| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1g |
|
||
| 5g |
|
||
| 10g |
|
||
| 25g |
|
||
| 50g |
|
||
| Other Sizes |
|

| 靶点 |
Aminoglycoside antibiotic; DNase I (IC50 = 0.57 mM)
|
|---|---|
| 体外研究 (In Vitro) |
庆大霉素对组织培养单层无害,不影响病毒复制,并且是比青霉素-链霉素联合使用更有效的抗生素激活剂[2]。庆大霉素已被证明对更广泛的生物体(铜绿假单胞菌、变形杆菌属和粪链球菌)具有杀菌作用,并已成功用作真菌学添加剂以限制细菌的生长。生成链霉素和青霉素[2]。庆大霉素在高压灭菌温度下稳定,对恒河猴肾、HeLa 和人羊膜细胞无害,并且不会干扰组织培养中某些脊髓灰质炎病毒和埃可病毒产生的细胞效应 [2]。几种小单孢菌产生庆大霉素[3]。核糖核酸[3]。
|
| 体内研究 (In Vivo) |
在使用小鼠的感染模型中,硫酸庆大霉素的注射剂和小鼠制剂显示出对鼠疫耶尔森氏菌的强大抗菌活性[3]。用硫酸庆大霉素(0.27 g/kg)治疗的小鼠表现出异物上的细菌显着减少[4]。】。
|
| 参考文献 | |
| 其他信息 |
硫酸庆大霉素是由发酵产生的抗生素复合物的硫酸盐。由三种化学结构密切相关的氨基糖组成。白色至淡黄色粉末。pH值(4%溶液)3.5至5.5。无味。(NTP, 1992) 美国国家毒理学计划,环境健康科学研究所,美国国立卫生研究院 (NTP)。1992。国家毒理学计划化学品库数据库。北卡罗来纳州三角研究园。
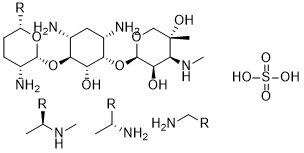
硫酸庆大霉素是庆大霉素的硫酸盐形式,庆大霉素是一种广谱氨基糖苷类抗生素复合物,由紫红小单孢菌 (Micromonospora purpurea) 或棘孢小单孢菌 (M. echinospora) 发酵产生,具有抗菌活性。庆大霉素是一种热稳定性复合物,包含庆大霉素C1、C1a、C2、C2a和C2b。硫酸庆大霉素是一种小分子药物,其临床试验阶段最高为IV期(涵盖所有适应症),于1970年首次获批,目前有7项已获批适应症和10项在研适应症。该药物已被美国食品药品监督管理局(FDA)列入黑框警告。它是一种由紫红微单孢菌(Micromonospora purpurea)产生的复杂抗生素物质,包含三种成分:硫酸庆大霉素C1、C2和C1A。 |
| 分子式 |
C24H55N7O11S3*
|
|---|---|
| 分子量 |
561.65 (Average)
|
| 精确质量 |
1487.86917
|
| CAS号 |
1405-41-0
|
| 相关CAS号 |
Gentamicin;1403-66-3
|
| PubChem CID |
9855350
|
| 外观&性状 |
White to off-white solid powder
|
| 沸点 |
797.6ºC at 760 mmHg
|
| 熔点 |
218-237°C
|
| 闪点 |
436.2ºC
|
| tPSA |
719.38
|
| 氢键供体(HBD)数目 |
26
|
| 氢键受体(HBA)数目 |
40
|
| 可旋转键数目(RBC) |
19
|
| 重原子数目 |
101
|
| 分子复杂度/Complexity |
1930
|
| 定义原子立体中心数目 |
36
|
| SMILES |
O=S(O)(O)=O.O[C@]1(C)C(NC)[C@@H](O)[C@@H](O[C@H]2C(N)C[C@H](N)[C@@H](O[C@@H]3[C@H](N)CC[C@@H]([R])O3)[C@@H]2O)OC1.NC[*].C[C@H]([*])N.C[C@H]([*])NC.[R=].[or].[or]
|
| InChi Key |
RDEIXVOBVLKYNT-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C21H43N5O7.C20H41N5O7.C19H39N5O7.H2O4S/c1-9(25-3)13-6-5-10(22)19(31-13)32-16-11(23)7-12(24)17(14(16)27)33-20-15(28)18(26-4)21(2,29)8-30-20;1-8(21)12-5-4-9(22)18(30-12)31-15-10(23)6-11(24)16(13(15)26)32-19-14(27)17(25-3)20(2,28)7-29-19;1-19(27)7-28-18(13(26)16(19)24-2)31-15-11(23)5-10(22)14(12(15)25)30-17-9(21)4-3-8(6-20)29-17;1-5(2,3)4/h9-20,25-29H,5-8,22-24H2,1-4H3;8-19,25-28H,4-7,21-24H2,1-3H3;8-18,24-27H,3-7,20-23H2,1-2H3;(H2,1,2,3,4)/t9?,10-,11+,12-,13+,14+,15-,16-,17+,18-,19-,20-,21+;8?,9-,10+,11-,12+,13+,14-,15-,16+,17-,18-,19-,20+;8-,9+,10-,11+,12-,13+,14+,15-,16+,17+,18+,19-;/m110./s1
|
| 化学名 |
mixtures of three isomers (Gentamicins C1, C2, and C1(subA).
|
| 别名 |
Gentamicin sulfate; Bristagen; U-Gencin; Gentacidin; Garacin; Garamycin; Gentak; Apogen; NSC-82261; Refobacin; Sch 9724; Gentafair
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ≥ 30 mg/mL
DMSO :< 1 mg/mL |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 100 mg/mL (Infinity mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
A Study to Compare Different Antibiotics and Different Modes of Fluid Treatment for Children With Severe Pneumonia
CTID: NCT04041791
Phase: Phase 3 Status: Completed
Date: 2024-07-08