| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
HT-1080 ferroptotic cell death (EC50 = 6 nM)
SRS11-92 targets ferroptosis pathway (inhibits lipid peroxidation-mediated ferroptosis) [1] SRS11-92 targets ferroptosis pathway in Friedreich's Ataxia (FRDA)-related cellular and animal models [2] |
|---|---|
| 体外研究 (In Vitro) |
在 100 nM 浓度下进行测试时,SRS11-92 可充分保护少突胶质细胞 (OL) 免受胱氨酸剥夺的影响。当 frataxin 被击倒时,SRS11-92 可防止原代人成纤维细胞死亡[2]。
在人纤维肉瘤HT-1080细胞和小鼠胚胎成纤维细胞(MEFs)中,SRS11-92 剂量依赖性抑制Erastin(10 μM)或RSL3(0.5 μM)诱导的铁死亡,保护HT-1080细胞免受Erastin诱导死亡的EC50约为0.8 μM [1] SRS11-92 可减少铁死亡诱导细胞中的脂质过氧化水平,通过流式细胞术检测脂质过氧化探针C11-BODIPY的荧光强度降低证实 [1] SRS11-92 不影响星形孢菌素诱导的HT-1080细胞凋亡或曲拉通X-100诱导的坏死,显示出对铁死亡抑制的选择性 [1] 在弗里德赖希共济失调(FRDA)患者来源的成纤维细胞中,SRS11-92(0.5-2 μM)显著减少铁死亡相关细胞死亡,降低线粒体活性氧(ROS)产生(MitoSOX Red染色),并下调脂质过氧化水平 [2] SRS11-92(1 μM)可改善FRDA成纤维细胞的线粒体呼吸功能,通过Seahorse实验检测到氧消耗率(OCR)升高证实 [2] |
| 体内研究 (In Vivo) |
与 caspase-3 抑制剂相比,SRS11-92 可有效保护经柠檬酸铁铵 (FAC) 和谷胱甘肽合成抑制剂 (BSO) 处理的人类和小鼠弗里德赖希共济失调 (FRDA) 细胞模型[2]。
在YG8R小鼠(FRDA疾病模型)中,腹腔注射 SRS11-92(5 mg/kg,每周3次,持续8周)可改善运动功能:与溶媒对照组相比,转棒实验中跌落潜伏期从约120秒延长至210秒,悬线实验中悬挂时间从约30秒延长至65秒 [2] SRS11-92 减少YG8R小鼠脊髓和心脏组织中的脂质过氧化,通过4-羟基壬烯醛(4-HNE)免疫组织化学(IHC)染色显示阳性信号降低证实 [2] SRS11-92 降低YG8R小鼠心脏和脊髓中的铁积累,通过电感耦合等离子体质谱法(ICP-MS)检测证实 [2] SRS11-92 改善YG8R小鼠心脏组织中的线粒体呼吸链复合物(复合物I和II)活性 [2] |
| 酶活实验 |
酿酒酵母活力测定[1]
所有实验均使用携带COQ3基因缺失(COQ3Δ)的酵母菌株。对于点S12稀释试验,从单个菌落中挑选携带coq3Δ突变的细胞,并在YPED培养基(1%Bacto酵母提取物、2%Bacto蛋白胨、2%葡萄糖)+G418中生长过夜。第二天早上,将细胞在YPED+G418中稀释至OD600=0.1-0.5,并使其生长2小时至对数期。然后用无菌水洗涤细胞2次,并在100mM磷酸盐缓冲液(pH 6.2)+0.2%葡萄糖中稀释至OD600=0.2。将0.5 mL等分试样孵育6小时+/-亚麻酸(500μM)和+/-DMSO、trolox、环吡酮胺或ferrostatin-1。6小时后,将培养物标准化至OD为0.2,并在YPED+琼脂平板上进行1:5的点稀释。将板生长72小时,并使用G:Box成像站进行成像。该实验进行了三次,结果相似,并显示了一次实验的代表性数据。 |
| 细胞实验 |
2,2-二苯基-1-苦肼基(DPPH)测定[1]
将稳定的自由基2,2-二苯基-1-苦肼基(DPPH)1溶解在甲醇中,使其最终工作浓度为0.05 mM。首先,通过将3.9mg DPPH溶解在2mL甲醇中制备100倍储备浓度(5mM)。然后,对于25 mL 0.05 mM的最终工作溶液,将S7 250μL的5 mM溶液加入24.75 mL甲醇中。将1 mL DPPH溶液加入到溶解在DMSO中的小体积(<5μL)的每种试验化合物中。每种测试化合物的最终浓度为0.05 mM。将样品倒置几次,并在室温下孵育30分钟。然后将样品等分到白色96孔固体底皿中,并使用TECAN M200平板读数器记录517nm处的吸光度。所有值均归一化为背景值(仅甲醇)。实验重复三次,取平均值。 细胞活力实验:HT-1080细胞或MEFs以5×10³个/孔的密度接种于96孔板,过夜培养。加入系列浓度 SRS11-92(0.01-10 μM)预处理1小时,随后加入Erastin(10 μM)或RSL3(0.5 μM)处理。孵育24-48小时后,使用CCK-8试剂检测细胞活力,根据活力曲线计算EC50值 [1] 脂质过氧化实验:细胞用5 μM C11-BODIPY探针负载30分钟(37°C),经1 μM SRS11-92 预处理1小时后,用Erastin(10 μM)刺激。6小时后,通过流式细胞术检测荧光强度以量化氧化脂质水平 [1] 凋亡/坏死选择性实验:HT-1080细胞经1 μM SRS11-92 预处理1小时,随后暴露于星形孢菌素(1 μM,凋亡诱导剂)或曲拉通X-100(0.1%,坏死诱导剂)24小时。通过Annexin V/PI染色和流式细胞术检测细胞死亡情况 [1] FRDA成纤维细胞实验:FRDA患者来源成纤维细胞接种于6孔板,培养至70%汇合度。加入 SRS11-92(0.5-2 μM)至培养基中,培养48小时。通过MitoSOX Red染色和荧光显微镜检测线粒体ROS;通过C11-BODIPY流式细胞术检测脂质过氧化 [2] 线粒体功能实验:FRDA成纤维细胞接种于Seahorse XF96板,经1 μM SRS11-92 处理24小时后,使用Seahorse分析仪检测氧消耗率(OCR),评估线粒体呼吸功能 [2] |
| 动物实验 |
用于HD检测的250 μm皮质纹状体脑切片取自出生后第10天的CD Sprague-Dawley大鼠幼崽,制备方法如前所述22。将脑切片外植体置于6孔板中进行界面培养,培养基为含15%热灭活马血清、10 mM KCl、10 mM HEPES、100 U/ml青霉素/链霉素、1 mM MEM丙酮酸钠和1 mM L-谷氨酰胺的Neurobasal A培养基,并在32℃、5% CO2的加湿培养箱中培养。使用定制的基因枪装置将含有73个CAG重复序列(“HttN90Q73”)的人类htt外显子1表达构建体(位于gWiz骨架S13中)与YFP表达构建体一起转染至脑切片,以可视化转染的神经元。对照组脑切片用等量的gWiz空载体和YFP进行转染。孵育4天后,根据纹状体内的位置和特征性树突形态识别中型多棘神经元(MSN),并根据以下标准将其评定为健康:整个脑组织中表达明亮且连续的YFP标记,胞体大小正常,且具有>2个长度>2个胞体的初级树突(如前所述)。数据以每个脑切片中每个纹状体区域健康MSN的平均数量表示,统计学显著性采用ANOVA方差分析,并进行Dunnett事后检验(置信水平为0.05)。在制备脑切片时,向培养基中添加Fer-1;阳性对照组脑切片用腺苷受体2A调节剂KW-6002(50 μM)和JNK抑制剂SP600125(30 μM)的组合处理。所有条件下DMSO的最终浓度均为0.1%。[1]
HD Fer-1类似物的脑片实验表明,该类似物能够保护发育中的少突胶质细胞免受胱氨酸剥夺诱导的细胞死亡。采用差速分离法,从P2 Sprague Dawley大鼠幼崽的前脑中制备原代前少突胶质细胞培养物。去除脑膜的前脑组织用含0.01%胰蛋白酶和10µg/ml DNase的Hanks平衡盐溶液进行解离,并用含10%热灭活胎牛血清、100 U/ml青霉素和100 µg/ml链霉素的DMEM培养基进行研磨。将解离的细胞接种到聚-D-赖氨酸包被的75 cm²培养瓶中,并每隔一天更换培养基,持续10-17天。在第10或17天,先在37℃、200 rpm下预摇晃1小时以去除小胶质细胞,然后将培养瓶在37℃下摇晃过夜,使前少突胶质细胞与星形胶质细胞层分离。将细胞悬液通过20 µm滤膜过滤,然后接种到未包被的(细菌培养皿)中,并在培养箱中孵育1小时以去除残留的小胶质细胞/星形胶质细胞。之后,将细胞悬液接种到预先包被聚-D-洛尼氨酸的培养皿中,培养基为DMEM,包含1×ITS、2 mM L-谷氨酰胺、1 mM丙酮酸钠、0.5% FBS、0.05%庆大霉素(Sigma)、10 ng/ml PDGF和10 ng/ml FGF。第二天完全更换培养基,之后每隔一天更换一半培养基。在第8天,用胱氨酸剥夺培养基洗涤细胞两次,然后在含有PDGF和FGF的胱氨酸剥夺培养基(处理培养基)中,用Fer-1及其类似物(DMSO储备液浓度为1 mM)处理细胞24小时。以含有100 µM胱氨酸的处理培养基作为阳性对照;以仅含处理培养基的细胞作为阴性对照。每个孔中的细胞均加入等量的DMSO作为溶剂。24小时后,用Alamar Blue进行荧光检测,方法是将培养基完全更换为含有1× AlamarBlue的Earle's Balance Salt Solution溶液,并在37℃、5% CO2条件下孵育2小时。使用配备Packard 3.0版酶标仪的FluoroCount酶标仪,以530 nm激发波长和590 nm发射波长检测每个孔的荧光强度。 [1] 分离小鼠近端肾小管的研究 肾小管制备:将 8-12 周龄的 C57/BL6 雌性小鼠用异氟烷安乐死。取出肾脏,立即将 S15 肾实质内注射冷的 95% O2/5% CO2 气体溶液,该溶液由以下成分组成(溶液 A):115 mM NaCl、2.1 mM KCl、25 mM NaHCO3、1.2 mM KH2PO4、2.5 mM CaCl2、1.2 mM MgCl2、1.2 mM MgSO4、25 mM 甘露醇、2.5 mg/ml 无脂肪酸牛血清白蛋白、5 mM 葡萄糖、4 mM 乳酸钠、1 mM 丙氨酸和 1 mM 丁酸钠,并添加 1 mg/ml 胶原酶。随后将皮质解剖并切碎,置于冰冷的瓷砖上,然后重悬于额外的溶液 A 中,于 37℃ 消化 8-10 分钟,之后使用自形成 Percoll 梯度离心法富集近端小管,方法与之前描述的兔肾小管富集方法相同。 YG8R 小鼠 FRDA 模型:将 6-8 周龄的 YG8R 小鼠随机分为载体对照组和 SRS11-92 治疗组(每组 n=8)。SRS11-92 溶解于 DMSO:PEG400:PBS (10:40:50, v/v/v) 混合溶液中,浓度为 1 mg/mL。治疗组每周3次腹腔注射5 mg/kg的SRS11-92,持续8周;对照组注射等体积的溶剂[2]。 行为学测试:治疗期间每周进行一次转棒测试,小鼠先以4 rpm的转速训练1分钟,然后在5分钟内从4 rpm加速至40 rpm;记录小鼠跌落潜伏期。治疗结束后进行悬挂测试,将小鼠置于金属丝网上,倒置,测量悬挂时间[2]。 组织采集与分析:治疗8周后,处死小鼠,采集脊髓和心脏组织。组织固定后进行4-HNE免疫组化染色,或匀浆后进行铁含量检测(ICP-MS)和线粒体复合物活性测定[2]。 |
| 参考文献 | |
| 其他信息 |
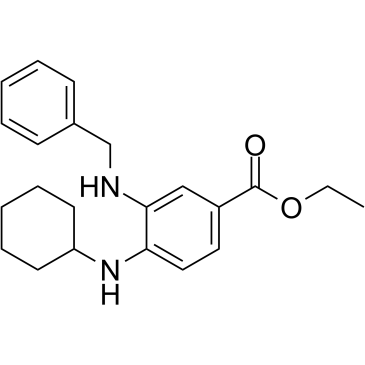
SRS11-92 是一种乙酯,由 3-(苄氨基)-4-(环己氨基)苯甲酸的羧基与乙醇缩合而成。它是 HT-1080 人纤维肉瘤细胞中 erastin 诱导的铁死亡的强效抑制剂(EC50 = 6 nM)。它具有铁死亡抑制剂的作用。它是一种取代苯胺、乙酯、仲胺化合物和二胺。它在功能上与铁死亡抑制剂-1 (ferrostatin-1) 相关。铁死亡抑制剂-1 (Fer-1) 抑制铁死亡,铁死亡是一种受调控的、氧化性的、非凋亡性的细胞死亡形式。我们发现 Fer-1 抑制了亨廷顿病 (HD)、脑室周围白质软化 (PVL) 和肾功能障碍细胞模型中的细胞死亡; Fer-1抑制脂质过氧化,但不影响线粒体活性氧的生成或溶酶体膜的通透性。我们建立了一个机制模型来解释Fer-1的活性,并以此指导开发具有更优特性的铁抑制剂。这些研究表明铁抑制剂具有多种治疗用途,并且脂质过氧化介导了多种疾病表型。[1]弗里德赖希共济失调(FRDA)是一种进行性神经和心脏退行性疾病,其特征是共济失调、感觉丧失和肥厚型心肌病。在大多数情况下,该疾病是由FXN基因两个等位基因第一个内含子中的GAA重复序列扩增引起的,导致编码蛋白弗拉塔辛的表达降低。弗拉塔辛定位于线粒体基质,是铁硫簇生物合成所必需的。弗拉塔辛蛋白表达降低与线粒体功能障碍、线粒体铁积累和氧化应激增加有关。铁死亡是近期发现的一种受调控的、铁依赖性细胞死亡途径,其生化机制与细胞凋亡截然不同。我们评估了FRDA细胞模型中是否存在铁死亡途径激活的证据。我们发现,原代患者来源的成纤维细胞、携带FRDA相关突变的小鼠成纤维细胞以及引入重复序列扩增(敲入/敲出)的小鼠成纤维细胞,与正常对照细胞相比,对已知的铁死亡诱导剂erastin更为敏感。我们还发现,浓度为 500 nM 的铁死亡抑制剂 3-(苄氨基)-4-(环己氨基)苯甲酸乙酯 (SRS11-92) 和 3-氨基-4-(环己氨基)苯甲酸乙酯能够有效保护经柠檬酸铁铵 (FAC) 和谷胱甘肽合成抑制剂 [L-丁硫氨酸 (S,R)-亚砜亚胺 (BSO)] 处理的 FRDA 人和小鼠细胞模型,而 caspase-3 抑制剂则未显示出显著的生物活性。经 FAC 和 BSO 处理的细胞均表现出谷胱甘肽依赖性过氧化物酶活性降低和脂质过氧化作用增强,这两者均为铁死亡的标志。最后,铁死亡抑制剂 SRS11-92 可降低健康人成纤维细胞中 frataxin 敲低引起的细胞死亡。综合来看,这些数据表明铁死亡抑制剂可能具有治疗弗里德赖希共济失调症 (FRDA) 的潜力。[2]
SRS11-92 是铁死亡抑制剂家族的成员,该家族是一类小分子铁死亡抑制剂。[1,2] SRS11-92 通过抑制脂质过氧化(铁死亡的关键步骤)发挥作用,而不干扰细胞凋亡或坏死信号通路。[1] SRS11-92 通过减轻疾病模型中与铁死亡相关的线粒体损伤和铁积累,显示出治疗弗里德赖希共济失调症 (FRDA) 的潜在价值。[2] SRS11-92 对铁死亡抑制具有选择性,在体外实验中未观察到对其他细胞死亡途径的脱靶效应。[1] |
| 分子式 |
C22H28N2O2
|
|---|---|
| 分子量 |
352.5
|
| 精确质量 |
352.215
|
| CAS号 |
1467047-25-1
|
| 相关CAS号 |
1467047-25-1
|
| PubChem CID |
71745064
|
| 外观&性状 |
White to off-white solid
|
| 密度 |
1.2±0.1 g/cm3
|
| 沸点 |
523.7±45.0 °C at 760 mmHg
|
| 闪点 |
270.5±28.7 °C
|
| 蒸汽压 |
0.0±1.4 mmHg at 25°C
|
| 折射率 |
1.619
|
| LogP |
6.37
|
| tPSA |
50.4
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
26
|
| 分子复杂度/Complexity |
416
|
| 定义原子立体中心数目 |
0
|
| SMILES |
C(C)OC(C1C=CC(NC2CCCCC2)=C(C=1)NCC1=CC=CC=C1)=O
|
| InChi Key |
VHQAJFNLPQULSV-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C22H28N2O2/c1-2-26-22(25)18-13-14-20(24-19-11-7-4-8-12-19)21(15-18)23-16-17-9-5-3-6-10-17/h3,5-6,9-10,13-15,19,23-24H,2,4,7-8,11-12,16H2,1H3
|
| 化学名 |
ethyl 3-(benzylamino)-4-(cyclohexylamino)benzoate
|
| 别名 |
SRS1192; SRS11 92; SRS-11-92; 1467047-25-1; ethyl 3-(benzylamino)-4-(cyclohexylamino)benzoate; 4-(cyclohexylamino)-3-[(phenylmethyl)amino]-benzoicacid,ethylester; CHEMBL3633564; SCHEMBL15320680; CHEBI:173095; VHQAJFNLPQULSV-UHFFFAOYSA-N; SRS11-92
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: 70~250 mg/mL (198.6~709.3 mM)
Ethanol: 70 mg/mL (~198.6 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 2.08 mg/mL (5.90 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (5.90 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL 澄清 DMSO 储备液添加到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8369 mL | 14.1844 mL | 28.3688 mL | |
| 5 mM | 0.5674 mL | 2.8369 mL | 5.6738 mL | |
| 10 mM | 0.2837 mL | 1.4184 mL | 2.8369 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
|