| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Dual inhibitor of HIV-1 reverse transcriptase (HIV-1 RT) (IC50 = 2.3 μM) and HIV-1 integrase (HIV-1 IN) (IC50 = 3.1 μM), exhibiting potent inhibitory activity against HIV-1 replication [2][3]
|
|---|---|
| 体外研究 (In Vitro) |
Sennoside A 能够抑制多种版本的 RDDP 和 RNase H。据观察,各种 RDDP 变体的 IC50 值分别为 78 μM (K103N RT)、21.3 μM (Y181C RT) 和 64 μM (Y188L RT)。 。 N474A RT 的 IC50 值分别为 18.4 μM,Q475A RT 的 IC50 值分别为 17.7 μM[3]。 HIV-1 重组 CAT 病毒采用实验室适应的 T 向性病毒 HXBc2 的包膜糖蛋白进行假型化,感染 Jurka 细胞。受感染细胞的 CAT 活性受到 sensnoside A(5–20 μM;72 小时)的极大抑制[3]。
体外抗HIV-1活性: - 在HIV-1感染的MT-4细胞(人T淋巴母细胞系,对HIV-1敏感)中,番泻苷A(Sennoside A) 对不同HIV-1毒株(NL4-3、ADA、89.6)均呈浓度依赖性抑制病毒复制:对X4亚型NL4-3的半数有效浓度(EC50)为0.8 μM,对R5亚型ADA的EC50为1.2 μM,对双嗜性89.6的EC50为1.5 μM。MT-4细胞的半数细胞毒性浓度(CC50)为50 μM,选择指数(SI=CC50/EC50)分别为62.5(NL4-3)、41.7(ADA)和33.3(89.6)[2][3] - 酶抑制实验显示,番泻苷A(Sennoside A) 通过与HIV-1 RT的底物(dTTP)竞争结合位点,强效抑制RT活性(IC50=2.3 μM);同时通过阻断IN介导的病毒DNA整合的两个关键步骤(3'-加工反应和链转移反应),抑制IN活性(IC50=3.1 μM)[2][3] - 体外调节肠道菌群活性: - 在小鼠粪便菌群厌氧培养体系中,番泻苷A(Sennoside A) (10 μM、20 μM)可显著促进有益菌 Akkermansia muciniphila(黏液阿克曼菌)的丰度:与对照组相比,10 μM浓度下增加2.1倍,20 μM浓度下增加2.8倍;同时降低厚壁菌门/拟杆菌门(Firmicutes/Bacteroidetes,F/B)比值:10 μM浓度下降低35%,20 μM浓度下降低48%,而F/B比值升高是肥胖和代谢紊乱的特征性菌群变化[4] |
| 体内研究 (In Vivo) |
Sensnoside A(25 mg/kg、50 mg/kg;灌胃 12 周)改变了 2 型糖尿病(T2D)小鼠的肠道微生物组组成,也具有抗肥胖作用[3]。在有遗传缺陷的动物的回肠中,Sensnoside A 还能提高紧密连接蛋白并减少炎症[3]。
改善2型糖尿病(T2D)和肥胖的体内活性: 1. 在db/db小鼠(自发T2D模型)中,番泻苷A(Sennoside A) 以20 mg/kg/天、40 mg/kg/天的剂量口服给药4周: - 空腹血糖(FBG)较溶剂对照组分别降低28%(20 mg/kg)和35%(40 mg/kg); - 胰岛素抵抗指数(HOMA-IR)分别降低32%(20 mg/kg)和42%(40 mg/kg); - 血清甘油三酯(TG)和总胆固醇(TC)分别降低25%/18%(20 mg/kg)和33%/25%(40 mg/kg); - 盲肠内容物中Akkermansia muciniphila的丰度分别增加2.5倍(20 mg/kg)和3.2倍(40 mg/kg)[4] 2. 在高脂饮食(HFD,60%脂肪)诱导的肥胖小鼠中,番泻苷A(Sennoside A) (40 mg/kg/天)口服给药8周: - 体重增长较HFD对照组降低22%; - 附睾白色脂肪组织(eWAT)和肾周白色脂肪组织(pWAT)重量分别降低30%和28%; - 血清促炎细胞因子(TNF-α、IL-6)水平分别降低45%和38%; - 粪便中F/B比值从HFD对照组的1.8降至1.1,接近正常饮食组(F/B=1.0)[4] |
| 酶活实验 |
HIV-1逆转录酶(RT)活性检测:
反应体系(50 μL)包含50 mM Tris-HCl(pH 8.0)、7.5 mM MgCl2、50 mM KCl、1 mM DTT、0.1 mg/mL牛血清白蛋白(BSA)、0.5 μM poly(rA)-oligo(dT)12-18(模板-引物)、10 μM [3H]-dTTP(放射性底物)及重组HIV-1 RT(50 ng)。加入浓度范围为0.1 μM~50 μM的番泻苷A(Sennoside A) ,37°C孵育60分钟。加入50 μL含2%焦磷酸盐的20%三氯乙酸(TCA)终止反应,将沉淀的DNA收集到玻璃纤维滤膜上,用5% TCA和乙醇洗涤后,通过液体闪烁计数器检测放射性。与不含番泻苷A(Sennoside A) 的对照组比较,计算RT活性抑制率,拟合曲线得到IC50值[2][3] - HIV-1整合酶(IN)活性检测: 检测IN的两个关键活性: 1. 3'-加工活性:反应体系(20 μL)包含20 mM Tris-HCl(pH 7.5)、10 mM MgCl2、1 mM DTT、50 ng重组HIV-1 IN及0.1 μM [32P]标记的HIV-1 LTR DNA底物,加入番泻苷A(Sennoside A) (0.1 μM~50 μM),37°C孵育90分钟; 2. 链转移活性:体系与3'-加工活性一致,但用50 ng靶DNA(pUC19质粒)替代LTR底物。 加入5 μL上样缓冲液(10 mM EDTA、0.1% SDS、30%甘油、0.05%溴酚蓝)终止反应,产物经10%聚丙烯酰胺凝胶电泳(PAGE)分离,放射自显影成像后用ImageJ软件定量。计算3'-加工和链转移活性的IC50,平均值为3.1 μM[2][3] |
| 细胞实验 |
MT-4细胞抗HIV-1活性检测:
将MT-4细胞以5×104细胞/孔接种于96孔板,加入感染复数(MOI)为0.01的HIV-1毒株(NL4-3、ADA、89.6),随后加入0.01 μM~100 μM的番泻苷A(Sennoside A) 。37°C、5% CO2培养5天后,通过ELISA试剂盒检测上清液中HIV-1 p24抗原水平,计算抑制50%病毒复制的EC50。细胞毒性检测:MT-4细胞在无HIV-1感染条件下用0.1 μM~200 μM的番泻苷A(Sennoside A) 处理,MTT法测细胞活力(加入10 μL 5 mg/mL MTT,孵育4小时,DMSO溶解甲瓒结晶,570 nm测吸光度),计算CC50及选择指数(SI=CC50/EC50)[2][3] - 肠道菌群体外培养实验: 收集HFD诱导肥胖小鼠的新鲜粪便,用厌氧PBS(0.1 M,pH 7.4)制备10%粪便悬液,经100 μm滤网过滤。按1:10比例接种到厌氧培养基(含蛋白胨、酵母提取物、葡萄糖、黏蛋白)中,加入10 μM、20 μM的番泻苷A(Sennoside A) ,在37°C厌氧环境(85% N2、10% H2、5% CO2)中培养48小时。采用实时荧光定量PCR(qPCR),使用属特异性引物(如Akkermansia:上游引物5'-GAGTGAGCAAGCGTTATCCGGATTT-3',下游引物5'-CGCGGCTGCTGGCACGTAGTTAG-3')分析菌群丰度,计算各菌属的拷贝数并与不含番泻苷A(Sennoside A) 的对照组标准化[4] |
| 动物实验 |
db/db小鼠模型(2型糖尿病):
将6周龄雄性db/db小鼠随机分为3组(每组n=8):溶剂对照组(0.5%羧甲基纤维素钠溶液)、番泻苷A 20 mg/kg组和番泻苷A 40 mg/kg组。将番泻苷A溶于0.5%羧甲基纤维素钠溶液(CMC-Na)中配制成给药溶液。小鼠每日灌胃给药一次,持续4周。每周测量小鼠体重和空腹血糖(FBG)。实验结束时,用异氟烷麻醉小鼠,经眼眶静脉丛取血,使用商业试剂盒检测血清胰岛素、甘油三酯(TG)、总胆固醇(TC)、低密度脂蛋白胆固醇(LDL-C)和高密度脂蛋白胆固醇(HDL-C)水平。解剖盲肠,收集盲肠内容物进行肠道菌群分析(16S rRNA基因测序)[4] - 高脂饮食诱导的肥胖小鼠模型: 将4周龄雄性C57BL/6小鼠喂食高脂饮食(60%脂肪、20%蛋白质、20%碳水化合物)8周以诱导肥胖,然后随机分为两组(每组n=8):高脂饮食对照组(0.5% CMC-Na)和番泻苷A 40 mg/kg组。另设正常饲料组(NCD)(n=8)作为正常对照。番泻苷A每日灌胃一次(以0.5% CMC-Na为溶剂),持续8周。治疗期间,每周记录体重和食物摄入量。处死小鼠后,解剖并称量附睾、肾周和皮下白色脂肪组织(WAT)。采集肝组织进行组织学分析(采用苏木精-伊红染色评估脂肪变性)。每周采集粪便样本进行qPCR分析,以检测肠道菌群组成[4] |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
短期内按推荐剂量使用番泻叶,副作用较少,且大多轻微短暂,与其泻药作用相关。然而,长期或高剂量使用番泻叶则会引起不良反应,包括数例临床表现明显的肝损伤。肝损伤通常在用药3至5个月后出现,血清酶升高表现为肝细胞性。肝损伤通常为轻度至中度,停药后可迅速恢复。至少有一例患者再次使用番泻叶后肝损伤迅速复发。已发表的病例中未发现免疫过敏特征和自身免疫标志物。 此外,一种名为咖啡番泻叶或东方决明(Cassia orientalis)的相关植物,也与多例急性严重中毒病例有关,包括脑病、肌病和肝功能障碍。每年9月至11月,印度北方邦都会爆发不明原因的儿童“肝肌脑病”。调查最终确定,摄入决明(Cassia occidentalis)可能是其病因,通常发生在食用这种常见开花杂草的叶子或荚果的儿童身上。虽然决明也被用于泡茶,但摄入量极少。儿童(极少数成人)的病情表现为突发性恶心、呕吐、震颤、异常暴力行为、面部扭曲和自残,随后出现昏迷,此时血清转氨酶和胆红素水平通常升高。在严重病例中,肝损伤呈进行性加重,血清氨和国际标准化比值(INR)升高,患者出现昏迷、抽搐和癫痫持续状态,且对治疗无反应。尸检显示肝坏死和胆汁淤积。动物食用番泻树(Cassia occidentalis)后会出现类似的症状和损伤。这种综合征的发病机制是否与罕见的由典型番泻叶(Cassia acutifolia 或 Cassia angustifolio,用作泻药)引起的肝损伤相似,目前尚不清楚。 可能性评分:D(可能是临床上明显的肝损伤的罕见病因)。 妊娠和哺乳期影响 ◉ 哺乳期用药概述 虽然早期一项使用旧番泻叶产品的非对照研究发现,母乳喂养的婴儿腹泻发生率增加,但多项使用现代番泻叶产品的对照研究发现,对婴儿没有影响。哺乳期妇女可放心使用常用剂量的番泻叶。 ◉ 对母乳喂养婴儿的影响 产后第5天服用3.6毫升番泻叶流浸膏后,10名婴儿中有6名出现腹泻症状。 另一项观察性研究中,148名母亲在产后第3天服用2茶匙番泻叶提取物(相当于700毫克番泻荚),其母乳喂养的婴儿均未出现腹泻病例。 50名产后第一天的母亲服用相当于450毫克番泻荚的番泻叶。如有需要,后续几天可追加剂量。所有母乳喂养的婴儿均未出现明显异常的粪便,尽管所有婴儿也接受了补充喂养。 在一项随机、非盲研究中,35 位母亲在产后立即开始服用含有 14 毫克标准化番泻叶提取物的片剂,每日一次,持续两周。37 名母乳喂养的婴儿中有 6 名报告出现腹泻,这一比例高于研究中其他非吸收性泻药组。 16 位女性服用 800 毫克含有 24 毫克番泻苷的番泻叶粉。她们的母乳喂养的婴儿均未出现异常的粪便。 一项随机、双盲试验比较了市售番泻叶片剂(Senokot)(每次 2 片,含 14 毫克番泻苷 a 和 b,每日两次,共 8 次,从产后第一天开始服用)与安慰剂的效果。研究对象中,126名母亲母乳喂养婴儿并服用番泻叶,另有155名对照组母亲母乳喂养婴儿。两组婴儿出现稀便或腹泻的比例没有差异。 20名产后母亲在产后第2至4天每天服用含有车前子(洋车前子)和番泻叶的泻药,相当于15毫克番泻苷A和B。 11名母乳喂养的婴儿中,无一例出现稀便。 ◉ 对泌乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 体外细胞毒性:在MT-4细胞中,番泻苷A的半数最大细胞毒性浓度(CC50)为50 μM,浓度≤10 μM时未观察到明显的细胞毒性(与对照组相比,细胞活力>90%)[2][3] 体内安全性:在db/db小鼠和高脂饮食诱导的肥胖小鼠中,用番泻苷A(剂量高达40 mg/kg/天,持续8周)治疗后,未观察到以下显著变化: - 血清肝功能指标(ALT、AST)和肾功能指标(BUN、肌酐); - 肝脏、肾脏和肠道的组织病理学特征(H&E染色未检测到炎症或组织损伤)。染色); - 体温和一般行为(无嗜睡或腹泻等中毒迹象)[4] |
| 参考文献 |
|
| 其他信息 |
番泻叶(粉末)是一种黄棕色粉末,略带气味和味道。(NTP, 1992)
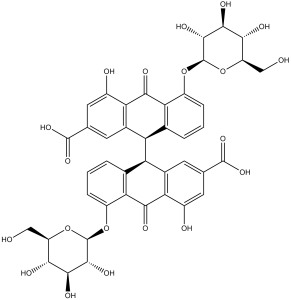
番泻苷A属于番泻苷类化合物,其化学名称为rel-(9R,9'R)-9,9',10,10'-四氢-9,9'-联蒽-2,2'-二羧酸,其4位和4'位被羟基取代,5位和5'位被β-D-吡喃葡萄糖基取代,10位和10'位被氧基取代。9位和9'位的确切立体化学构型尚不清楚,可能是R,R(如图所示)或S,S。它属于番泻苷类化合物,也是一种氧代二羧酸。 番泻叶(决明属植物)是一种常用的草药泻药,无需处方即可购买。番泻叶通常安全且耐受性良好,但若长期高剂量使用,可能引起包括临床上明显的肝损伤在内的不良反应。 据报道,番泻苷A存在于掌叶大黄、唐古特大黄和其他有相关数据的生物体中。 Senokot是由普渡制药公司生产的一种标准化浓缩制剂,含有从番泻叶中提取的蒽醌苷类化合物番泻苷,具有泻药活性。番泻苷作用于肠壁并刺激肠壁内层,从而引起肠道肌肉收缩增强,最终导致剧烈排便。 番泻植物制剂。它们含有番泻苷,番泻苷是一种蒽醌类泻药,被广泛用于多种制剂中作为泻药。 番泻苷A是一种天然从番泻属植物(例如狭叶番泻叶)和大黄属植物(例如大黄)中分离得到的蒽醌糖苷,传统上因其能够刺激肠道蠕动而被用作泻药[1]。 - 作为一种双重HIV-1抑制剂,番泻苷A对不同的HIV-1亚型(X4、R5、双嗜性)具有广谱活性,并显示出克服耐药性的潜力,因为它靶向两种不同的病毒酶(逆转录酶和整合酶),这两种酶不太可能同时产生耐药突变[2][3]。 - 番泻苷A缓解2型糖尿病和肥胖的机制是通过肠道菌群介导的:它选择性地促进有益菌(例如粘液嗜酸阿克曼菌)的生长。它能增强肠道屏障功能,抑制有害细菌(例如,某些促进脂质吸收的厚壁菌门细菌)的增殖,从而减少炎症,提高胰岛素敏感性,并调节脂质代谢[4] |
| 分子式 |
C42H38O20
|
|
|---|---|---|
| 分子量 |
862.74
|
|
| 精确质量 |
862.195
|
|
| CAS号 |
81-27-6
|
|
| 相关CAS号 |
|
|
| PubChem CID |
73111
|
|
| 外观&性状 |
Light yellow to yellow solid powder
|
|
| 密度 |
1.7±0.1 g/cm3
|
|
| 沸点 |
1144.8±65.0 °C at 760 mmHg
|
|
| 熔点 |
200-240ºC
|
|
| 闪点 |
348.6±27.8 °C
|
|
| 蒸汽压 |
0.0±0.3 mmHg at 25°C
|
|
| 折射率 |
1.763
|
|
| LogP |
1.88
|
|
| tPSA |
347.96
|
|
| 氢键供体(HBD)数目 |
12
|
|
| 氢键受体(HBA)数目 |
20
|
|
| 可旋转键数目(RBC) |
9
|
|
| 重原子数目 |
62
|
|
| 分子复杂度/Complexity |
1550
|
|
| 定义原子立体中心数目 |
12
|
|
| SMILES |
C1=CC2=C(C(=C1)O[C@H]3[C@@H]([C@H]([C@@H]([C@H](O3)CO)O)O)O)C(=O)C4=C([C@@H]2[C@@H]5C6=C(C(=CC=C6)O[C@H]7[C@@H]([C@H]([C@@H]([C@H](O7)CO)O)O)O)C(=O)C8=C5C=C(C=C8O)C(=O)O)C=C(C=C4O)C(=O)O
|
|
| InChi Key |
IPQVTOJGNYVQEO-KGFNBKMBSA-N
|
|
| InChi Code |
InChI=1S/C42H38O20/c43-11-23-31(47)35(51)37(53)41(61-23)59-21-5-1-3-15-25(17-7-13(39(55)56)9-19(45)27(17)33(49)29(15)21)26-16-4-2-6-22(60-42-38(54)36(52)32(48)24(12-44)62-42)30(16)34(50)28-18(26)8-14(40(57)58)10-20(28)46/h1-10,23-26,31-32,35-38,41-48,51-54H,11-12H2,(H,55,56)(H,57,58)/t23-,24-,25-,26-,31-,32-,35+,36+,37-,38-,41-,42-/m1/s1
|
|
| 化学名 |
(9R)-9-[(9R)-2-carboxy-4-hydroxy-10-oxo-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-9H-anthracen-9-yl]-4-hydroxy-10-oxo-5-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-9H-anthracene-2-carboxylic acid
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 6.25 mg/mL (7.24 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
例如,若需制备1 mL的工作液,将 100 μL 62.5 mg/mL 澄清 DMSO 储备液加入 900 μL 20% SBE-β-CD 生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (2.41 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清的DMSO储备液加入到400 μL PEG300中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1591 mL | 5.7955 mL | 11.5910 mL | |
| 5 mM | 0.2318 mL | 1.1591 mL | 2.3182 mL | |
| 10 mM | 0.1159 mL | 0.5795 mL | 1.1591 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Link: https://clinicaltrials.gov/ct2/show/NCT04930237
Conditions:OICLink: https://clinicaltrials.gov/ct2/show/NCT06335797
Conditions:ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT06825260
Conditions:Obstructive Defecation Syndrome|Postoperative Constipation
Title:Naloxegol to Prevent Lower Gastrointestinal Paralysis in Critically Ill Adults Administered Opioids
Status:Terminated
updateDate:2023-02-16
Ctid:NCT02977286
Link: https://clinicaltrials.gov/ct2/show/NCT02977286
Conditions:ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT03501498
Conditions:Intestinal TransitLink: https://clinicaltrials.gov/ct2/show/NCT03593252
Conditions:Colostomy|Hirschsprung Disease - Pull Through|Necrotizing Enterocolitis|Inflammatory Bowel Diseases|Meconium Ileus|Bowel Obstruction|Elective SurgeryLink: https://clinicaltrials.gov/ct2/show/NCT01695850
Conditions:Functional Constipation|Gastrointestinal DisordersLink: https://clinicaltrials.gov/ct2/show/NCT02239510
Conditions:Chronic Idiopathic ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT02383758
Conditions:Encopresis|Autistic DisorderLink: https://clinicaltrials.gov/ct2/show/NCT02481219
Conditions:Colorectal Cancer ScreeningLink: https://clinicaltrials.gov/ct2/show/NCT02008864
Conditions:End Stage Renal Disease|PruritusLink: https://clinicaltrials.gov/ct2/show/NCT00994851
Conditions:ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT00662363
Conditions:ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT00931853
Conditions:ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT00571896
Conditions:ConstipationLink: https://clinicaltrials.gov/ct2/show/NCT00275184
Conditions:Small Bowel Disease|Gastrointestinal Hemorrhage|Refractory Anemia