| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 500mg |
|
||
| 1g |
|
||
| 5g |
|
||
| Other Sizes |
|

| 体外研究 (In Vitro) |
盐酸甲哌卡因通过与神经元细胞膜中特定的电压门控钠通道结合来抑制钠内流和膜去极化。这会导致可逆的感觉丧失以及神经冲动的开始和传导受阻。与其他局部麻醉剂相比,这种药物起效更快,持续时间也较短[2]。与普鲁卡因相比,盐酸甲哌卡因起效更快,中间作用时间更长[3]。盐酸甲哌卡因可优先阻断 Na(v)1.8,且依赖于使用,而 S(-)-布比卡因则优先阻断 TTX Na(+) 通道 [4]。
|
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
妊娠期和哺乳期影响
◉ 哺乳期用药概述 目前尚无关于哺乳期使用甲哌卡因的信息。鉴于其他局部麻醉药在母乳中的分泌量较低,哺乳期单次使用甲哌卡因不太可能对母乳喂养的婴儿产生不良影响。然而,尤其是在哺乳新生儿或早产儿时,可能更倾向于选择其他药物。 据报道,分娩期间作为局部麻醉药给予母亲的甲哌卡因会影响部分婴儿的初始哺乳行为,但不会影响产后前5天的体重增长。虽然针对甲哌卡因的研究尚不充分,但似乎在良好的母乳喂养支持下,无论是否联合芬太尼或其衍生物,硬膜外局部麻醉药对母乳喂养的成功率几乎没有或完全没有不良影响。分娩镇痛药物可能会延迟泌乳的开始。需要更多研究来阐明分娩期间使用甲哌卡因对母乳喂养结局的影响。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 一项研究比较了在正常分娩过程中使用甲哌卡因、布比卡因和利多卡因进行硬膜外镇痛的效果,结果发现,3组母乳喂养婴儿在产后前5天的体重变化无差异。所有组的总体体重增长均在正常范围内。 在分娩前1小时内接受甲哌卡因阴部神经阻滞的6名婴儿中,有4名婴儿开始哺乳的时间较晚,且最初的哺乳量少于10名分娩期间未接受麻醉的婴儿。这些差异的长期后果尚未报告。 一项针对孕晚期至产后12个月的妇女及其婴儿的全国性调查比较了分娩期间接受和未接受镇痛药物的母亲的泌乳第二阶段(lactroogenesis II)时间。药物类别包括:仅使用脊髓或硬膜外麻醉、脊髓或硬膜外麻醉联合其他药物,以及仅使用其他镇痛药物。与未接受分娩镇痛药物的妇女相比,接受任何类别药物的妇女发生泌乳第二阶段延迟(>72小时)的风险大约是前者的两倍。 |
| 参考文献 | |
| 其他信息 |
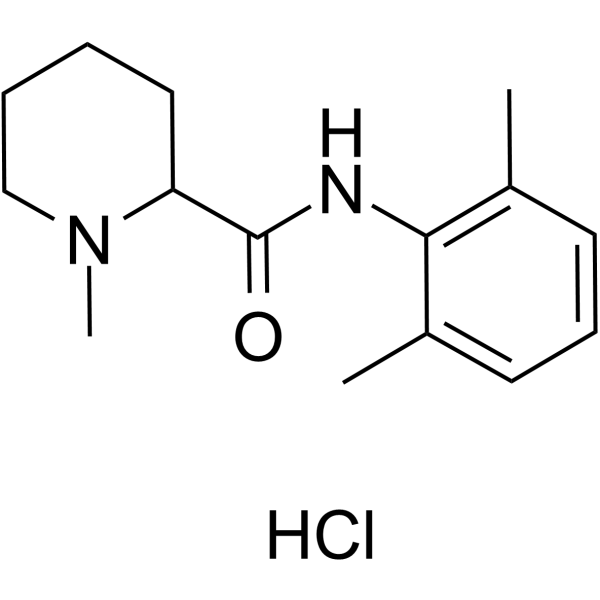
盐酸甲哌卡因是甲哌卡因的盐酸盐,用作局部麻醉剂。它是一种哌啶甲酰胺类化合物,具有局部麻醉作用。其功能与甲哌卡因相关。
盐酸甲哌卡因是甲哌卡因的盐酸盐形式,甲哌卡因是一种具有局部麻醉特性的酰胺衍生物。在注射部位,盐酸甲哌卡因通过与神经元细胞膜上的特定钠离子通道结合发挥作用,从而抑制钠离子内流。这会导致神经冲动传导阻滞,最终导致感觉丧失。 甲哌卡因是一种化学结构与布比卡因相关,但药理学上与利多卡因相关的局部麻醉剂。它适用于浸润麻醉、神经阻滞和硬膜外麻醉。甲哌卡因仅在大剂量局部使用时有效,因此不应通过此途径使用。 (摘自 AMA 药物评价,1994 年,第 168 页) 另见:甲哌卡因(具有活性部分);左去甲肾上腺素;盐酸甲哌卡因(成分)。 |
| 分子式 |
C15H23CLN2O
|
|---|---|
| 分子量 |
282.8089
|
| 精确质量 |
282.149
|
| CAS号 |
1722-62-9
|
| 相关CAS号 |
(+)-Mepivacaine;24358-84-7;Mepivacaine;96-88-8
|
| PubChem CID |
66070
|
| 外观&性状 |
White to off-white solid powder
|
| 沸点 |
383.1ºC at 760 mmHg
|
| 熔点 |
255-257ºC (dec.)
|
| 闪点 |
185.5ºC
|
| 蒸汽压 |
4.52E-06mmHg at 25°C
|
| LogP |
3.539
|
| tPSA |
32.34
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
19
|
| 分子复杂度/Complexity |
282
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
RETIMRUQNCDCQB-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C15H22N2O.ClH/c1-11-7-6-8-12(2)14(11)16-15(18)13-9-4-5-10-17(13)3;/h6-8,13H,4-5,9-10H2,1-3H3,(H,16,18);1H
|
| 化学名 |
N-(2,6-dimethylphenyl)-1-methylpiperidine-2-carboxamide;hydrochloride
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮和光照。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ~100 mg/mL (~353.59 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 100 mg/mL (353.59 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5359 mL | 17.6797 mL | 35.3594 mL | |
| 5 mM | 0.7072 mL | 3.5359 mL | 7.0719 mL | |
| 10 mM | 0.3536 mL | 1.7680 mL | 3.5359 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04561921 | Unknown status | Drug: Magnesium sulfate Drug: Mepivacaine-Levonordefrin Hydrochloride |
Success of Inferior Alveolar Nerve Block | Cairo University | 2020-10-22 | Not Applicable |
| NCT03725579 | Unknown status | Drug: mepivicane hydrochloride Drug: articane hydrochloride |
Symptomatic Irreversible Pulpitis | Cairo University | 2018-09-15 | Phase 2 Phase 3 |
| NCT04822415 | Completed | Drug: Mepivacaine Drug: Articaine |
Symptomatic Irreversible Pulpitis | Cairo University | 2014-12 | Not Applicable |
| NCT04947267 | Completed | Drug: 3% Mepivacaine Hydrochloride Drug: 2% Mepivacaine with 1:100,000 epinephrine |
LOCAL ANESTHESIA | Afshan Amjad Ali | 2018-05-30 | Phase 2 |
| NCT05765682 | Completed | Other: Mepivacaine Spinal Other: Bupivacaine Spinal |
Total Knee Replacement | Medical University of South Carolina | 2023-03-13 | Not Applicable |