| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
SIRT1 (EC50 = 0.10 μM); SIRT1 (EC1.5 = 0.16 μM5); SIRT2 (EC1.5 = 37 μM)
Because SRT 1720 inhibits histone acetyltransferase p300, it effectively lowers p53 acetylation in cells even when SIRT1 is not present[3]. SRT1720 is a selective activator of SIRT1 with an EC50 of 0.16 μM, which is more than 230 times lower than SIRT2 and SIRT3. Here, researchers examined the anti-multiple myeloma (MM) activity of a novel oral agent, SRT1720, which targets SIRT1. Treatment of MM cells with SRT1720 inhibited growth and induced apoptosis in MM cells resistant to conventional and bortezomib therapies without significantly affecting the viability of normal cells. Mechanistic studies showed that anti-MM activity of SRT1720 is associated with: 1) activation of caspase-8, caspase-9, caspase-3, poly (ADP) ribose polymerase; 2) increase in reactive oxygen species; 2) induction of phosphorylated ataxia telangiectasia mutated/checkpoint kinase 2 signalling; 3) decrease in vascular endothelial growth factor-induced migration of MM cells and associated angiogenesis; and 4) inhibition of nuclear factor-κB. Blockade of ATM attenuated SRT1720-induced MM cell death. [2] To determine whether SRT1460 and SRT1720 bind and activate the enzyme at the same molecular site as resveratrol, an isobologram analysis was performed. A concentration matrix of two compounds, resveratrol versus SRT1720 and SRT1720 versus SRT1460, was examined to determine whether the combination was antagonistic, additive or synergistic. In both cases the compound combination resulted in additivity consistent with the hypothesis that a single allosteric site exists on the SIRT1—substrate complex to which structurally diverse compounds can bind (Fig. 2c). [1] |
|---|---|
| 体外研究 (In Vitro) |
由于 SRT 1720 抑制组蛋白乙酰转移酶 p300,因此即使不存在 SIRT1,它也能有效降低细胞中的 p53 乙酰化[3]。
SRT1720 是 SIRT1 的选择性激活剂,EC50 为 0.16 μM,比 SIRT2 和 SIRT3 低 230 倍以上。 在这里,研究人员检测了一种新型口服药物SRT1720的抗多发性骨髓瘤(MM)活性,该药物靶向SIRT1。SRT1720可抑制MM细胞的生长,诱导对常规和硼替佐米治疗有抗性的MM细胞凋亡,但不显著影响正常细胞的活力。机制研究表明,SRT1720的抗mm活性与:1)激活caspase-8、caspase-9、caspase-3、聚(ADP)核糖聚合酶有关;2)活性氧增加;2)诱导磷酸化的共济失调毛细血管扩张突变/检查点激酶2信号传导;3)血管内皮生长因子诱导的MM细胞迁移和相关血管生成减少;4)抑制核因子-κB。阻断ATM可减弱srt1720诱导的MM细胞死亡。[2] 为了确定SRT1460和SRT1720是否与白藜芦醇在相同的分子位点结合并激活酶,我们进行了等距图分析。研究了白藜芦醇对SRT1720和SRT1720对SRT1460两种化合物的浓度矩阵,以确定这两种化合物的组合是拮抗、相加还是协同作用。在这两种情况下,化合物组合产生的可加性与假设一致,即sirt1 -底物复合物上存在单一的变构位点,结构上不同的化合物可以与之结合(图2c)。[1] SRT1720 在生化测定中激活人 SIRT1 酶,与白藜芦醇(EC₁.₅ = 46.2 µM,最大激活 = 201%)相比,显示出显著更高的效力(EC₁.₅ = 0.16 µM)和最大激活能力(781%)。 [1] 在基于细胞的去乙酰化测定(In Cell Western)中,SRT1720(约 0.10 µM,近似 EC₅₀)能降低 U2OS 细胞中 p53(一种 SIRT1 底物)的乙酰化状态。该效应可被选择性 SIRT1 抑制剂阻断,证实了其 SIRT1 依赖性。 [1] 机制上,SRT1720 能降低 SIRT1 对其乙酰化肽底物的 Km,而不影响其对 NAD+ 的 Km 或 Vmax,表明它增强了底物结合/催化效率。 [1] 等效线图分析表明,SRT1720 与白藜芦醇的组合产生相加的激活效应,这与它们结合于 SIRT1-底物复合物上相同的变构位点一致。 [1] SIRT1 的截短突变实验表明,氨基酸 183-225(位于核心催化结构域的 N 端)对于化合物诱导的激活至关重要,从而定义了变构结合位点。 [1] |
| 体内研究 (In Vivo) |
与罗格列酮治疗类似,SRT 1720(10、30、100 mg/kg,口服)可显着降低高胰岛素血症,并在 4 周后很大程度上恢复高胰岛素水平。使用 SRT 1720 治疗的 Lepob/ob 小鼠的空腹血糖水平显着降低 [1],几乎正常。通过 SIRT1 的下游靶标(例如 PGC1α 和 FOXO1),SRT 1720 可以保护小鼠免受饮食引起的肥胖的有害影响,并与脂肪酸代谢适应性和氧化代谢相关 [3]。在 WT 小鼠中,SRT 1720(50-100 mg/kg,口服)可降低动脉氧饱和度,并减弱肺气肿发展过程中弹性蛋白酶诱导的气隙扩大和肺功能退化[4]。
在 DIO 小鼠中,SRT1720 模拟了热量限制后观察到的几种效果,包括改善胰岛素敏感性、标准化葡萄糖和胰岛素水平以及增加线粒体容量。此外,在饮食诱导的肥胖和遗传性肥胖小鼠中,SRT1720 可以改善胰岛素敏感性、降低血浆葡萄糖并增加线粒体容量。因此,SRT1720是一种有前途的新型治疗剂,用于治疗2型糖尿病等衰老疾病。与糖耐量改善一致,SRT1720治疗的fa/fa大鼠中维持血糖正常所需的葡萄糖输注率增加了约35%,总葡萄糖处理率增加了约20%。 SRT1720 还可以预防多发性骨髓瘤肿瘤的生长。 SRT1720 增加硼替佐米或地塞米松的细胞毒活性。 通过基因过表达和选择性药理激活剂SRT1720激活SIRT1,可以减轻应激诱导的细胞过早衰老,并保护小鼠免受香烟烟雾和弹性蛋白酶诱导的肺气肿。气道上皮Sirt1消融,但髓细胞不消融,气道扩大加重,肺功能受损,运动耐受性降低。这些影响是由于SIRT1能够使FOXO3转录因子去乙酰化,因为FOXO3缺乏降低了SRT1720对细胞衰老和肺气肿变化的保护作用。[4] 然而,SIRT1是否是治疗胆汁淤积症的合适治疗靶点尚不清楚。在本研究中,检测了SRT1720 (SIRT1的特异性激活剂)对17α-炔雌醇(EE)诱导的小鼠胆汁淤积的保护作用。数据表明,SRT1720显著阻止ee诱导的血清总胆汁酸(TBA)、总胆红素(TBIL)、γ-谷氨酰转肽酶(γ-GGT)和碱性磷酸酶(ALP)水平的变化。血红素和伊红(H&E)染色显示,SRT1720还能减轻ee诱导的肝脏病理损伤。SRT1720通过HNF1α/FXR信号通路,上调肝外排转运蛋白(Bsep和Mrp2)和肝摄取转运蛋白(Ntcp和Oatp1b2)的表达,对ee诱导的肝损伤有保护作用。SRT1720显著抑制EE诱导的TNF-α和IL-6水平。这些结果表明,SRT1720对ee诱导的小鼠胆汁淤积性肝损伤具有剂量依赖性的保护作用,其机制可能与激活HNF1α/FXR信号通路和抗炎机制有关。[5] 在饮食诱导的肥胖(DIO)小鼠中,口服给予 SRT1720(100 mg/kg/天)10 周可显著降低进食后血浆葡萄糖水平,改善葡萄糖耐量(腹腔葡萄糖耐量试验),降低高胰岛素血症,并增强胰岛素敏感性(胰岛素耐量试验),效果与罗格列酮相当。 [1] SRT1720 处理(100 mg/kg/天,持续 10 周)使 DIO 小鼠腓肠肌中的线粒体容量(柠檬酸合酶活性)增加 15%。 [1] 在高脂饮食的遗传性肥胖糖尿病 Lep^ob/ob 小鼠中,SRT1720(100 mg/kg/天)治疗 1 周后,可显著降低空腹血糖至接近正常水平。 [1] 在 Zucker fa/fa 大鼠(遗传性肥胖、胰岛素抵抗模型)中,SRT1720 治疗(100 mg/kg/天,持续 4 周)可显著降低进食后血糖,改善口服葡萄糖耐量(降低葡萄糖和胰岛素 AUC),并通过高胰岛素-正常血糖钳夹实验证实了其增强全身胰岛素敏感性的作用。具体而言,它增加了葡萄糖输注率(约35%)、总葡萄糖清除率(约20%)、胰岛素刺激的葡萄糖清除率(主要是骨骼肌,约55%),并抑制了肝脏葡萄糖输出。它还降低了血浆游离脂肪酸水平,表明脂肪组织胰岛素敏感性得到改善。 [1] 在 Zucker fa/fa 大鼠中,SRT1720 治疗(100 mg/kg/天,持续 3 周)可减少肝脏糖异生,这通过丙酮酸耐量试验中葡萄糖反应减弱得到证明。 [1] 在所研究的所有肥胖啮齿动物模型中,SRT1720 均未影响体重。 [1] 在正常饮食喂养的非胰岛素抵抗小鼠中,SRT1720 未降低空腹血糖或胰岛素水平,表明其在非胰岛素抵抗状态下不会诱发低血糖。 [1] |
| 酶活实验 |
SIRT1荧光偏振测定和HTS [1]
在SIRT1 FP检测中,使用从p53序列中提取的20个氨基酸肽(AcGlu-Glu-Lys(生物素)- gly - gln - ser - thr - ser - ser - his - ser -Lys(Ac)- nle - ser - thr - glu - gly -Lys(MR121或Tamra)- gluu - gluu - nh2)来监测SIRT1的活性。该肽n端与生物素连接,c端用荧光标记修饰。监测酶活性的反应是一个偶联酶试验,第一个反应是由SIRT1催化的去乙酰化反应,第二个反应是由胰蛋白酶在新暴露的赖氨酸残基上切割。停止反应,加入链霉亲和素,以强调底物和产物之间的质量差异。总共筛选了29万种化合物,确认了127种。FP检测的灵敏度允许鉴定出SIRT1低水平激活(20 μM下激活≥17%)的化合物,产生代表不同结构类别的多种激活剂。荧光极化反应条件为:0.5 μM肽底物,150 μM βNAD+, 0-10 nM SIRT1, 25 mM Tris-acetate pH 8, 137 mM Na-Ac, 2.7 mM K-Ac, 1 mM Mg-Ac, 0.05% Tween-20, 0.1% Pluronic F127, 10 mM CaCl2, 5 mM DTT, 0.025% BSA, 0.15 mM烟酰胺。37℃孵育,加入烟酰胺停止反应,加入胰蛋白酶裂解去乙酰化底物。在1 μM链亲和素存在下,37℃孵育反应。在激发(650 nm)和发射(680 nm)波长处测定荧光偏振。 作用机制研究[1] 测试化合物对乙酰化肽底物SIRT1酶Km的影响采用上述SIRT1质谱法检测。采用无细胞质谱法测定了9种化合物浓度(100、33、11、3.7、1.2、0.41、0.14、0.046和0.015 μM)和DMSO载体单独存在下肽底物SIRT1酶的Km。为了确定Km,对每种化合物浓度和对照进行了12种乙酰化肽底物浓度(50、25、12.5、6.25、3.12、1.56、0.78、0.39、0.19、0.098、0.049和0.024 μM)下的线性去乙酰化速率测定。SIRT1酶、2 mM NAD+和0-50 μM乙酰化肽底物与0-100 μM化合物在25°C下孵育。在0、3、6、9、12、15、20和25分钟,用10%甲酸和50 mM烟酰胺停止反应,并通过质谱测定底物到产物的转化。 等温滴定量热法(ITC) [1] 人SIRT1-E5c蛋白(41 μM;ITC使用质谱肽底物(1.0 mM)和SRT1460 (0.84 mM)原液。缓冲条件为50 mM Tris-HCl (pH 8.0)、137 mM NaCl、2.7 mM KCl、1 mM MgCl2、2 mM TCEP和5%甘油。滴定在26℃的VP-ITC上进行。选择SRT1460进行这些研究是因为它在缓冲液中可溶到实验所需的毫摩尔浓度。 等温图研究/Isobologram studies[1] 白藜芦醇与SRT1720和SRT1720与SRT1460联合使用的效果使用上述SIRT1质谱法测定。建立了两种化合物的浓度基质,并对SIRT1酶进行了检测。在基质中存在的每种组合下,测定乙酰化肽底物转化为去乙酰化肽产物的百分比。所得的等线图用于评价组合的效果。在分析中,产生相同效应水平的剂量组合的笛卡尔坐标图是等线图的基础。如果两种化合物具有可变效价,则选择恒定的相对效价(R) -即达到相同倍活性所需的化合物量(例如白藜芦醇vs SRT1720 EC1.25和SRT1720 vs SRT1460 EC2.5) -用于X和Y截距进行等温图分析。对应于各自EC值的两种化合物的浓度用作X和Y轴上的截距。利用这两个截距,在两点之间画出一条叫做可加性线的理论线。将两种化合物作为剂量对混合在一个矩阵中,产生相同的效应水平(EC值),通过对数滴定得到的实验数据绘制在等温线图上。通过对两种药物剂量组合的可加性曲线和可加性曲线进行统计比较,可以看出一种效应是否具有可加性。落在可加性线以下和以上的点进行回归分析。高于可加性线的实验数据被解释为拮抗,低于可加性线的实验数据被解释为协同,落在可加性线上的实验数据被认为是可加性。 p53去乙酰化试验[1] 将人骨肉瘤细胞(U-2 OS)以每孔1.5 X 104个细胞的比例在96孔板中进行镀膜。24小时后,将测试化合物和对照(均为100% DMSO)添加到细胞板中。为了证明该检测结果对sirt1的依赖性,重复板组与测试化合物和sirt1特异性小分子抑制剂(6-氯-2,3,4,9-四氢-1- h -咔唑-1-carboxamide)共同处理。化合物添加后,每孔中加入阿霉素(终浓度1 μg/ml)诱导p53表达和乙酰化。p53诱导后,将细胞固定,然后用PBS-0.1% Triton-X-100渗透。在PBS-0.1% TWEEN 20(阻断溶液)中加入5%牛血清白蛋白阻断非特异性蛋白结合。一抗抗p53-acetyl-lysine-382(兔多克隆)和抗β - tubulin(小鼠单克隆)分别在Block Solution中稀释1:400和1:1000,加入孔中在40℃孵育过夜,然后在PBS-0.1% TWEEN 20 (Wash Buffer)中洗涤。二抗IR800CW山羊抗兔IgG和Alexa-Fluor 680山羊抗小鼠IgG在Block Solution中稀释,加入孔中室温孵育1小时。在Wash Buffer中再次洗涤5次。然后用Li-Cor奥德赛红外扫描仪扫描底片。使用制造商的软件提取数据。Ac-Lys382-p53和β -微管蛋白的信号均使用仅含二抗孵育的孔进行背景校正。然后将每个孔的ac - lys382信号归一化为相应的β -微管蛋白信号,以校正细胞数量的差异。然后将这些值归一化到载体中,以生成每口井的乙酰化p53 %值。 使用高通量荧光偏振测定法进行初步筛选。该测定使用了一种基于 p53 的生物素化和荧光标记的肽。去乙酰化反应与暴露的赖氨酸残基处的胰蛋白酶切割步骤偶联。 [1] 使用质谱测定法进行所有构效关系研究和效力确定(EC₁.₅)。该测定使用与荧光测定相同的肽底物。 [1] 为了确定激活机制,使用质谱测定法检测了 SRT1720 对 SIRT1 动力学参数的影响。评估了该化合物对乙酰化肽底物和 NAD+ 的 Km 以及 Vmax 的影响。 [1] 通过创建两种化合物(例如白藜芦醇和 SRT1720)的浓度矩阵,并使用质谱测定法测试它们对 SIRT1 的作用,进行等效线图分析,以确定其效应是相加、协同还是拮抗。 [1] |
| 细胞实验 |
免疫组织化学(IHC)原位检测细胞凋亡[2]
从小鼠(对照)和SRT1720 -处理小鼠中切除肿瘤并保存在10%福尔马林中。如前所述,肿瘤中的凋亡细胞通过免疫组化染色检测caspase-3激活(Chauhan等,2010)。 体外迁移和毛细血管样管结构形成测定[2] 如前所述,Transwell插入测定法用于测量迁移(Podar等,2001)。通过Matrigel毛细管样管结构形成试验评估体外血管生成(Chauhan et al, 2010)。为了进行内皮管形成实验,从克隆公司获得人血管内皮细胞(HUVECs),并将其保存在含有5%胎牛血清的内皮细胞生长培养基2 (EGM2 MV singlequotes)中。3代后,用台盼蓝排斥法测定HUVEC细胞活力,SRT1720处理HUVEC细胞死亡率<5%。 细胞活力和凋亡测定[2] 采用先前描述的3-(4,5 -二甲基噻唑-2-基)- 2,5 -二苯基溴化四唑(MTT)比色法评估细胞活力(Hideshima等人,2000年)。凋亡测定采用Annexin V-FITC/碘化丙啶(PI)细胞凋亡检测试剂盒,按照制造商的说明进行定量,然后在FACS Calibur上进行分析。 Western blotting和蛋白定量[2] 采用caspase-3、caspase-7、caspase-8、caspase-9、多聚(ADP)核糖聚合酶(PARP)、Ace-Lys 382 p53、磷酸化-失调性毛细血管扩张突变(pATM)、磷酸化-检查点激酶2 (pCHK2)、磷酸化- i - κ b ser32/36和GAPDH抗体进行免疫印迹分析。然后通过增强化学发光形成印迹。使用AlphaImager EC凝胶记录系统获取蛋白质条带的密度,并使用斑点密度分析工具对条带进行分析。 在 U2OS 细胞中进行基于细胞的去乙酰化测定(In Cell Western, ICW),以监测 p53 在赖氨酸 382 位的乙酰化状态。用化合物处理细胞,然后固定并使用针对乙酰化 p53(K382)的多克隆抗体进行染色。β-微管蛋白用作标准化对照。通过同时使用选择性 SIRT1 抑制剂处理,证实了 SRT1720 的去乙酰化作用是 SIRT1 依赖性的。 [1] |
| 动物实验 |
溶于 20% PEG400/0.5% Tween80/79.5% 去离子水中;200 mg/kg/天;口服给药 Chase-SCID 小鼠(MM.1S 细胞) SRT1720 的药代动力学[1] 将溶于载体(2% HPMC + 0.2% DOSS)的 SRT1720 通过灌胃法给予 C57BL/6 雄性小鼠(18-22 克;每个剂量组每个时间点 3 只小鼠),剂量见 doi: 10.1038/nature06261 补充信息 www.nature.com/nature 4。所有体内研究中,SRT1720 均以盐酸盐形式给药。小鼠采用二氧化碳窒息法处死,分别于给药后 5、30、120 和 360 分钟采集血液。采集血液后,将血浆送至查尔斯河实验室 (CRL) 进行药物浓度分析。为测定口服生物利用度,将溶于载体(10% 乙醇/40% 聚乙二醇/50% 水)的 SRT1720 以指定剂量经尾静脉注射至 C57BL/6 雄性小鼠(18-22 克;每个剂量组每个时间点 3 只小鼠)。分别于给药后 5、30、120 和 360 分钟采集血液,并按上述方法分析药物浓度。此外,将溶于载体(2% 羟丙甲纤维素 + 0.2% 多西环己醇)的 SRT1720 以 100 mg/kg 的剂量灌胃给予 Sprague-Dawley 雄性大鼠(250 克;每个剂量组 3 只大鼠)。分别于给药后 0.033、0.083、0.25、0.5、1、2、4、6、8 和 24 小时采集血液样本,并分析药物浓度。为测定口服生物利用度,将 SRT1720 以 10 mg/kg 的剂量经尾静脉注射给药。SRT1720 用于静脉注射研究时,其溶于 10% 乙醇/ 40% 聚乙二醇/ 50% 水的混合溶液中。 饮食诱导肥胖模型 [1] 9 周龄 C57BL/6 雄性小鼠喂食高脂饮食(60% 的能量来自脂肪),直至其平均体重达到约 40 g。然后将小鼠分为实验组(每组 6-10 只)。 SRT1460 (100 mg/kg)、SRT1720 (100 mg/kg)、SRT501 (500 mg/kg) 和罗格列酮 (5 mg/kg) 每日一次通过灌胃给药。所用溶剂为 2% HPMC + 0.2% DOSS。每周测量两次小鼠体重。分别在给药后第 2、4、6、8 和 10 周进行餐后血糖测定;治疗 5 周后,对各组所有小鼠进行腹腔葡萄糖耐量试验 (IPGTT);治疗 10 周后,进行胰岛素耐量试验 (ITT)。柠檬酸合酶活性测定[1] 5 采用Srere和Moyes29,30描述的方法,在治疗11周后测定骨骼肌(腓肠肌)和白色脂肪组织(附睾)中的柠檬酸合酶(CS)活性。选择每组(DIO Vehicle组和DIO SRT1720组)中空腹血糖水平最具代表性的5只小鼠进行分析。 ob/ob模型[1] 6周龄时接收nob/ob小鼠和一只杂合子ob/+小鼠。在研究开始前至少一周,小鼠喂食高脂饮食(60%的能量来自脂肪),并在整个研究期间保持高脂饮食。适应一周后,对所有小鼠进行称重并测量血糖。本研究中使用的ob/ob小鼠平均体重约为40-45克。所有小鼠均按体重和血糖水平进行分选,然后分组。动物每日一次通过灌胃给予SRT1720(100 mg/kg)、SRT501(1000 mg/kg)或载体(2% HPMC + 0.2% DOSS)。血糖和胰岛素的测定方法如上所述。 Zucker fa/fa模型[1] 6周龄雄性肥胖(fa/fa)Zucker (ZF)大鼠在光照(12:12光照:黑暗)和温度控制条件下单独饲养。7周龄时,动物随机分配接受SIRT1激活剂(SRT1720)或载体(即2% HPMC + 0.2% DOSS)。药物通过灌胃法每日给药(下午3点至5点之间),持续4周,动物可自由摄取食物和水。在开始给药前一晚,动物禁食12小时。次日早晨(第1天),测量血糖浓度,并从尾静脉采集血样至肝素化毛细管中。该血样以13,000 rpm离心5分钟,并将血浆储存于-80℃以备分析。在给药的前3周(即第8、15和22天),重复上述步骤。此外,在第22天下午,从尾静脉采集餐后血糖进行测量。人浆细胞瘤异种移植模型[2] 异种移植肿瘤模型的构建方法如前所述(LeBlanc等,2002)。该动物模型在广泛验证新型抗多发性骨髓瘤疗法硼替佐米和来那度胺方面发挥了极其重要的作用,最终促成了这两种疗法进入临床试验,并获得美国食品药品监督管理局批准用于治疗多发性骨髓瘤。将含有 6.0 × 10⁶ 个 MM.1S 细胞的 100 μl 无血清 RPMI-1640 培养基皮下接种到 Fox Chase-SCID 小鼠(每组 6 只)体内。 MM 细胞注射后约三周,当肿瘤可测量(~100 mm3)时,小鼠分别口服给予载体(20% PEG400/0.5% Tween80/79.5% 去离子水)或 SRT1720(200 mg/kg),连续四周,每周五天。使用免疫组织化学 (IHC) 原位检测细胞凋亡[2] 切除载体(对照)和 SRT1720 处理组小鼠的肿瘤,并保存在 10% 福尔马林中。肿瘤中的凋亡细胞通过 caspase-3 激活的 IHC 染色进行鉴定,如前所述(Chauhan 等,2010)。 在药代动力学研究中,将 SRT1720 以指定剂量通过灌胃或静脉注射给予 C57BL/6 雄性小鼠。并随时间推移测量血浆浓度。[1] 在饮食诱导肥胖 (DIO) 小鼠的疗效研究中,将雄性 C57BL/6 小鼠喂食高脂饮食,直至其平均体重达到约 40 g。SRT1720(100 mg/kg 体重)或载体每日一次通过灌胃给药,持续长达 10 周。[1] 在 Lep^ob/ob 小鼠的研究中,小鼠在研究开始前和研究期间至少 1 周喂食高脂饮食。每日一次通过灌胃给予SRT1720(100 mg/kg)或载体。[1] 在Zucker fa/fa大鼠的研究中,遗传性肥胖雄性大鼠每日一次通过灌胃给予SRT1720(100 mg/kg)或载体,持续4周。监测体重和食物摄入量。[1] 在治疗开始后的指定时间点进行葡萄糖耐量试验(腹腔或口服)、胰岛素耐量试验、高胰岛素-正常血糖钳夹试验和丙酮酸耐量试验,以评估代谢参数。[1] |
| 药代性质 (ADME/PK) |
SRT1720 的药代动力学特征(图 3a)适用于小鼠(生物利用度 = 50%,末端半衰期约为 5 小时,曲线下面积 (AUC) = 7,892 ng h⁻¹ ml⁻¹)和大鼠(生物利用度 = 25%,末端半衰期约为 8.4 小时,AUC = 3,714 ng h⁻¹ ml⁻¹)的体内评价。SRT501 是白藜芦醇的改良版本,具有更高的生物利用度(生物利用度为 11%,末端半衰期约为 1 小时,AUC 为 10,524 ng h⁻¹ ml⁻¹),也已在遗传性肥胖小鼠 (Lepob/ob) 和饮食诱导肥胖 (DIO) 小鼠中进行了研究。 [1]
在小鼠中,口服给药后,SRT1720的生物利用度约为50%,末端半衰期(t₁/₂)约为5小时,曲线下面积(AUC)为7,892 ng·h/mL。[1] 在大鼠中,口服给药后,SRT1720的生物利用度约为25%,末端半衰期(t₁/₂)约为8.4小时,AUC为3,714 ng·h/mL。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
一项被引用的研究报告称,早期研究中使用的SRT1720剂量具有毒性,而较低剂量则会导致体重增加。但具体的毒性作用或参数(例如,LD50、器官毒性)并未详细说明。[3]
另一项被引用的研究涉及对肥胖小鼠进行终生100 mg/kg SRT1720治疗,结果显示小鼠存活率有所提高,但未提及毒性,这表明该剂量在该模型中具有耐受性。[3] |
| 参考文献 | |
| 其他信息 |
SRT-1720 是 NAD(+) 依赖性组蛋白去乙酰化酶 SIRT1 的激活剂。
热量限制可延长寿命,并产生有利于治疗衰老相关疾病(例如 2 型糖尿病)的代谢特征。SIRT1 是一种 NAD+ 依赖性去乙酰化酶,是热量限制下游通路的主要调节因子,这些通路对葡萄糖稳态和胰岛素敏感性具有有益作用。白藜芦醇是一种多酚类 SIRT1 激活剂,它模拟了热量限制在低等生物体中的抗衰老作用,并且在喂食高脂饮食的小鼠中,白藜芦醇可改善胰岛素抵抗、增加线粒体含量并延长寿命。本文描述了我们鉴定和表征的 SIRT1 小分子激活剂,这些激活剂在结构上与白藜芦醇无关,但其效力比白藜芦醇高 1000 倍。这些化合物与SIRT1酶-肽底物复合物结合,结合位点位于催化结构域氨基末端的变构位点,从而降低乙酰化底物的米氏常数。在饮食诱导的肥胖小鼠和遗传性肥胖小鼠中,这些化合物可改善胰岛素敏感性,降低血糖水平,并提高线粒体功能。在Zucker fa/fa大鼠中,高胰岛素-正常血糖钳夹试验表明,SIRT1激活剂可改善全身葡萄糖稳态,并提高脂肪组织、骨骼肌和肝脏的胰岛素敏感性。因此,SIRT1激活是一种有前景的治疗衰老相关疾病(如2型糖尿病)的新方法。[1] SIRT1属于沉默信息调节因子2 (Sir2) 蛋白家族,是一种NAD(+)依赖性III类组蛋白去乙酰化酶。本文研究了一种新型口服药物SRT1720的抗多发性骨髓瘤(MM)活性,该药物靶向SIRT1。 SRT1720 处理多发性骨髓瘤 (MM) 细胞可抑制对传统疗法和硼替佐米耐药的 MM 细胞的生长并诱导其凋亡,且对正常细胞的活力无显著影响。机制研究表明,SRT1720 的抗 MM 活性与以下机制相关:(i) 激活 caspase-8、caspase-9、caspase-3 和聚 (ADP) 核糖聚合酶;(ii) 增加活性氧;(iii) 诱导磷酸化共济失调毛细血管扩张突变蛋白/检查点激酶 2 (ATM/CK2) 信号通路;(iv) 降低血管内皮生长因子 (VEGF) 诱导的 MM 细胞迁移和相关血管生成;以及 (v) 抑制核因子-κB (NF-κB)。阻断 ATM 可减弱 SRT1720 诱导的 MM 细胞死亡。在动物肿瘤模型研究中,SRT1720 可抑制 MM 肿瘤的生长。最后,SRT1720增强了硼替佐米或地塞米松的细胞毒活性。我们的临床前研究为针对多发性骨髓瘤(MM)中SIRT1靶点的新型疗法提供了理论依据。[2] 尽管人类寿命的延长体现了现代医学的成功,但罹患多种疾病的风险也随着年龄的增长呈指数级增长。已知热量限制可以延缓衰老,减缓功能衰退,并延缓大多数生物体疾病的发生。研究表明,sirtuins(SIRT1-SIRT7)是衰老过程中热量限制关键效应的介质。两种不相关的分子——白藜芦醇和SRT1720——已被证明在某些情况下可以增强SIRT1活性,它们是哺乳动物对抗代谢应激的优秀保护剂,这使得SIRT1成为一个潜在的、有吸引力的治疗靶点。本综述涵盖了sirtuins作为新型药理靶点的现状和争议,重点关注SIRT1。[3] 慢性阻塞性肺疾病/肺气肿(COPD/肺气肿)的特征是慢性炎症和肺部过早衰老。抗衰老蛋白sirtuin 1 (SIRT1) 是一种NAD+依赖性蛋白/组蛋白去乙酰化酶,在COPD患者的肺部表达降低。然而,肺部过早衰老的分子机制,以及SIRT1是否能抵抗细胞衰老和肺气肿的各种病理生理改变,仍然未知。本文中,我们发现COPD患者肺部细胞衰老增加。 SIRT1的激活,无论是通过基因过表达还是选择性药理学激活剂SRT1720,都能减轻应激诱导的细胞过早衰老,并保护小鼠免受香烟烟雾和弹性蛋白酶诱导的肺气肿。在气道上皮细胞(而非髓系细胞)中敲除Sirt1,会加剧肺泡扩大、损害肺功能并降低运动耐量。这些影响是由于SIRT1能够使FOXO3转录因子去乙酰化,因为Foxo3缺陷会减弱SRT1720对细胞衰老和肺气肿改变的保护作用。使用NF-κB/IKK2抑制剂抑制肺部炎症对肺气肿没有益处。因此,SIRT1通过FOXO3介导的细胞衰老减少来保护肺气肿,而与炎症无关。激活SIRT1可能是治疗慢性阻塞性肺疾病/肺气肿的一种有前景的策略。[4] Sirtuin 1 (SIRT1) 是哺乳动物中最保守的NAD+依赖性蛋白脱乙酰酶,属于沉默信息调节因子2 (Sir2) 蛋白家族(也称为Sirtuins)。在肝脏中,肝脏SIRT1通过调节法尼醇X受体 (FXR) 的表达来调控胆汁酸代谢。FXR是参与胆汁酸代谢调控的最重要的核受体之一。SIRT1在多个层面上调控FXR的表达,包括直接对该转录因子进行脱乙酰化以及通过肝细胞核因子1α (HNF1α) 进行转录调控。因此,肝脏SIRT1是HNF1α/FXR信号通路和肝脏胆汁酸代谢的重要调节因子。然而,SIRT1是否是治疗胆汁淤积的合适靶点尚不清楚。本研究探讨了SIRT1特异性激活剂SRT1720对17α-炔雌醇(EE)诱导的小鼠胆汁淤积的保护作用。结果表明,SRT1720显著抑制了EE诱导的血清总胆汁酸(TBA)、总胆红素(TBIL)、γ-谷氨酰转肽酶(γ-GGT)和碱性磷酸酶(ALP)水平的变化。苏木精-伊红(H&E)染色结果显示,SRT1720还减轻了EE诱导的肝脏病理损伤。SRT1720通过HNF1α/FXR信号通路发挥保护作用,上调肝脏外排转运蛋白(Bsep和Mrp2)和肝脏摄取转运蛋白(Ntcp和Oatp1b2)的表达,从而减轻EE诱导的肝损伤。此外,SRT1720显著抑制了EE诱导的TNF-α和IL-6水平。这些结果表明,SRT1720对EE诱导的小鼠胆汁淤积性肝损伤具有剂量依赖性的保护作用,其作用机制与HNF1α/FXR信号通路的激活和抗炎机制有关。[5] SRT1720在结构上与天然SIRT1激活剂白藜芦醇无关,但其体外活性比白藜芦醇高约1000倍。[1] 它与SIRT1酶-乙酰化肽底物复合物上的变构位点结合,该位点位于SIRT1的N端区域(氨基酸183-225)。这种结合降低了乙酰化底物的Km值,从而促进了更高效的催化构象。 [1] 该化合物模拟了热量限制的多种有益代谢效应,包括提高胰岛素敏感性、使血糖和胰岛素水平正常化以及增强线粒体功能。[1] SRT1720被认为是一种有前景的治疗与年龄相关的疾病(例如2型糖尿病)的新疗法。[1] |
| 分子式 |
C25H23N7OS
|
|
|---|---|---|
| 分子量 |
469.56
|
|
| 精确质量 |
469.168
|
|
| 元素分析 |
C, 63.95; H, 4.94; N, 20.88; O, 3.41; S, 6.83
|
|
| CAS号 |
925434-55-5
|
|
| 相关CAS号 |
SRT 1720 monohydrochloride;2060259-60-9;SRT 1720 dihydrochloride;2468639-77-0;SRT 1720 hydrochloride;1001645-58-4
|
|
| PubChem CID |
24180125
|
|
| 外观&性状 |
Light yellow to yellow solid powder
|
|
| 密度 |
1.46
|
|
| 熔点 |
221ºC
|
|
| LogP |
4.003
|
|
| tPSA |
115.69
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
7
|
|
| 可旋转键数目(RBC) |
5
|
|
| 重原子数目 |
34
|
|
| 分子复杂度/Complexity |
707
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
C1CN(CCN1)CC2=CSC3=NC(=CN23)C4=CC=CC=C4NC(=O)C5=NC6=CC=CC=C6N=C5
|
|
| InChi Key |
IASPBORHOMBZMY-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C25H23N7OS/c33-24(22-13-27-20-7-3-4-8-21(20)28-22)29-19-6-2-1-5-18(19)23-15-32-17(16-34-25(32)30-23)14-31-11-9-26-10-12-31/h1-8,13,15-16,26H,9-12,14H2,(H,29,33)
|
|
| 化学名 |
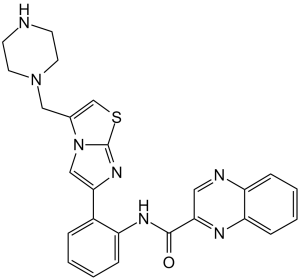
N-[2-[3-(piperazin-1-ylmethyl)imidazo[2,1-b][1,3]thiazol-6-yl]phenyl]quinoxaline-2-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (4.43 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (4.43 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: 25 mg/mL (53.24 mM) in 50% PEG300 50% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1297 mL | 10.6483 mL | 21.2965 mL | |
| 5 mM | 0.4259 mL | 2.1297 mL | 4.2593 mL | |
| 10 mM | 0.2130 mL | 1.0648 mL | 2.1297 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。