| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Benzodiazepine (BZD) receptors [1]
- Benzodiazepine (BZD) receptors (partial agonist-like binding in BALB/c mice, no affinity data specified) [2] - Benzodiazepine (BZD) receptors (mediating allopregnanolone's anxiolytic action blockade) [3] - Benzodiazepine (BZD) receptors and non-NMDA receptors (interactions with non-NMDA antagonists in seizure models) [4] - Benzodiazepine (BZD) receptors (blocking anxiety in ethanol-withdrawn rats) [5] |
|---|---|
| 体内研究 (In Vivo) |
氟马西尼通过与中枢苯二氮卓受体相互作用,中和或逆转苯二氮卓激动剂和反向激动剂的行为、神经和电生理作用。在某些情况下,肝性脑病可以受益于氟马西尼,但是在进行周密计划的临床试验之前,肝性脑病必须作为探索性适应症进行治疗。虽然氟马西尼单独或与其他药物联合使用可以恢复苯二氮卓类药物引起的睡意,但如果怀疑存在周期性抗抑郁药物毒性,则不应服用氟马西尼[1]。在凸起十字迷宫和光/暗测试中,氟马西尼 (1 mg/kg) 显着降低 BALB/c 小鼠的焦虑[2]。在大鼠中,氟马西尼 (10 mg/kg) 可以成功阻止异孕酮的减少[3]。在小鼠中,地西泮的抗惊厥作用和副作用可以被氟马西尼 (5-20 mg/kg) 抵消,但不能被 GYKI 52466 抵消。在 MES 模型中,氟马西尼在一定程度上减弱了 NBQX 的抗惊厥活性,但在 PTZ 试验中则不然。 [4]。减少开臂时间和开臂进入百分比是长期乙醇治疗的结果,因为氟马西尼(3.0 mg/kg)抑制戒断改变[5]。
Flumazenil(氟马西尼)在多种动物模型和人体研究中,可逆转苯二氮䓬类药物的镇静、催眠、抗焦虑、抗惊厥和肌肉松弛作用[1] - Flumazenil在BALB/c小鼠中诱导部分激动剂样效应(降低自发活动、增加Vogel冲突试验中的受惩罚反应),但在C57BL/6小鼠中无此效应[2] - Flumazenil(10 mg/kg,腹腔注射)可完全阻断孕烷醇酮(5 mg/kg,腹腔注射)在小鼠高架十字迷宫试验中表现出的抗焦虑作用[3] - Flumazenil(10 mg/kg,腹腔注射)可拮抗地西泮对戊四氮诱导的小鼠惊厥的抗惊厥作用,但不影响NBQX或GYKI 52466的抗惊厥活性[4] - Flumazenil(1-10 mg/kg,腹腔注射)以剂量依赖性方式阻断乙醇戒断大鼠的焦虑样行为(增加高架十字迷宫开放臂进入次数)[5] |
| 酶活实验 |
采用放射性配体结合实验评估Flumazenil对脑细胞膜制剂中苯二氮䓬受体的亲和力。将细胞膜与氚标记的苯二氮䓬配体在有或无Flumazenil的条件下孵育,随后通过过滤和闪烁计数测量特异性结合抑制情况[1]
- 小鼠脑匀浆中的受体结合研究显示,Flumazenil可置换放射性标记的苯二氮䓬配体,且在BALB/c和C57BL/6小鼠品系间存在结合特征差异[2] |
| 动物实验 |
Mice (BALB/c and C57BL/6 strains) were used to assess behavioral effects of Flumazenil. The drug was dissolved in a suitable vehicle and administered intraperitoneally at doses ranging from 0.1 to 10 mg/kg. Behavioral tests included locomotor activity monitoring and the Vogel conflict test, performed 30 minutes after drug administration [2]
- Mice were treated with Flumazenil via intraperitoneal injection at a dose of 10 mg/kg, 15 minutes prior to allopregnanolone administration (5 mg/kg, i.p.). The elevated plus-maze test was conducted 30 minutes after allopregnanolone administration to evaluate anxiolytic activity [3] - Mice were pretreated with Flumazenil (10 mg/kg, i.p.) 30 minutes before administration of diazepam, NBQX, or GYKI 52466. Seizures were induced by pentylenetetrazol, and seizure severity and latency were recorded for 30 minutes post-induction [4] - Rats were rendered ethanol-dependent by chronic ethanol vapor exposure. After ethanol withdrawal, Flumazenil was administered intraperitoneally at doses of 1, 3, and 10 mg/kg. Anxiety-like behavior was evaluated using the elevated plus-maze test 30 minutes after drug administration [5] - Human subjects with benzodiazepine overdose or therapeutic benzodiazepine effects were administered Flumazenil via intravenous infusion at a rate of 0.2 mg/min (total dose up to 1 mg) to reverse benzodiazepine-induced sedation and respiratory depression [1] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Flumazenil is completely metabolized (99%). The elimination of the radiolabeled drug is essentially completed within 72 hours, with 90% to 95% of the radioactive material appearing in urine and 5% to 10% in feces. Dosage: 0.9 to 1.1 L/kg 1 L/hr/kg [Healthy volunteers receive 1 mg infusion over 5 minutes] Metabolism/Metabolites Hepatic metabolism. Flumazenil is completely metabolized (99%). The major metabolites of flumazenil detected in urine are deethylated free acid and its glucuronide conjugates. Biological half-life Initial distribution half-life is 4 to 11 minutes, and terminal half-life is 40 to 80 minutes. The half-life is prolonged to 1.3 hours in patients with moderate hepatic impairment and to 2.4 hours in patients with severe hepatic impairment. Compared with adults, the elimination half-life of pediatric patients was more variable, averaging 40 minutes (range: 20 to 75 minutes). Flumazenil is rapidly absorbed after intravenous administration, with a volume of distribution in the human body of approximately 0.6–1.0 L/kg. The elimination half-life is 0.7–1.3 hours [1] - Flumazenil is extensively metabolized in the liver through oxidation and conjugation reactions, with less than 1% of the original drug excreted unchanged in the urine [1] - Due to significant first-pass metabolism, the oral bioavailability of flumazenil is low (approximately 16%) [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
Effects During Pregnancy and Lactation
◉ Overview of Use During Lactation There is currently no information regarding the clinical use of flumazenil during lactation. Since flumazenil is not absorbed orally, it is unlikely to have adverse effects on breastfed infants. If the mother needs to take flumazenil, there is no need to stop breastfeeding. The drug has a half-life of 54 minutes, therefore stopping breastfeeding 4 to 5 hours after administration minimizes drug transfer to the infant. ◉ Effects on Breastfed Infants As of the revision date, no relevant published information was found. ◉ Effects on Lactation and Breast Milk As of the revision date, no relevant published information was found. Protein Binding The protein binding rate is approximately 50%, primarily (66%) bound to albumin. Protein binding is reduced in patients with cirrhosis. Flumazenil has low toxicity in humans. Adverse reactions are mild and transient, including nausea, vomiting, anxiety, and seizures (rare, mainly seen in patients with epilepsy or benzodiazepine dependence) [1] - Flumazenil has a plasma protein binding rate of approximately 40-50% [1] |
| 参考文献 | |
| 其他信息 |
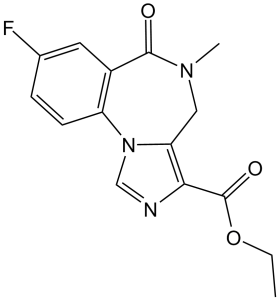
Flumazenil is an organic heterocyclic compound with the chemical name 5,6-dihydro-4H-imidazo[1,5-a][1,4]benzodiazepine, in which the 3, 5, 6, and 8 positions are substituted with ethoxycarbonyl, methyl, oxo, and fluorine atoms, respectively. It can be used as an antidote for benzodiazepine overdose. Flumazenil acts as a γ-aminobutyric acid (GABA) antagonist and is also an antidote for benzodiazepine poisoning. It is an ethyl ester, an organofluorine compound, and an imidazobenzodiazepine. Flumazenil is an imidazobenzodiazepine derivative and a potent benzodiazepine receptor antagonist. It competitively inhibits the activity of the benzodiazepine recognition site on the GABA/benzodiazepine receptor complex, thereby reversing the effects of benzodiazepines on the central nervous system. Flumazenil is a benzodiazepine antagonist. Flumazenil is an imidazobenzodiazepine derivative that effectively reverses benzodiazepine-induced activity. Flumazenil antagonizes the benzodiazepine binding site of the GABA/benzodiazepine receptor complex in the central nervous system (CNS), thereby preventing chloride channel opening and inhibiting neuronal hyperpolarization. Therefore, flumazenil can reverse the sedation, psychomotor disturbances, amnesia, and hypoventilation caused by benzodiazepines in a dose-dependent manner. It is a potent benzodiazepine receptor antagonist. Because it can reverse the sedative and other effects of benzodiazepines, it is considered an antidote for benzodiazepine overdose. Indications: For complete or partial reversal of the sedative effects of benzodiazepines, suitable for induction and/or maintenance of general anesthesia with benzodiazepines, and for sedation with benzodiazepines during diagnostic and therapeutic procedures. In addition, it can be used as an adjunct therapy to treat benzodiazepine overdose, in conjunction with appropriate supportive and symptomatic treatment.
FDA Label Mechanism of Action Flumazenil is an imidazobenzodiazepine derivative and belongs to the benzodiazepine antagonist class. It exerts its effect by competitively inhibiting the benzodiazepine binding site on the GABA/benzodiazepine receptor complex. In some animal models, flumazenil is a weak partial agonist, but in humans it has little or no agonist activity. Pharmacodynamics Flumazenil antagonizes the central nervous system effects of benzodiazepines, but it does not antagonize the central nervous system effects of drugs that affect GABAergic neurons through pathways other than benzodiazepine receptors (including ethanol, barbiturates, or general anesthetics), nor does it reverse the effects of opioids. Flumazenil (Ro 15-1788) is a selective competitive benzodiazepine receptor antagonist used clinically to reverse the effects of benzodiazepine overdose and terminate conscious sedation [1] -The partial agonist-like effect of flumazenil in BALB/c mice suggests strain-dependent differences in benzodiazepine receptor signaling or regulation [2] -Flumazenil blocks the anxiolytic effect of allogeneinolone, indicating that allogeneinolone exerts its anxiolytic effect through benzodiazepine receptor regulation [3] -Flumazenil does not interact with non-NMDA glutamate receptors because it failed to affect the anticonvulsant activity of NBQX or GYKI 52466 [4] -The anxiolytic blocking effect of flumazenil in ethanol withdrawal rats suggests that benzodiazepine receptor activation is involved in ethanol withdrawal-induced anxiety [5] |
| 分子式 |
C15H14FN3O3
|
|
|---|---|---|
| 分子量 |
303.29
|
|
| 精确质量 |
303.101
|
|
| CAS号 |
78755-81-4
|
|
| 相关CAS号 |
|
|
| PubChem CID |
3373
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.4±0.1 g/cm3
|
|
| 沸点 |
528.0±50.0 °C at 760 mmHg
|
|
| 熔点 |
201-203°C
|
|
| 闪点 |
273.1±30.1 °C
|
|
| 蒸汽压 |
0.0±1.4 mmHg at 25°C
|
|
| 折射率 |
1.634
|
|
| LogP |
0.67
|
|
| tPSA |
64.43
|
|
| 氢键供体(HBD)数目 |
0
|
|
| 氢键受体(HBA)数目 |
5
|
|
| 可旋转键数目(RBC) |
3
|
|
| 重原子数目 |
22
|
|
| 分子复杂度/Complexity |
461
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
OFBIFZUFASYYRE-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C15H14FN3O3/c1-3-22-15(21)13-12-7-18(2)14(20)10-6-9(16)4-5-11(10)19(12)8-17-13/h4-6,8H,3,7H2,1-2H3
|
|
| 化学名 |
ethyl 8-fluoro-5-methyl-6-oxo-4H-imidazo[1,5-a][1,4]benzodiazepine-3-carboxylate
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2 mg/mL (6.59 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.0 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2 mg/mL (6.59 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.0mg/mL澄清的DMSO储备液加入到900μL 20%SBE-β-CD生理盐水中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2 mg/mL (6.59 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2972 mL | 16.4859 mL | 32.9717 mL | |
| 5 mM | 0.6594 mL | 3.2972 mL | 6.5943 mL | |
| 10 mM | 0.3297 mL | 1.6486 mL | 3.2972 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。