| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
AMPK α2β1γ1 (EC50 = 6.8 nM); AMPK α1β1γ1 (EC50 = 7 nM)
The target of PF-06409577 is adenosine monophosphate-activated protein kinase (AMPK), specifically acting as a direct activator of AMPK. [1] PF-06409577 is a direct activator of AMPK, with a preference for the β1 subunit of AMPK (α1β1γ1 isoform), [2] |
|---|---|
| 体外研究 (In Vitro) |
PF-06409577 激活 AMPK 亚型 α1β1γ1 和 α2β1γ1,EC50 值分别为 7.0 nM 和 6.8 nM,但针对 α1β2γ1/α2β2γ1/α2β2γ3 亚型 α1β1γ1 和 α2β1γ1 的活性明显较低,EC50 值大于 4000 nM[1].10 0 µM)主要人类细胞色素 P450 亚型的微粒体活性。
1. 在分子对接研究中,将PF-06409577对接至AMPK的α1β1γ1亚型。PF-06409577的吲哚母核相较于其在X射线晶体结构中的原始构象出现轻微偏转,且该化合物与AMPK α1β1γ1亚型的活性位点之间形成氢键,这是其能够直接激活AMPK的结构基础[2] |
| 体内研究 (In Vivo) |
PF-06409577 在糖尿病肾病临床前模型中表现出功效[1]。它在大鼠(血浆未结合部分,fu,p = 0.0044)、狗(fu,p = 0.028)、猴(fu,p = 0.032)和人(fu,p = 0.017)中表现出高血浆蛋白结合。静脉注射给药后,PF-06409577 在大鼠 (22.6 mL/min/kg)、狗 (12.9 mL/min/kg) 和猴子 (8.57 mL/min/kg) 中表现出中等的血浆清除率 (CLp),并且分布良好稳态分布体积 (Vdss) 范围为 0.846-3.15 L/kg。当大鼠、狗和猴子口服给药时,0.5% 甲基纤维素悬浮液中的结晶 PF-06409577 很快被吸收(Tmax = 0.25-1.20 小时)。大鼠、狗和猴子的口服生物利用度 (F) 值分别为 15%、100% 和 59%。与其他临床前物种和人类相比,PF-06409577 在大鼠体内遭受更大程度的首过肠道葡萄糖醛酸化[2]。
|
| 酶活实验 |
PF-06409577 在 DMSO 中制备。将 PF-06409577 与完全磷酸化的 AMPK 在测定缓冲液中在室温下孵育 15 分钟,然后添加 PP2a,并在室温下再次孵育 60 分钟。在停止磷酸酶处理并开始激酶测定之前,添加冈田酸(最终 50 nM)、50 nM Cy-5 SAMS 肽和等于每种亚型 Km 的 ATP。反应再孵育 60 分钟后,通过向检测缓冲液中添加 10 mM EDTA 和 2 nM Eu-pACC 抗体来猝灭激酶反应。分别使用 320 nM 的激发和 665 和 615 nM 的发射测量来测量激酶活性。
|
| 细胞实验 |
PF-06409577 对人类和大鼠 α1β1γ1 亚型具有相似的效力。在针对其他受体、通道、PDE 和激酶的广泛筛选中,PF-06409577 表现出最小的脱靶药理学。 PF-06409577 在膜片钳测定 (100 µM) 中未显示出可检测到的 hERG 抑制作用,并且不是主要人细胞色素 P450 亚型的微粒体活性的抑制剂 (IC50>100 µM)。
|
| 动物实验 |
大鼠:连续68天,每日分别给予饮用水中添加雷米普利(1 mg/kg/天)、PF-06409577(10、30或100 mg/kg,口服)、PF-249(3、10或30 mg/kg,口服)或0.5%甲基纤维素溶液(口服)。分别于给药14、28、42和60天后,收集所有瘦鼠和肥胖鼠的24小时尿液,并记录尿量。所有大鼠在第63天禁食16小时后接受最后一次给药。最后一次给药1小时后,采集100 μL尾静脉血样,用于测定总蛋白和胰岛素水平。同时使用血糖仪测量血糖。随后使用异氟烷麻醉每只大鼠。取出右肾,立即进行冷冻钳夹,并转移至液氮中保存。将左肾固定于 10% 福尔马林溶液中。然后通过下腔静脉放血处死大鼠[1]
|
| 药代性质 (ADME/PK) |
对先导化合物(吲唑酸)的核心和芳基侧链进行优化,提高了PF-06409577的口服吸收率,但文献[1]中未描述具体的ADME参数(如吸收率、分布、代谢、排泄、半衰期、口服生物利用度等)。
|
| 参考文献 | |
| 其他信息 |
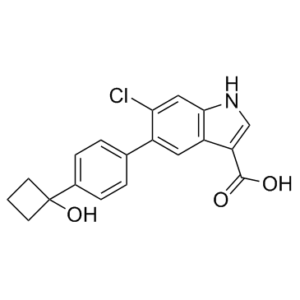
1. PF-06409577(化学名称:6-氯-5-[4-(1-羟基环丁基)苯基]-1H-吲哚-3-羧酸)是一种吲哚酸化合物,也是AMPK的直接激活剂。它是基于人类遗传关联数据开发的,具有治疗糖尿病肾病的潜力。该化合物的发现源于高通量筛选 (HTS) 鉴定出的吲唑酰胺先导化合物,在截短至其最小药效团得到吲唑酸先导化合物后,对核心和芳基侧链进行优化,提高了口服吸收率,最终鉴定出 PF-06409577,该化合物已进入首次人体试验,用于治疗糖尿病肾病 [1]
2. AMPK 是一种参与维持细胞能量稳态的蛋白激酶,直接 β1 选择性 AMPK 激活剂(包括 PF-06409577)因其在治疗糖尿病肾病患者方面的潜力而重新引起人们的关注。在基于结构的虚拟筛选和分子对接研究中,PF-06409577 被用作 AMPK 激活剂筛选的参考化合物,并分析了其与 AMPK α1β1γ1 亚型的结合模式,以指导新型 β1 选择性 AMPK 激活剂的鉴定 [2] |
| 分子式 |
C19H16CLNO3
|
|
|---|---|---|
| 分子量 |
341.79
|
|
| 精确质量 |
341.082
|
|
| 元素分析 |
C, 66.77; H, 4.72; Cl, 10.37; N, 4.10; O, 14.04
|
|
| CAS号 |
1467057-23-3
|
|
| 相关CAS号 |
|
|
| PubChem CID |
71748255
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
4.558
|
|
| tPSA |
73.32
|
|
| 氢键供体(HBD)数目 |
3
|
|
| 氢键受体(HBA)数目 |
3
|
|
| 可旋转键数目(RBC) |
3
|
|
| 重原子数目 |
24
|
|
| 分子复杂度/Complexity |
487
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
ClC1=CC2=C(C(C(=O)O)=CN2)C=C1C1C=CC(=CC=1)C1(CCC1)O
|
|
| InChi Key |
FHQXLWCFSUSXBF-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C19H16ClNO3/c20-16-9-17-14(15(10-21-17)18(22)23)8-13(16)11-2-4-12(5-3-11)19(24)6-1-7-19/h2-5,8-10,21,24H,1,6-7H2,(H,22,23)
|
|
| 化学名 |
6-Chloro-5-[4-(1-hydroxycyclobutyl)phenyl]-1H-indole-3-carboxylic acid
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (6.09 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (6.09 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (6.09 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9258 mL | 14.6289 mL | 29.2577 mL | |
| 5 mM | 0.5852 mL | 2.9258 mL | 5.8515 mL | |
| 10 mM | 0.2926 mL | 1.4629 mL | 2.9258 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Status | Interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02286882 | Terminated | Drug: PF-06409577 or Placebo |
Healthy | Pfizer | November 2014 | Phase 1 |
(A) In vitro AMPK activity for selected AMPK heterotrimers (αβγ) activated with PF-06409577. (B) In vitro AMPK activity for selected AMPK heterotrimers (αβγ) activated with PF-249. (C) Ribbon representation of crystal structure of AMPKα1β1γ1 bound to PF-249. (D) Close-up view of the ligand-protein interface. (E) AMPKβsubunit levels in kidney tissue were measured by quantitative ELISA and plotted as percentage of AMPK heterotrimer containing theβ1 subunit.J Pharmacol Exp Ther.2017 May;361(2):303-311. |
|---|
(A) Western blot for AMPKβ1,β2, and pan-αin 293FT cells transfected with scrambled siRNA or siRNA targeting AMPKβ1. (B) ELISA quantification of ACC phosphorylation status in 293FT treated with siRNA and PF-06409577. (C) ELISA quantification of ACC phosphorylation status in 293FT treated with siRNA and PF-06409577.J Pharmacol Exp Ther.2017 May;361(2):303-311. |
(A) Cumulative urinary albumin excretion over a 24-hour period at multiple time points during a dosing study in obese ZSF1 rats treated with vehicle or PF-06409577; 10 or 11 animals per group. (B) Kidney AMPK phosphorylation status in terminal samples after 8 weeks of dosing, 1 hour after the last dose.J Pharmacol Exp Ther.2017 May;361(2):303-311. |
(A) Cumulative albumin excretion over a 24-hour period at multiple time points during a dosing study in ZSF1 rats treated with vehicle, PF-249, PF-06409577, or ramipril (1 mg/kg/d in drinking water); 12 animals per group. (B) Kidney AMPK phosphorylation status in terminal samples after 8 weeks of dosing, 1 hour after the last dose.J Pharmacol Exp Ther.2017 May;361(2):303-311. |
|---|
(A–C) Representative histology images of phospo-S6 (p-S6) reactivity in kidneys collected after 8 weeks of dosing vehicle or PF-06409577 to ZSF1 rats; 12 animals per group. Glomeruli are indicated by the arrows and stain area was quantified. (D) Quantitative measure of p-S6 stain area in glomeruli of five lean ZSF1 treated with vehicle, seven obese ZSF1 treated with vehicle, and seven obese ZSF1 animals treated with PF-06409577.J Pharmacol Exp Ther.2017 May;361(2):303-311. |
Quantitative PCR measurements of mRNA for (A)Col1a1, (B)Col4a1, (C)Nox4, and (D)Ppargc1ain kidney samples from animals treated with 3, 10, and 30 mg/kg PF-249, 100 mg/kg PF-06409577 (PF-577), or ramipril for 8 weeks; 8–10 animals per group. Significance compared with the obese vehicle group.J Pharmacol Exp Ther.2017 May;361(2):303-311. |