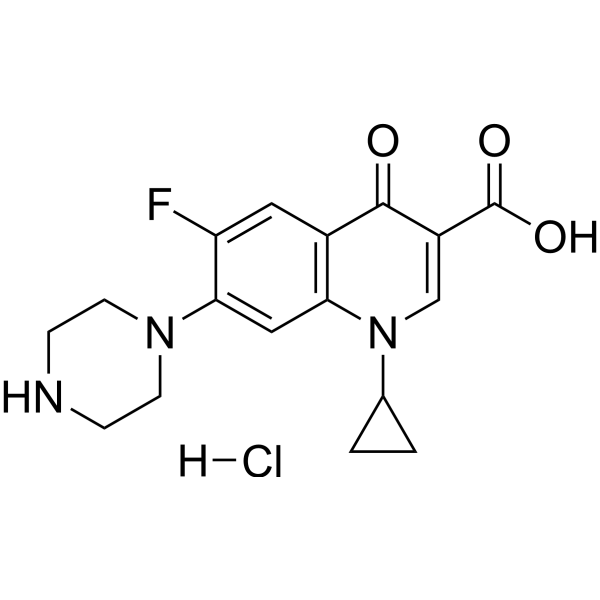
Ciprofloxacin (Bay-09867) monoHCl 是一种口服生物可利用的拓扑异构酶 IV 抑制剂。 Ciprofloxacin monoHCl 会诱导线粒体 DNA 和核 DNA 损伤,并导致线粒体功能障碍和活性氧产生。 Ciprofloxacin monoHCl 具有抗增殖活性并导致细胞凋亡。 Ciprofloxacin monoHCl 是一种氟喹诺酮类抗生素,具有有效的抗菌作用。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Clinical Trial Testing Whether Targeted Antibiotic Prophylaxis Can Reduce Infections After Cystectomy Compared to Empiric Prophylaxis
CTID: NCT06709196
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-11-29
Tobramycin Inhalation Solution for Pseudomonas Aeruginosa Eradication in Bronchiectasis
CTID: NCT06093191
Phase: Phase 4 Status: Recruiting
Date: 2024-10-16
Modulation of the Gut Microbiome With Pembrolizumab Following Chemotherapy in Resectable Pancreatic Cancer
CTID: NCT05462496
Phase: Phase 2 Status: Recruiting
Date: 2024-10-15
CAT BITE Antibiotic Prophylaxis for the Hand/Forearm (CATBITE)
CTID: NCT05846399
Phase: Phase 4 Status: Recruiting
Date: 2024-10-01
Antibiotic Prophylaxis for Neurogenic Bladder Botox
CTID: NCT04791579
Phase: Phase 2 Status: Not yet recruiting
Date: 2024-09-19
View More
Ciprofloxacin Versus Levofloxacin in Stem Cell Transplant
CTID: NCT03850379
Phase: Phase 2 Status: Completed
Date: 2024-09-19
Efficacy of Immunization With 4C-MenB in Preventing Experimental Urethral Infection With Neisseria Gonorrhoeae
CTID: NCT05294588
Phase: Phase 2 Status: Recruiting
Date: 2024-09-19
LiveSpo Navax® Supports the Treatment of Acute Rhinosinusitis and Otitis Media
CTID: NCT05804123
Phase: N/A Status: Completed
Date: 2024-08-22
Comparing the Effect of Different Intracanal Dressing on Failed Root Canal Treated Cases on Periapical Healing
CTID: NCT06342830
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-08-20
Pharmacokinetics and Safety of Commonly Used Drugs in Lactating Women and Breastfed Infants
CTID: NCT03511118
Phase: Status: Recruiting
Date: 2024-07-24
Ciprofloxacin Versus an Aminoglycoside Followed by Ciprofloxacin for Bubonic Plague
CTID: NCT04110340
Phase: Phase 3 Status: Recruiting
Date: 2024-06-17
Shigella Sonnei 53G Human Infection Study in Kenyan Adults
CTID: NCT05959616
Phase: Phase 1 Status: Not yet recruiting
Date: 2024-05-23
Velocity 2: An Anthrax Vaccine and Antibiotics Clinical Study
CTID: NCT04067011
Phase: Phase 2 Status: Completed
Date: 2024-03-21
Ciprofloxacin BioThrax Co-Administration Study
CTID: NCT01753115
Phase: Phase 2 Status: Completed
Date: 2024-03-18
Early Versus Late Stopping of Antibiotics in Children With Cancer and High-risk Febrile Neutropenia
CTID: NCT04948463
Phase: Phase 4 Status: Recruiting
Date: 2024-03-15
CiproPAL (Ciprofloxacin Prophylaxis in Acute Leukaemia)
CTID: NCT04678869
Phase: N/A Status: Recruiting
Date: 2024-03-08
Single Versus Combined Antibiotic Therapy for Bacterial Peritonitis in CAPD Patients
CTID: NCT01785641
Phase: N/A Status: Completed
Date: 2024-02-21
Antibiotic Prophylaxis for Bladder Botox
CTID: NCT04444440
Phase: Phase 4 Status: Recruiting
Date: 2024-02-08
Collaborative Urological Prosthetics Investigation Directive Research Group
CTID: NCT05100654
PhaseEarly Phase 1 Status: Active, not recruiting
Date: 2024-02-07
Fundamental Modification of the Gut Microbiota in the Treatment of Refractory Crohn's Disease
CTID: NCT02765256
Phase: Phase 2 Status: Completed
Date: 2024-02-06
Rifaximin as a Prophylaxis of Spontaneous Bacterial Peritonitis in Comparison With Ciprofloxacin
CTID: NCT06234046
Phase: Phase 3 Status: Completed
Date: 2024-01-31
A Randomized Placebo- and Active Comparator-controlled Study to Evaluate the Photosafety of SAR441566
CTID: NCT05844735
Phase: Phase 1 Status: Completed
Date: 2024-01-18
Antibiotic Treatment foLlowing Surgical drAinage of Perianal abScess; the ATLAS Trial
CTID: NCT05385887
Phase: N/A Status: Recruiting
Date: 2023-11-13
Combination Study of Antibiotics With Enzalutamide (PROMIZE)
CTID: NCT06126731
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2023-11-13
Pharmacokinetics of Ciprofloxacin in Pediatric Patients
CTID: NCT02598362
Phase: Phase 4 Status: Completed
Date: 2023-09-05
Evaluation of Adherent Invasive E. Coli Eradication in Adult Crohn Disease
CTID: NCT02620007
Phase: Phase 2 Status: Terminated
Date: 2023-08-23
Evaluation of Photosafety of BI 730357 Compared to Placebo and the Known Photosensitizing Agent Ciprofloxacin
CTID: NCT04147260
Phase: Phase 1 Status: Completed
Date: 2023-07-17
Ciprofloxacin Utilization and Adverse Events Occurrence in Community Pharmacy Setting
CTID: NCT05916105
Phase: Status: Completed
Date: 2023-06-23
Ciprofloxacin Versus Azithromycin for Children Hospitalised With Dysentery
CTID: NCT03854929
Phase: Phase 4 Status: Completed
Date: 2023-06-07
A Phase 1, Open-Label Study to Evaluate the Effect of a Low-Fat Meal and Multiple Doses of Ciprofloxacin on the Pharmacokinetics of Vorasidenib in Healthy Subjects
CTID: NCT05843708
Phase: Phase 1 Status: Recruiting
Date: 2023-06-05
Targeted AntiBiotics for Chronic Pulmonary Diseases
CTID: NCT03262142
Phase: Phase 4 Status: Terminated
Date: 2023-05-25
A Multiple-Dose PK Study to Evaluate the Comparative Bioavailability of PrimeC Tablets to Ciprofloxacin Tablets Co-administered With Celecoxib Capsules, in Healthy Adult Subject
CTID: NCT05436678
Phase: Phase 1 Status: Completed
Date: 2023-02-08
A Pilot Study to Evaluate the PK Profile of PrimeC-ER Tablets in Healthy Adult Subjects
CTID: NCT05232461
Phase: Phase 1 Status: Completed
Date: 2022-10-25
Bioequivalence Study of Ciprofloxacin in Healthy Adult Subjects Under Fasting Condition
CTID: NCT05532267
Phase: Phase 1 Status: Completed
Date: 2022-09-08
Antimicrobial Treatment in Patients With Ventilator-associated Tracheobronchitis
CTID: NCT03012360
Phase: Phase 4 Status: Unknown status
Date: 2022-08-17
Clinical Trial of Oral Ciprofloxacin and Etoposide in Subjects With Resistant Acute Myeloid Leukemia (AML)(UF-AML-CE-101)
CTID: NCT02773732
Phase: Phase 1/Phase 2 Status: Terminated
Date: 2022-08-05
Efficacy Of Doxycycline & Versus Rifampin In Treatment Of Rhinoscleroma
CTID: NCT05431673
Phase: Phase 1 Status: Unknown status
Date: 2022-07-07
Modified Surface of PLGA Nanoparticles in Smart Hydrogel
CTID: NCT05442736
PhaseEarly Phase 1 Status: Completed
Date: 2022-07-05
Oral Antimicrobial Treatment vs. Outpatient Parenteral for Infective Endocarditis
CTID: NCT05398679
Phase: Phase 4 Status: Not yet recruiting
Date: 2022-06-01
S. Aureus Decolonization in HPN Patients.
CTID: NCT03173053
Phase: N/A Status: Terminated
Date: 2022-05-31
Culture-guided Antimicrobial Prophylaxis in Men Undergoing Prostate Biopsy.
CTID: NCT03228108
Phase: Phase 4 Status: Completed
Date: 2022-05-10
Amoxicillin-clavulanate Alone or in Combination With Ciprofloxacin in Low-Risk Febrile Neutropenic Adult Patients: A Prospective, Double-blind, Randomized, Non-Inferiority Multicenter, Phase III Clinical Trial.
CTID: NCT04698057
Phase: Phase 3 Status: Withdrawn
Date: 2022-03-31
SpeeDx Ciprofloxacin gyrA Assay for N. Gonorrhoeae Gonococcal Infection
CTID: NCT05286931
Phase: N/A Status: Recruiting
Date: 2022-03-18
Antibiotic Prophylaxis for HDR Brachytherapy in the Treatment of Prostate Cancer
CTID: NCT03862170
Phase: Phase 3 Status: Suspended
Date: 2022-03-17
Role of Prophylactic Postoperative Antibiotics in HoLEP
CTID: NCT05274672
Phase: Phase 4 Status: Unknown status
Date: 2022-03-11
Short-course Methenamine Hippurate for Prevention of Post-operative UTI
CTID: NCT02358993
Phase: N/A Status: Completed
Date: 2022-02-25
A Study to Evaluate the Efficacy and Safety of Vedolizumab in the Treatment of Chronic Pouchitis
CTID: NCT02790138
Phase: Phase 4 Status: Completed
Date: 2022-02-24
Single Dose Ciprofloxacin in the Treatment of Childhood Cholera:Randomized Controlled Clinical Trial
CTID: NCT00142272
Phase: Phase 3 Status: Completed
Date: 2022-02-11
Pilot Study of Fundamental Modification of the Gut Microbiota in the Treatment of Refractory Crohn's Disease
CTID: NCT03476317
Phase: Phase 2 Status: Completed
Date: 2022-02-09
Experimental Human Infection With Neisseria Gonorrhoeae (LptA Trial)
CTID: NCT04870138
Phase: Phase 1 Status: Completed
Date: 2021-12-02
Antibiotic Profile of Pathogenic Bacteria Isolated in Public Hospitals in Northern Jordan
CTID: NCT05106803
Phase: Status: Completed
Date: 2021-11-04
Antibiotic Prophylaxis Before Shock Wave Lithotripsy
CTID: NCT03692715
Phase: Phase 4 Status: Recruiting
Date: 2021-10-18
Evaluation of Innovative Tools in Development of Antibiotics
CTID: NCT03177720
Phase: Phase 1 Status: Completed
Date: 2021-09-01
Safety and Immunogenicity of Peru-15-pCTB in Healthy Adult Subjects
CTID: NCT00654108
Phase: Phase 1 Status: Completed
Date: 2021-07-29
Longitudinal Study of the Human Intestinal Microbiome
CTID: NCT00832286
Phase: Phase 1 Status: Completed
Date: 2021-07-29
Clinical Efficacy of Crano-cure inTreatment of Urinary Tract Infection
CTID: NCT04575493
Phase: N/A Status: Completed
Date: 2021-06-24
Experimental Human Infection With Neisseria Gonorrhoeae
CTID: NCT03840811
Phase: Phase 1 Status: Completed
Date: 2021-03-23
Targeted Retreatment of COPD Exacerbations
CTID: NCT02300220
Phase: Phase 3 Status: Completed
Date: 2021-03-08
Efficacy of Ciprofloxacin Therapy in Avoidance of Sepsis in Patient Undergoing Percutanous Nephrolithotomy
CTID: NCT04374188
Phase: N/A Status: Unknown status
Date: 2021-03-03
Oral Sulopenem-etzadroxil/Probenecid Versus Ciprofloxacin for Uncomplicated Urinary Tract Infection in Adult Women
CTID: NCT03354598
Phase: Phase 3 Status: Completed
Date: 2021-01-12
Sulopenem Followed by Sulopenem-etzadroxil/Probenecid vs Ertapenem Followed by Cipro for Complicated UTI in Adults
CTID: NCT03357614
Phase: Phase 3 Status: Completed
Date: 2020-12-29
Sulopenem Versus Ertapenem for Complicated Intra-abdominal Infection (cIAI)
CTID: NCT03358576
Phase: Phase 3 Status: Completed
Date: 2020-12-01
A Phase III Study of Ciprofloxacin Plus Fluocinolone in Acute Otitis Externa (AOE)
CTID: NCT04636957
Phase: Phase 3 Status: Unknown status
Date: 2020-11-19
Impact of Formulation on Ciprofloxacin Oral Absorption
CTID: NCT00992329
Phase: Phase 1 Status: Completed
Date: 2020-11-06
Dose Ranging Study of OTO-201 in AOMT
CTID: NCT02719158
Phase: Phase 2 Status: Completed
Date: 2020-10-19
Phase 3 Study of OTO-201 in Acute Otitis Externa
CTID: NCT02801370
Phase: Phase 3 Status: Completed
Date: 2020-10-19
Chronic Prostatitis Collaborative Research Network Clinical Trial- Ciprofloxacin and Tamsulosin
CTID: NCT04552431
Phase: Phase 2 Status: Completed
Date: 2020-09-18
Clinical Validation of a Molecular Test for Ciprofloxacin-Susceptibility in Neisseria Gonorrhoeae
CTID: NCT02961751
Phase: N/A Status: Completed
Date: 2020-08-25
A Pilot Study of Ciprofloxacin Plus Gemcitabine and Nab-Paclitaxel Chemotherapy in Patients With Metastatic Pancreatic Ductal Adenocarcinoma.
CTID: NCT04523987
Phase: Phase 1 Status: Unknown status
Date: 2020-08-24
Granulocyte-colony Stimulating Factors or Antibiotics for Primary Prophylaxis for Febrile Neutropenia
CTID: NCT02816112
Phase: Phase 4 Status: Completed
Date: 2020-05-27
Comparison of Efficacy of Cefotaxime, Ceftriaxone, and Ciprofloxacin for the Treatment of SBP in Patients With LC
CTID: NCT01265173
Phase: Phase 4 Status: Completed
Date: 2020-04-06
Antibiotic Prophylaxis in Laparoscopic Cholecystectomy
CTID: NCT01888822
Phase: Phase 4 Status: Terminated
Date: 2020-03-25
Antibiotic Prophylaxis With Routine Ureteral Stent Removal
CTID: NCT02944825
Phase: N/A Status: Recruiting
Date: 2020-03-24
Special Drug Use Investigation of Ciproxan Injection in Pediatrics
CTID: NCT02555059
Phase: Status: Completed
Date: 2020-02-28
Antibiotic/COPD in Acute Exacerbation of Chronic Obstructive Pulmonary Disease (COPD) Requiring Mechanical Ventilation
CTID: NCT00791505
Phase: Phase 3 Status: Completed
Date: 2020-02-24
Parenteral Antibiotics Compared to Combination of Oral and Parenteral Antibiotics in Colorectal Surgery Prophylaxis
CTID: NCT02505581
Phase: Phase 4 Status: Completed
Date: 2020-01-14
Ciprofloxacin for Prevention of BK Infection
CTID: NCT01789203
Phase: Phase 4 Status: Completed
Date: 2019-11-13
Antibiotics and Hydroxychloroquine in Crohn's
CTID: NCT01783106
Phase: Phase 2 Status: Completed
Date: 2019-10-31
Efficacy of Ciprofloxacin for the Treatment of Uncomplicated Urinary Tract Infection (uUTI)
CTID: NCT03366207
Phase: Phase 4 Status: Completed
Date: 2019-09-06
Ciprofloxacin for the Prevention of Meningococcal Meningitis 2018
CTID: NCT03431675
Phase: Phase 4 Status: Withdrawn
Date: 2019-08-14
Ciprofloxacin Plus Metronidazole Vs Cefixime Plus Metronidazole Therapy for the Treatment of Liver Abscess
CTID: NCT03969758
Phase: Phase 3 Status: Unknown status
Date: 2019-07-22
BK Virus in Salivary Gland Disease: Treating the Potential Etiologic Agent
CTID: NCT02068846
Phase: Phase 3 Status: Completed
Date: 2019-07-11
Antibiotic Prophylaxis for Transrectal Prostate Biopsy
CTID: NCT01659866
Phase: Phase 4 Status: Completed
Date: 2019-06-25
Safety, Tolerability, and Efficacy of MK-7655 (Relebactam) + Imipenem/Cilastatin Versus Imipenem/Cilastatin Alone for Treating Complicated Urinary Tract Infection (cUTI) (MK-7655-003)
CTID: NCT01505634
Phase: Phase 2 Status: Completed
Date: 2019-05-24
PEriToneal Catheter Versus Repeated Paracentesis for Ascites in Cirrhosis
CTID: NCT03027635
Phase: N/A Status: Terminated
Date: 2019-05-20
Antibiotic Prophylaxis for Transrectal Prostate Biopsy-Ciprofloxacin vs. Trimethoprim/Sulfamethoxazole
CTID: NCT02734732
Phase: Phase 2 Status: Unknown status
Date: 2019-05-15
Safety, Tolerability and PK of Intravenous (IV) ETI-204 Alone and in Presence of Ciprofloxacin in Adult Volunteers
CTID: NCT01952444
Phase: Phase 1 Status: Completed
Date: 2019-04-16
Antibiotic Prophylaxis for Endoscopic Ultrasound Guided Fine Needle Aspiration of Pancreatic Cystic Lesions
CTID: NCT02261896
Phase: Phase 4 Status: Completed
Date: 2019-04-12
Rifamycin SV-MMX® Tablets Versus Ciprofloxacin Capsules in Acute Traveller's Diarrhoea
CTID: NCT01208922
Phase: Phase 3 Status: Completed
Date: 2019-02-27
Otiprio Versus Ciprodex Tympanostomy Tube Outcomes
CTID: NCT03347461
Phase: Phase 4 Status: Withdrawn
Date: 2018-09-14
Antibiotics for Klebsiella Liver Abscess Study
CTID: NCT01723150
Phase: Phase 4 Status: Completed
Date: 2018-08-27
Ciprofloxacin Compared to Placebo in Diagnosing Prostate Cancer in Patients Undergoing Prostate Biopsy
CTID: NCT02252978
Phase: Phase 2 Status: Withdrawn
Date: 2018-07-05
Comparing Ciprofloxacin (CPFX) With Cefepime (CFPM) in Febrile Neutropenic Patients With Hematologic Diseases
CTID: NCT00137787
Phase: Phase 3 Status: Completed
Date: 2018-06-26
Efficiency of Triple Antibiotic Paste, Ciprofloxacin/Propolis, Ciprofloxacin/Metronidazole, Propolis/Metronidazole Combinations on Revascularization Process of Immature Necrotic Maxillary Incisors of Patients 8-18 Years Old.
CTID: NCT03533231
Phase: Phase 4 Status: Completed
Date: 2018-05-30
Comparison Between Rifampicin and Gemifloxacin and Ciprofloxacin in Treatment of Rhinoscleroma
CTID: NCT03326050
PhaseEarly Phase 1 Status: Unknown status
Date: 2018-02-13
Ciprofloxacin for the Prevention of Meningococcal Meningitis
CTID: NCT02724046
Phase: Phase 4 Status: Completed
Date: 2018-02-08
Efficacy Study of Prophylaxis With Fosfomycin Versus Ciprofloxacin Prior Prostate Biopsy
CTID: NCT01803191
Phase: Phase 4 Status: Completed
Date: 2017-12-13
Study In Healthy Subjects To Evaluate The Photo-Irritant Potential Of Eltrombopag
CTID: NCT00688272
Phase: Phase 1 Status: Completed
Date: 2017-11-17
Bacterial Resistance in Patients Receiving Post-Intravitreal Injection Antibiotics
CTID: NCT02223338
Phase: N/A Status: Completed
Date: 2017-11-07
REaCT Integrated Consent Model to Compare Two Standard of Care Regimens
CTID: NCT02173262
Phase: Phase 4 Status: Completed
Date: 2017-11-06
Phase III Comparison of Adjuvant Chemotherapy W/High-Dose Cyclophosphamide Plus Doxorubicin (AC) vs Sequential Doxorubicin Fol by Cyclophosphamide (A-C) in High Risk Breast Cancer Patients With 0-3 Positive Nodes (Intergroup, CALGB 9394)
CTID: NCT00590785
Phase: Phase 3 Status: Completed
Date: 2017-08-25
Rifaximin for the Secondary Prevention of Spontaneous Bacterial Peritonitis Recurrence in Cirrhotic Patients
CTID: NCT02011841
Phase: Phase 3 Status: Withdrawn
Date: 2017-04-25
Antibiotic Prophylaxis for Urinary Catheter Removal After Radical Prostatectomy
CTID: NCT02247960
Phase: N/A Status: Terminated
Date: 2017-04-17
A Trial Assessing Peri-procedure Chemoprophylaxis During Transrectal Prostate Needle Biopsy
CTID: NCT02423759
Phase: Phase 4 Status: Completed
Date: 2017-04-04
Evaluate the Effects of Itraconazole and Ciprofloxacin on Single-Dose PK of Pracinostat in Healthy Nonsmoking Subjects
CTID: NCT02118909
Phase: Phase 1 Status: Completed
Date: 2017-02-23
Randomised Open-label Multicenter Study Evaluating Ciprofloxacin in Severe Alcoholic Hepatitis
CTID: NCT02326103
PhaseEarly Phase 1 Status: Completed
Date: 2017-01-19
Norfloxacin Versus Ciprofloxacin for Spontaneous Bacterial Peritonitis (SBP) Prevention
CTID: NCT01542801
Phase: Phase 4 Status: Completed
Date: 2016-12-28
Pharmacokinetics and Pharmacodynamics and Selected Antibiotics During Pregnancy
CTID: NCT00214331
Phase: Status: Completed
Date: 2016-10-27
Cerebral Antibiotics Distribution After Acute Brain Injury
CTID: NCT01059890
Phase: Phase 1 Status: Completed
Date: 2016-10-11
Study of Cabazitaxel Combined With Prednisone and Prophylaxis of Neutropenia Complications in the Treatment of Patients With Metastatic Castration-resistant Prostate Cancer
CTID: NCT01649635
Phase: Phase 4 Status: Completed
Date: 2016-07-06
A Pilot Study on the Use of Prophylactic Antibiotics for EUS-guided Pancreatic Cyst Aspiration
CTID: NCT01929460
Phase: N/A Status: Completed
Date: 2016-02-15
Efficacy of Antimicrobial Prophylaxis for Shock Wave Lithotripsy (SWL) on Reducing Urinary Tract Infection (UTI)
CTID: NCT01873690
Phase: Phase 3 Status: Terminated
Date: 2016-02-08
A Phase Ib Study of Belinostat With RDHAP Chemotherapy (Dexamethasone, Cytarabine, Cisplatinum) in Adults With Relapsed or Refractory
A Multicentre Randomised Control Trial Assessing the Efficacy of Antimicrobial Prophylaxis for Extracorporeal Shock Wave Lithotripsy on reducing Urinary Tract Infections
CTID: null
Phase: Phase 4 Status: Completed
Date: 2021-11-26
A multicenter randomized trial of fosfomycin versus ciprofloxacin for febrile neutropenia in hematologic patients: efficacy and microbiologic safety.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2021-11-11
Prospective randomized controlled study of two antibiotic treatment times (3 versus 6 weeks) of diabetic foot osteomyelitis
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2021-07-22
Pathophysiological mechanisms in the development of anal fistula. Oral antibiotics after anal abscess drainage to diminish perianal Fistula Formation: a multicenter, randomized, observer-blind, placebo-controlled clinical trial.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2021-05-04
Pivmecillinam with Amoxicillin/clavulanic acid for Step Down Oral Therapy in Febrile UTIs Caused by ESBL-producing Enterobacterales.
CTID: null
Phase: Phase 4 Status: Trial now transitioned
Date: 2021-02-16
Amoxicillin-clavulanate alone or in combination with Ciprofloxacin in Low-Risk Febrile Neutropenic adult Patients:
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2020-09-15
Use of repeated Multiple Breath Washout to detect and treat pulmonary exacerbation in children with Cystic Fibrosis, a multicenter randomized controlled study.
CTID: null
Phase: Phase 4 Status: Ongoing, Prematurely Ended
Date: 2020-04-08
ANTIBIOTIC THERAPY IN RESPIRATORY TRACT INFECTIONS: AIR.
CTID: null
Phase: Phase 4 Status: Trial now transitioned
Date: 2020-04-03
Revised dosing recommendations of ciprofloxacin for patients with impaired renal function: a bioequivalence study.
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2020-03-10
ABSORB 2:An exploratie study determining the oral antibiotic drug absorption in patients with short bowel syndrome.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2020-02-25
Short course antibiotic treatment of Gram-negative bacteremia: A multicenter, randomized, non-blinded, non-inferiority interventional study
CTID: null
Phase: Phase 4 Status: Trial now transitioned
Date: 2020-02-11
Exposure to orally administered antibiotics during the initial phase of infection in non-critically ill, febrile patients
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-07-03
A Phase 2/3 Open-label, Randomized, Active-controlled Clinical Study to Evaluate the Safety, Tolerability, Efficacy and Pharmacokinetics of MK-7655A in Pediatric Participants From Birth to Less Than 18 Years of Age With Confirmed or Suspected Gram-negative Bacterial Infection
CTID: null
Phase: Phase 2, Phase 3 Status: Restarted, Completed
Date: 2019-06-26
Impact on the intestinal microbiota during antibiotic treatment
CTID: null
Phase: Phase 2 Status: Completed
Date: 2019-05-17
Shortened Antibiotic Treatment in Community-Acquired Pneumonia: A Nationwide Danish Randomized Controlled Trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-04-29
Effects of antibiotics on micobiota, pulmonary immune response and incidence of ventilator-associated infections
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2019-01-14
A Phase III, Randomized, Double-Blind, Multicenter, Comparative Study to Determine the Efficacy and Safety of Cefepime-Tazobactam vs. Meropenem followed by Optional Oral Therapy in the Treatment of Complicated Urinary Tract Infection or Acute Pyelonephritis in Adults
CTID: null
Phase: Phase 3 Status: Temporarily Halted, Prematurely Ended, Completed
Date: 2018-10-17
Pharmacokinetics of different antibiotics in cerebrospinal fluid in children with malignant brain tumors – a pilot study
CTID: null
Phase: Phase 1 Status: Ongoing
Date: 2018-09-27
Optimized treatment for uncomplicated acute appendicitis - active observation with or without antibiotic treatment. A phase IV consecutive clinical treatment trial.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2018-07-30
Immediate oral, immediate topical or delayed oral antibiotics for acute otitis media with discharge (the Runny Ear STudy: REST)
CTID: null
Phase: Phase 4 Status: Completed
Date: 2018-05-23
The effect of rectal swab culture-guided antimicrobial prophylaxis in men undergoing prostate biopsy on infectious complications and cost of care: A randomized controlled trial in the Netherlands.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2018-02-06
A multicenter, open-label, randomized, active-controlled, parallel group, pivotal study to investigate the efficacy, safety and tolerability, and pharmacokinetics of murepavadin combined with one anti-pseudomonal antibiotic versus two anti-pseudomonal antibiotics in adult subjects with ventilator-associated bacterial pneumonia suspected or confirmed to be due to Pseudomonas aeruginosa.
CTID: null
Phase: Phase 3 Status: Ongoing, Prematurely Ended
Date: 2018-01-29
Right Dose, Right Now: Randomized Clinical Trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2018-01-11
Acute uncomplicated diverticulitis: prospective, controlled, randomized, multicenter clinical trial of non-antibiotic outpatient treatment (TASDA trial).
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2017-12-14
Fosfomycin vs Ciprofloxacin for transrectal biopsy - a randomized
CTID: null
Phase: Phase 2 Status: Completed
Date: 2017-05-16
Target-ABC (Targeted AntiBiotics for Chronic pulmonary disease):
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2017-01-14
Placebo-kontrollierte, multizentrische, randomisierte, doppelblinde Phase III-Studie zur Verbesserung der gastrointestinalen Verträglichkeit einer per-oralen Antibiotikatherapie durch add-on-Gabe von Lactobacillus rhamnosus GG (InfectoDiarrstop® LGG® Mono Kapseln) bezogen auf die Häufigkeit einer AAD bei Kindern unter 2 Jahren
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2016-11-17
Combined Effect of CFTR Modifiers and Intensive Antibiotic Treatment
CTID: null
Phase: Phase 4 Status: Completed
Date: 2016-08-15
Antibiotic prophylaxis before extracorporeal shock wave lithotripsy (APPEAL)
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-06-28
Individualized dosing of aminoglycosides, quinolones and glycopeptide antibiotics in (morbidly) obese patients
CTID: null
Phase: Phase 4 Status: Completed
Date: 2016-05-18
Randomized, multicenter, open, phase III, controlled clinical trial, to demonstrate the non-inferiority of reduced antibiotic treatment directed against the treatment of a broad spectrum betalactam antipseudomonal in treating patients with bacteremia spectrum Enterobacteriaceae
CTID: null
Phase: Phase 3 Status: Completed
Date: 2016-03-18
Effect of Intravenous Administration of C1-inhibitor on Inflammation and Coagulation after Bronchial Instillation of House Dust Mite Allergen and Lipopolysaccharide in Allergic Asthma Patients
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2015-03-16
Pharmacokinetics of ciprofloxacine in pediatric patients, a pilot study – SAFE PEDRUG.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-03-10
Antibiotic prophylaxis oral vs parenteral + parenteral in colonic surgery: a prospective, randomized, multicenter clinical trial.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-01-19
Development of a tool for adapting dosage of fluoroquinolones by using a population pharmacokinetic model
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2014-12-12
Multicenter, randomized and double-blinded clinical trial on the use of antibiotic prophylaxis for EUS guided FNA of pancreatic cystic lesions
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-08-18
MEROPENEM and CIPROFLOXACIN DOSING IN THE CRITICALLY ILL PATIENT WITH SEPTIC SHOCK –
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-07-25
Randomized, double-blind, placebo-controlled, multicenter study comparing Ciprofloxacin DPI 32.5 mg BID intermittently administered for 28 days on / 28 days off or 14 days on / 14 days off versus placebo to evaluate the time to first pulmonary exacerbation and frequency of exacerbations in subjects with non–cystic f
e.querySelector("font strong").innerText = 'View More'
} else if(up_display === 'none' || up_display === '') {
icon_angle_down.style.display = 'none';