| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |

| 药代性质 (ADME/PK) |
吸收、分布和排泄
肌内注射和皮下注射后均能迅速吸收。肠外给药后初始血药浓度较高,但持续时间短暂。空腹健康人口服吸收率仅为15-30%左右,因为它极易被酸催化水解。 青霉素G主要经肾脏排泄。非肾清除途径包括肝脏代谢,以及少量胆汁排泄。 肾功能正常的成年人每日血药浓度为0.53-0.67 L/kg 健康人每日血药浓度为560 ml/min ……广泛分布于全身……其表观分布容积约占体液总量的50%。血液中超过90%的药物存在于血浆中,不到10%存在于红细胞中;约65%可逆性地与血浆白蛋白结合。蛋白质浓度低……结合程度低……药物疗效。 肝脏、胆汁、肾脏、精液、淋巴和肠道中均可见显著的活性药物成分。……当脑膜正常时,青霉素不易进入脑脊液。 人体口服500毫克钾青霉素后,2小时内尿液中青霉素浓度为600微克/毫升,4小时内为300微克/毫升。 ……胎盘转运效率低下与青霉素G的低脂溶性和低电离常数相符,并且没有证据表明存在胎盘转运。 ……迅速从体内清除,主要通过肾脏,但少量通过胆汁和其他途径。由于肾功能发育不完全,新生儿和婴儿的清除率明显较低…… 有关青霉素G(共21种)的更多吸收、分布和排泄(完整)数据,请访问HSDB记录页面。 代谢/代谢物 肌注剂量约有16-30%代谢为青霉素酸,一种无活性代谢物。在服用青霉素G的患者尿液中检测到少量6-氨基青霉烷酸。少量药物似乎被羟基化为一种或多种活性代谢物,这些代谢物也通过尿液排出。肌注青霉素G钠约有16-30%代谢为青霉酸,而青霉酸在微生物学上无活性。在接受青霉素G治疗的患者尿液中也发现了少量6-氨基青霉烷酸(6-APA)。此外,该药物似乎会被少量羟基化,生成一种或多种具有微生物活性的代谢物,这些代谢物也会随尿液排出。 生物半衰期 据报道,肾功能正常的成年人的生物半衰期为0.4-0.9小时。 正常成年人的消除半衰期约为30分钟。 青霉素在人血清中的半衰期从年轻成年人的约25分钟增加到老年人的2小时,并且肾小管主动分泌的药物也会显著延长其半衰期。 /青霉素/ 据报道,肾功能正常的成年人血清中青霉素G的半衰期为0.4-0.9小时。 新生儿血清中青霉素G的半衰期与年龄呈反比,且似乎与出生体重无关。据报道,6日龄及以下新生儿的血清半衰期为3.2-3.4小时,7-13日龄新生儿为1.2-2.2小时,14日龄及以上新生儿为0.9-1.9小时。 |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
妊娠期和哺乳期用药
◉ 哺乳期用药概述 现有信息有限,表明青霉素G在乳汁中的浓度较低,预计不会对母乳喂养的婴儿造成不良影响。有报道称,青霉素类药物偶尔会扰乱婴儿的胃肠道菌群,导致腹泻或鹅口疮,但这些影响尚未得到充分评估。哺乳期妇女可以安全使用青霉素G。 ◉ 对母乳喂养婴儿的影响 一名患有先天性梅毒的1月龄母乳喂养婴儿,在其母亲肌注240万单位苄星青霉素G 6小时后出现赫氏反应。然而,该婴儿在母亲注射青霉素G的同时,也接受了10单位青霉素G。该反应可能是由母乳中的青霉素引起的。 ◉ 对哺乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 蛋白质结合 与血清蛋白结合(45-68%),主要与白蛋白结合。 相互作用 一名49岁男性患者报告,大剂量肠外注射青霉素G可显著增强华法林的低凝血酶原血症作用。青霉素G引起华法林蛋白质结合置换相互作用。 青霉素通常在高温、碱性或酸性pH值、氧化剂、醇类、乙二醇类以及铜、汞或锌等金属离子存在下失活。目前市售的青霉素类药物,其核心结构(包括β-内酰胺环)的任何部位发生断裂都会导致抗菌活性完全丧失。青霉素失活的主要原因是β-内酰胺环的水解。水解过程和降解产物的性质可能有所不同,并且通常受pH值的影响。/青霉素/ 青霉素G可能与某些药物存在物理和/或化学不相容性,包括氨基糖苷类和四环素类药物,但这种相容性取决于多种因素(例如,药物浓度、使用的特定稀释剂、最终pH值、温度)。 青霉素通常在高温、碱性或酸性pH值、氧化剂、醇类、乙二醇类以及铜、汞或锌等金属离子存在下失活。目前市售的青霉素类药物,其分子核内任何位置(包括β-内酰胺环)的断裂都会导致抗菌活性完全丧失。青霉素失活的主要原因是β-内酰胺环的水解。水解过程和降解产物的性质可能有所不同,并且通常受pH值的影响。/青霉素/ 如需查看更多关于青霉素G(共18种)的相互作用(完整)数据,请访问HSDB记录页面。 |
| 其他信息 |
治疗用途
惊厥药;GABA调节剂;青霉素 由呼吸道中存在的具核梭杆菌(梭形杆菌)和螺旋体协同作用引起的龈口炎、肺部感染和生殖器疾病,均可用青霉素轻松治疗。/青霉素/ 导致鼠咬热的两种微生物对青霉素G敏感。……是治疗单核细胞增生菌感染的首选药物……唯一对青霉素高度敏感的巴斯德氏菌属细菌是多杀性巴斯德氏菌。丹毒的病原体对青霉素敏感。 青霉素G治疗梅毒几乎是理想的安全、廉价且高效的药物。……是治疗所有临床表现的放线菌病、炭疽、气性坏疽的首选药物。 有关青霉素G(共35种)的更多治疗用途(完整)数据,请访问HSDB记录页面。 药物警告 当使用大剂量青霉素G钠时,会引入大量的钠负荷,这会扩张细胞外间隙,并可能导致心力衰竭患者出现水肿。青霉素G钠 对普鲁卡因成分可能产生过敏反应,但普鲁卡因的其他毒性作用非常罕见。 /普鲁卡因/ 无尿症可使青霉素G的半衰期从正常的0.5小时延长至约10小时。 尽管用于吸入疗法和皮肤及黏膜局部应用的青霉素G制剂(PrePN)仍然可用,但不建议使用,因为缺乏其疗效充分的证据,且其超敏反应发生率高。 有关青霉素G(共23条)的更多药物警告(完整)数据,请访问HSDB记录页面。 药效学 青霉素G是一种青霉素类β-内酰胺类抗生素,用于治疗由敏感菌株引起的细菌感染,通常革兰氏阳性菌。 “青霉素”这个名称既可以指几种现有的青霉素衍生物,也可以指一类源自青霉素的抗生素。青霉素G对革兰氏阳性菌和革兰氏阴性需氧菌和厌氧菌均具有体外活性。青霉素G的杀菌活性源于其抑制细胞壁合成,并通过与青霉素结合蛋白(PBPs)结合而发挥作用。青霉素G对多种β-内酰胺酶(包括青霉素酶、头孢菌素酶和超广谱β-内酰胺酶)的水解作用稳定。 |
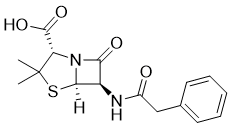
| 分子式 |
C16H18N2O4S
|
|---|---|
| 分子量 |
334.4
|
| 精确质量 |
334.098
|
| CAS号 |
61-33-6
|
| 相关CAS号 |
Penicillin G potassium;113-98-4;Penicillin G sodium salt;69-57-8;Streptomycin;57-92-1;Penicillin G procaine hydrate;6130-64-9;Penicillin G benzathine;1538-09-6;Penicillin G benzathine tetrahydrate;41372-02-5;Penicillin G-d7 potassium;352323-25-2
|
| PubChem CID |
5904
|
| 外观&性状 |
AMORPHOUS WHITE POWDER
|
| 密度 |
1.4±0.1 g/cm3
|
| 沸点 |
663.3±55.0 °C at 760 mmHg
|
| 熔点 |
214-217 °C
214 - 217 °C |
| 闪点 |
355.0±31.5 °C
|
| 蒸汽压 |
0.0±2.1 mmHg at 25°C
|
| 折射率 |
1.655
|
| LogP |
1.67
|
| tPSA |
112.01
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
4
|
| 重原子数目 |
23
|
| 分子复杂度/Complexity |
530
|
| 定义原子立体中心数目 |
3
|
| SMILES |
CC1([C@@H](N2C([C@@H](NC(CC3=CC=CC=C3)=O)[C@H]2S1)=O)C(O)=O)C
|
| InChi Key |
JGSARLDLIJGVTE-MBNYWOFBSA-N
|
| InChi Code |
InChI=1S/C16H18N2O4S/c1-16(2)12(15(21)22)18-13(20)11(14(18)23-16)17-10(19)8-9-6-4-3-5-7-9/h3-7,11-12,14H,8H2,1-2H3,(H,17,19)(H,21,22)/t11-,12+,14-/m1/s1
|
| 化学名 |
(2S,5R,6R)-3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
|
| 别名 |
Galofak CilopenBenzylpenicillin Pradupen
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9904 mL | 14.9522 mL | 29.9043 mL | |
| 5 mM | 0.5981 mL | 2.9904 mL | 5.9809 mL | |
| 10 mM | 0.2990 mL | 1.4952 mL | 2.9904 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。