 与 TEM-1 β-内酰胺酶相互作用,导致酶的静态荧光猝灭和构象变化。结合由焓变驱动,结合常数随温度升高而降低(278 K 时为 1.41×10⁷ L/mol,288 K 时为 7.62×10⁶ L/mol)。结合位点数约为 1,表明形成 1:1 复合物。TEM-1 与青霉素 G 之间的能量转移距离为 2.08 nm。[1] |
| 体内研究 (In Vivo) |
根据logistic回归模型,青霉素G钠盐处理组的猪拭子阳性概率比对照组低1.6倍(P<0.05)。与每板零菌落相比,对照组拭子含有10至99个菌落的风险是接受青霉素G钠盐处理的猪的2.3倍(P=0.022)[2]。
在一项涉及896头断奶仔猪(18日龄)的田间试验中,通过饮用水给予Potassium Penicillin G(剂量为每升水297,000 IU),进行两个为期5天的治疗期(断奶后第1-5天和第21-25天),显著降低了死亡率和猪链球菌携带率。治疗组的总死亡率为7.1%(32/448头猪),而对照组为14.0%(59/420头猪)。治疗组中归因于猪链球菌的死亡率(29头猪)也显著低于对照组(53头猪)。与对照组相比,治疗组猪的扁桃体猪链球菌携带量(每板≥1000菌落)也显著降低。[2] |
| 酶活实验 |
将 TEM-1β-内酰胺酶溶液 (5×10-6 M) 与不同浓度的头孢氨苄、头孢西丁和青霉素 G 钠盐溶液在 278 K 下混合。将三种抗生素逐渐添加至浓度 0 至 25×10 -6 M. 混合和相互作用 2 分钟后,用 2 nm 狭缝和 400 nm/min 扫描速度的分光光度计记录紫外-可见 (UV-Vis) 吸收光谱,并加入 0.02 M 磷酸盐缓冲液 (pH 7.0)作为参考[1]。
在 278 K 下,于磷酸缓冲液(pH 7.0)中,记录 TEM-1 β-内酰胺酶(5.0×10⁻⁶ mol/L)与不同浓度 penicillin G(0–25×10⁻⁶ mol/L)的紫外-可见吸收光谱。光谱显示增色效应和红移,表明复合物形成。[1] 在 278 K、283 K 和 288 K 下,于激发波长 278 nm 和 295 nm 处,测量 TEM-1 β-内酰胺酶(2.5×10⁻⁷ mol/L)与不同浓度 penicillin G(0–11.25×10⁻⁸ mol/L)的荧光发射光谱。使用 Stern-Volmer 方程和修正的 Stern-Volmer 方程分析猝灭数据,以确定猝灭常数和结合参数。[1] 记录同步荧光光谱(Δλ = 15 nm 用于酪氨酸,Δλ = 60 nm 用于色氨酸)以监测结合后的构象变化。[1] 进行碘化钾猝灭实验,以评估 TEM-1 和 TEM-1-penicillin G 复合物中发色团残基的表面可及性。[1] |
| 动物实验 |
本研究采用随机完全区组设计,设置两个重复。每个重复包含16个猪栏,每个猪栏饲养28头猪,共计448头猪。猪按体重分拣,并根据目测体重相近将猪分组到同一区组内。通过正式的随机化程序,在每个区组内的两个相邻猪栏中随机分配两种处理。两个处理组分别为处理组(青霉素G钠盐处理)和对照组(不处理)。在两个处理阶段,连续五天给予含青霉素G钠盐的饮用水。第1天(仔猪被转移到保育舍的当天)标志着第一个治疗阶段的开始,该阶段于第5天结束。第21天标志着第二个治疗阶段的开始,该阶段持续到第25天。对照组不接受任何治疗[2]。
本研究共使用了来自一家有2型猪链球菌感染史的商业养殖场的896头18日龄断奶仔猪。仔猪按性别和体重分组,并在猪栏内随机分配到治疗组或对照组。治疗组在饮用水中添加了青霉素钾,最终计算剂量为每升297,000 IU。该药物的制备方法是将85克等分试样溶于水中,制成储备液,然后通过比例分配器以1:128(储备液:水)的比例将其添加到饮水系统中。每天配制两次新鲜的储备液。治疗分两个阶段进行,每个阶段持续5天:从断奶当天(第1天)开始,再从断奶后第21天开始。对照组不接受任何治疗。在三个时间点(第1天(治疗前)、第6天(第一次治疗后)和第26天(第二次治疗后))从每个猪栏随机选择4头猪采集扁桃体拭子,用于细菌培养和定量。[2] 出现猪链球菌感染临床症状(共济失调、震颤、侧卧)的猪接受单独肌注头孢噻呋盐酸盐(3 mg/kg)治疗。如果一个猪栏中有超过3头猪出现症状,则对整个猪栏进行治疗。重病或濒死的猪被实施安乐死,并进行尸检以确定死因。[2] |
| 参考文献 |
|
| 其他信息 |
青霉素G钠盐是一种白色至微黄色结晶性粉末,略带气味。pH值(10%溶液)为5.5-7.5。(NTP,1992)
苄青霉素钠是一种有机钠盐。它含有苄青霉素(1-)基团。 青霉素G(钾或钠)是一种处方抗菌药物,经美国食品药品监督管理局(FDA)批准,用于治疗某些严重的细菌感染,例如肺炎、脑膜炎、淋病、梅毒等。 肺炎和梅毒可能是HIV的机会性感染。 青霉素G钠是苄青霉素的钠盐形式,苄青霉素是一种半合成的广谱青霉素类抗生素,具有杀菌活性。苄青霉素钠与位于细菌细胞壁内膜上的青霉素结合蛋白 (PBP) 结合并使其失活。PBP 的失活会干扰肽聚糖链的交联,而肽聚糖链的交联对于维持细菌细胞壁的强度和刚性至关重要。这会导致细菌细胞壁的弱化,最终导致细胞裂解。 苄青霉素钠是一种青霉素衍生物,通常以钠盐或钾盐的形式用于治疗多种感染。它对大多数革兰氏阳性菌和革兰氏阴性球菌有效。由于其对 γ-氨基丁酸 (GABA) 介导的突触传递的作用,它也曾被用作实验性惊厥剂。 另见:青霉素 G(具有活性部分)。 青霉素 G 是一种 β-内酰胺类抗生素,其结构包含一个与噻唑烷环稠合的 β-内酰胺环。它主要通过与 Ala237、Ser70、Ser130、Asn132、Arg244 和 Ser235 残基形成氢键与 TEM-1 β-内酰胺酶结合,共形成 6 个氢键。这种结合诱导酶的构象变化,增加色氨酸残基周围的极性。该研究为设计新型抗 TEM-1 β-内酰胺酶抗生素提供了思路。[1] |
| 分子式 |
C16H17N2NAO4S
|
|---|---|
| 分子量 |
356.3720
|
| 精确质量 |
356.08
|
| 元素分析 |
C, 53.93; H, 4.81; N, 7.86; Na, 6.45; O, 17.96; S, 9.00
|
| CAS号 |
69-57-8
|
| 相关CAS号 |
Penicillin G potassium;113-98-4;Penicillin G procaine hydrate;6130-64-9;Penicillin G benzathine;1538-09-6;Penicillin G benzathine tetrahydrate;41372-02-5;Penicillin G;61-33-6
|
| PubChem CID |
23668834
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.41
|
| 沸点 |
663.3ºC at 760 mmHg
|
| 熔点 |
209-212°C
|
| 折射率 |
300 ° (C=2, H2O)
|
| tPSA |
114.84
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
4
|
| 重原子数目 |
24
|
| 分子复杂度/Complexity |
536
|
| 定义原子立体中心数目 |
3
|
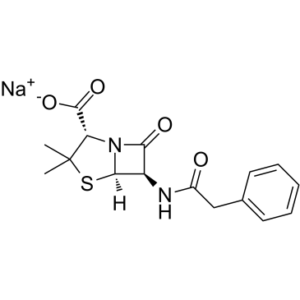
| SMILES |
S1C(C([H])([H])[H])(C([H])([H])[H])[C@]([H])(C(=O)[O-])N2C([C@]([H])([C@@]12[H])N([H])C(C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H])=O)=O.[Na+]
|
| InChi Key |
FCPVYOBCFFNJFS-LQDWTQKMSA-M
|
| InChi Code |
InChI=1S/C16H18N2O4S.Na/c1-16(2)12(15(21)22)18-13(20)11(14(18)23-16)17-10(19)8-9-6-4-3-5-7-9;/h3-7,11-12,14H,8H2,1-2H3,(H,17,19)(H,21,22);/q;+1/p-1/t11-,12+,14-;/m1./s1
|
| 化学名 |
4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-((2-phenylacetyl)amino)- (2S,5R,6R)-, sodium salt (1:1)
|
| 别名 |
Benzylpenicillin sodium salt; Penicillin G sodium salt; Monosodium benzylpenicillin; Mycofarm; Novocillin; NSC 402815; OK 431; Pen-A-Brasive;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: (1). 该产品在溶液状态不稳定,请现配现用。 (2). 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : 71~100 mg/mL ( 199.23~280.61 mM )
Water : 71~125 mg/mL(~350.76 mM) Ethanol : ~1 mg/mL |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (7.02 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (7.02 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (7.02 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 2.5 mg/mL (7.02 mM) 配方 5 中的溶解度: 100 mg/mL (280.61 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8061 mL | 14.0304 mL | 28.0607 mL | |
| 5 mM | 0.5612 mL | 2.8061 mL | 5.6121 mL | |
| 10 mM | 0.2806 mL | 1.4030 mL | 2.8061 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
|