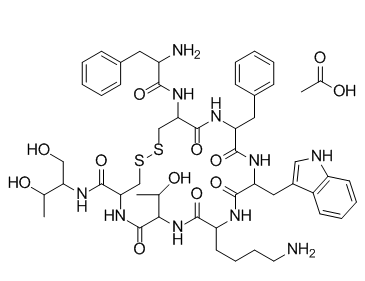
Octreotideacetate (SMS201-995; Sandostatin, SMS201995; Samilstin; Sandostatina; Octreotide-LAR; Longastatin) 是奥曲肽的醋酸盐,是一种八肽和生长抑素类似物,作为 sst2、sst3 和 sst5 生长抑素受体的激动剂。已被批准用于治疗激素分泌肿瘤;高胃泌素血症;糖尿病、高血压和小肠瘘。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Study of RYZ101 Compared with SOC in Pts W Inoperable SSTR+ Well-differentiated GEP-NET That Has Progressed Following 177Lu-SSA Therapy
CTID: NCT05477576
Phase: Phase 3 Status: Recruiting
Date: 2024-11-13
Octreotide Acetate and Recombinant Interferon Alfa-2b or Bevacizumab in Treating Patients With Metastatic or Locally Advanced, High-Risk Neuroendocrine Tumor
CTID: NCT00569127
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-13
Octreotide Treatment to Improve Nutritional Recovery After Surgery for Patients with Esophageal or Gastric Cancer
CTID: NCT04871204
Phase: Phase 2 Status: Completed
Date: 2024-10-31
Treatment of Orthostatic Intolerance
CTID: NCT00262470
Phase: Phase 1/Phase 2 Status: Active, not recruiting
Date: 2024-10-02
Human Models of Selective Insulin Resistance: Pancreatic Clamp
CTID: NCT06558422
Phase: Phase 1 Status: Not yet recruiting
Date: 2024-09-20
View More
Pancreatic Clamp in NAFLD
CTID: NCT05724134
Phase: Phase 1 Status: Recruiting
Date: 2024-09-19
Dexamethasone/Pancreatic Clamp P&F
CTID: NCT06126354
Phase: Phase 1 Status: Withdrawn
Date: 2024-07-10
Outcome of ADPKD With Octreotide LAR
CTID: NCT06193616
Phase: Status: Recruiting
Date: 2024-03-15
Angiotensin 2 for Hepatorenal Syndrome
CTID: NCT04048707
Phase: Phase 2 Status: Withdrawn
Date: 2024-03-06
Trial Using Octreotide to Enhance Liver Recovery After Hepatectomy
CTID: NCT03179995
Phase: Phase 2 Status: Terminated
Date: 2024-02-14
Comparison of 24-hours Versus 72-hours of Octreotide Infusion in Preventing Early Rebleed From Esophageal Varices
CTID: NCT03624517
Phase: Phase 4 Status: Recruiting
Date: 2024-01-08
Survivin Long Peptide Vaccine in Treating Patients With Metastatic Neuroendocrine Tumors
CTID: NCT03879694
Phase: Phase 1 Status: Recruiting
Date: 2023-12-29
The Effect of Satiety Gut Hormone Modulation on Appetitive Drive After Upper Gastrointestinal Surgery
CTID: NCT02381249
Phase: N/A Status: Completed
Date: 2023-11-27
Food Intake and Gut Hormones in Patients Who Have Undergone Upper Gastrointestinal Surgery for Cancer
CTID: NCT02385617
Phase: N/A Status: Completed
Date: 2023-11-27
A Study to Evaluate Patient Experience in the Therapy of Neuroendocrine Tumors Treated With Octreotide Long Acting Release Versus Lanreotide
CTID: NCT03289741
Phase: Phase 4 Status: Completed
Date: 2023-11-07
Efficacy and Safety of Octreotide in Laparoscopic Hepatectomy Surgery: Effect on Blood Loss, Need for Vasoactive Drugs, Transfusion Requirements.
CTID: NCT06085976
Phase: Phase 2 Status: Recruiting
Date: 2023-10-17
AMG 706 and Octreotide in Treating Patients With Low-Grade Neuroendocrine Tumors
CTID: NCT00427349
Phase: Phase 2 Status: Completed
Date: 2023-07-05
Octreotide Compared With Loperamide Hydrochloride for Chemotherapy-Related Diarrhea in Patients With Colorectal Cancer
CTID: NCT00003057
Phase: Phase 3 Status: Completed
Date: 2023-06-18
Octreotide With or Without Prednisone in Treating Patients With Metastatic or Recurrent Thymoma
CTID: NCT00003283
Phase: Phase 2 Status: Completed
Date: 2023-06-15
Combination of Everolimus and Octreotide LAR in Aggressive Recurrent Meningiomas
CTID: NCT02333565
Phase: Phase 2 Status: Completed
Date: 2023-05-25
Efficacy of Octreotide on Blood and Iron Requirements in Patients With Anemia Due to Angiodysplasias
CTID: NCT02384122
Phase: Phase 3 Status: Completed
Date: 2023-05-24
A Feasibility Study of Octreotide Infusion During Liver Transplant.
CTID: NCT04941911
Phase: Phase 2 Status: Active, not recruiting
Date: 2023-05-03
Macro and Microcirculatory Effects of the Combination of Norepinephrine and Octreotide for the Treatment of Cirrhotic Patients With Hemorrhagic Shock
CTID: NCT03891849
Phase: N/A Status: Withdrawn
Date: 2023-02-09
The Effect of Octreotide on Wound Drainage After Mastectomy
CTID: NCT05682209
Phase: Phase 4 Status: Completed
Date: 2023-01-18
Ghrelin Suppression by Octreotide in Prader-Willi
CTID: NCT01613495
Phase: N/A Status: Unknown status
Date: 2022-12-16
Palliative Management of Inoperable Malignant Bowel Obstruction
CTID: NCT04027348
Phase: Phase 2 Status: Terminated
Date: 2022-11-29
Everolimus and Octreotide Acetate With or Without Bevacizumab in Treating Patients With Locally Advanced or Metastatic Pancreatic Neuroendocrine Tumors That Cannot Be Removed by Surgery
CTID: NCT01229943
Phase: Phase 2 Status: Completed
Date: 2022-08-04
Investigation of the Effects of Obesity Surgery on Appetitive Behaviour
CTID: NCT02010385
Phase: N/A Status: Completed
Date: 2022-05-23
Comparison of 2 Days Versus 5 Days of Octreotide After Endoscopic Therapy in Preventing Early Esophageal Varices Rebleed : A Randomized Controlled Study
CTID: NCT05199038
Phase: Phase 4 Status: Unknown status
Date: 2022-05-18
Effect of Hyperglycemia on Microvascular Perfusion in Healthy Adults
CTID: NCT03520569
PhaseEarly Phase 1 Status: Completed
Date: 2022-05-13
Effect of Octreotide on Saliva
CTID: NCT05340192
Phase: Phase 1 Status: Completed
Date: 2022-04-22
Comparison of Oral Octreotide Capsules to Injectable Somatostatin Analogs in Acromegaly
CTID: NCT02685709
Phase: Phase 3 Status: Completed
Date: 2022-04-22
The Longitudinal Approach to Acromegaly: A Pattern of Treatment and Comparative Effectiveness Research
CTID: NCT03158090
Phase: Status: Recruiting
Date: 2022-03-16
Efficacy and Safety Evaluation of Octreotide in the Treatment of Congenital Hyperinsulinemia
CTID: NCT05171751
Phase: Status: Completed
Date: 2021-12-29
Cattell-Warren Versus Blumgart Techniques of Pancreatico-jejunostomy Following Pancreato-duodenectomy
CTID: NCT02457156
Phase: Phase 3 Status: Unknown status
Date: 2021-12-10
Functional MRI-based Assessment of Terlipressin vs. Octreotide on Renal Function in Cirrhotic Patients With Acute Variceal Bleeding (CHESS1903)
CTID: NCT04028323
Phase: Phase 4 Status: Unknown status
Date: 2021-08-17
Gastrointestinal Nutrient Transit and Enteroendocrine Function After Upper Gastrointestinal Surgery
CTID: NCT03734627
Phase: Status: Completed
Date: 2021-08-16
Prevention of Postoperative Pancreatic Fistula by Somatostatin
CTID: NCT03000946
Phase: Phase 3 Status: Completed
Date: 2021-05-03
Efficacy and Safety of Octreotide Capsules (MYCAPSSA) in Acromegaly
CTID: NCT03252353
Phase: Phase 3 Status: Unknown status
Date: 2020-11-23
Octreotide for Management of Bronchorrhea in Mechanically Ventilated Patients
CTID: NCT02916433
Phase: Phase 2 Status: Completed
Date: 2020-10-08
Octreotide LAR in the Induction of Immunologic Response in NENs Patients
CTID: NCT04129255
Phase: Phase 2 Status: Completed
Date: 2020-09-10
Prophylactic Octreotide to Prevent Post Duodenal EMR and Ampullectomy Bleeding
CTID: NCT02032784
Phase: Phase 4 Status: Terminated
Date: 2020-07-16
Hemodynamic Profile of Terlipressin and Octreotide in Patients With Cirrhosis and Portal Hypertension. A Randomised, Single Blinded Clinical Trial.
CTID: NCT04353193
Phase: Phase 4 Status: Unknown status
Date: 2020-04-20
Tamoxifen With or Without Octreotide in Treating Postmenopausal Women With Stage I, Stage II, or Stage III Breast Cancer
CTID: NCT00002864
Phase: Phase 3 Status: Completed
Date: 2020-04-01
Stress, Diurnal Cortisol, and Breast Cancer Survival
CTID: NCT00226967
Phase: Status: Completed
Date: 2020-01-27
Etiology, Assessment and Treatment of Post-gastric Bypass Severe Hypoglycemia
CTID: NCT01865760
Phase: N/A Status: Completed
Date: 2019-10-28
Sandostatin (Octreotide LAR) May Lead to Clinical Improvement Through Receptor Occupation Optimisation
CTID: NCT04140409
Phase: Phase 4 Status: Terminated
Date: 2019-10-25
Octreotide VS Placebo in Prevention of Salivary Fistulae After Post Radiation Salvage Surgery
CTID: NCT02437825
Phase: Phase 2 Status: Unknown status
Date: 2019-09-12
Study of Prophylactic Octreotide to Prevent or Reduce the Frequency and Severity of Diarrhoea in Subjects Receiving Lapatinib With Capecitabine for the Treatment of Metastatic Breast Cancer
CTID: NCT02294786
Phase: Phase 2 Status: Terminated
Date: 2019-07-15
Terlipressin on Effect of Renal Function in Cirrhotic Patients With Acute Gastrointestinal Hemorrhage
CTID: NCT03846180
Phase: Status: Completed
Date: 2019-07-05
A Four-Part Study to Assess the Safety, Tolerability, PK and PD of ONO-5788 in Healthy Adult Volunteers
CTID: NCT03571594
Phase: Phase 1 Status: Terminated
Date: 2019-06-05
Midodrine, Octreotide and Albumin: Impact on Renal Function of Patients With Liver Cirrhosis and Renal Failure
CTID: NCT01587222
Phase: Phase 2 Status: Withdrawn
Date: 2019-02-15
Recurrence Rates of Type I Gastric Neuroendocrine Tumors Treated With Long-acting Somatostatin Analogs
CTID: NCT03812939
Phase: Status: Unknown status
Date: 2019-01-23
Effects of Glucagon Administration on Energy Expenditure
CTID: NCT02237053
Phase: Phase 1 Status: Completed
Date: 2018-04-26
Comparison of Terlipressin, Somatostatin, and Octreotide for Control of Variceal Bleeding
CTID: NCT00966355
Phase: Phase 4 Status: Completed
Date: 2018-03-19
The Effect of Subcutaneous Infusions of 3 Doses of DG3173 on Growth Hormone Levels in Untreated Acromegalics
CTID: NCT02217800
Phase: Phase 2 Status: Completed
Date: 2018-02-15
Single Dose Pharmacology Study of DG3173 and Octreotide in Acromegalic Patients.
CTID: NCT02235987
Phase: Phase 2 Status: Completed
Date: 2018-02-15
Somatostatin In Patients With Autosomal Dominant Polycystic Kidney Disease And Moderate To Severe Renal Insufficiency
CTID: NCT01377246
Phase: Phase 3 Status: Completed
Date: 2018-01-10
RCT to Determine the Efficacy of Combining Hemospray With Medical Treatment in Acute Variceal Bleeding
CTID: NCT03061604
Phase: N/A Status: Completed
Date: 2017-11-14
Octreotide in the Prevention of Postoperative Complications After Pancreaticoduodenectomy
CTID: NCT02474914
Phase: N/A Status: Completed
Date: 2017-08-22
Efficacy and Safety of Octreotide (MYCAPSSA™ [Formerly Octreolin™]) for Acromegaly
CTID: NCT01412424
Phase: Phase 3 Status: Completed
Date: 2017-08-17
Safety and Efficacy of Pasireotide Long Acting Release (LAR) vs. Octreotide LAR in Patients With Active Acromegaly
CTID: NCT00600886
Phase: Phase 3 Status: Completed
Date: 2017-07-02
Octreotide in Treating Patients With Cancer-Related Malignant Ascites
CTID: NCT00182754
Phase: Phase 3 Status: Completed
Date: 2017-04-11
Efficacy of Octreotide Treatment in Patients With Primary Inoperable Thymoma
CTID: NCT00332969
Phase: Phase 2 Status: Completed
Date: 2017-03-31
Octreotide Efficacy and Safety in First-line Acromegalic Patients
CTID: NCT00171886
Phase: Phase 4 Status: Completed
Date: 2017-02-24
Treatment of Orthostatic Hypotension in Autonomic Failure
CTID: NCT00223691
Phase: Phase 1 Status: Completed
Date: 2017-01-18
Fludarabine Plus Octreotide in Treating Patients With Relapsed Low-Grade Non-Hodgkin's Lymphoma
CTID: NCT00002779
Phase: Phase 2 Status: Completed
Date: 2016-12-15
Fecal Calprotectin Levels in Mycophenolate Mofetil Induced Diarrhea and Treatment With Octreotide
CTID: NCT02977897
Phase: Status: Unknown status
Date: 2016-11-30
Impact of Prophylactic Octreotide to Pancreatic Exocrine Secretion
CTID: NCT02920567
Phase: N/A Status: Unknown status
Date: 2016-09-30
Phase 1, Open-label, Drug-drug Interaction Study With Octreotide Acetate Injection and Telotristat Etiprate in Healthy Subjects
CTID: NCT02195635
Phase: Phase 1 Status: Completed
Date: 2016-09-07
Cixutumumab, Everolimus, and Octreotide Acetate in Treating Patients With Advanced Low to Intermediate Grade Neuroendocrine Carcinoma
CTID: NCT01204476
Phase: Phase 1 Status: Completed
Date: 2016-07-15
Octreotide in Preventing Diarrhea in Patients Who Are Undergoing Radiation Therapy to the Pelvis
CTID: NCT00033605
Phase: Phase 3 Status: Completed
Date: 2016-07-13
Phase II Study With ITF2984 in Acromegalic Patients
CTID: NCT02111044
Phase: Phase 2 Status: Completed
Date: 2016-06-16
Hemodynamic Effects of Terlipressin and High Dose Octreotide
CTID: NCT02119884
Phase: Phase 4 Status: Completed
Date: 2016-06-13
Evaluation of the Efficacy of Long-acting Release Octreotide in Patients With Advanced Hepatocellular Carcinoma
CTID: NCT00241020
Phase: Phase 3 Status: Completed
Date: 2016-04-12
Effect of Octreotide on the Colonic Motility in Pediatric Patients
CTID: NCT01917773
Phase: Phase 4 Status: Completed
Date: 2015-12-21
Octreotide in Preventing or Reducing Diarrhea in Patients Receiving Chemoradiotherapy for Anal or Rectal Cancer
CTID: NCT00075868
Phase: Phase 3 Status: Completed
Date: 2015-11-17
Vatalanib and Octreotide in Treating Patients With Progressive Neuroendocrine Tumors
CTID: NCT00227773
Phase: Phase 2 Status: Withdrawn
Date: 2015-10-08
Hormonal Outcomes in Acromegalic Patients With Treated Surgery With or Without Long Acting Somatostatin Analogues
CTID: NCT02427295
Phase: Phase 4 Status: Unknown status
Date: 2015-04-28
Effect of Obesity-derived Cytokines on Protein Turnover and Carbohydrate Metabolism in Human Skeletal Muscle
CTID: NCT02305069
Phase: N/A Status: Completed
Date: 2014-12-02
Cabazitaxel Plus Prednisone With Octreotide For Castration-Resistant Prostate Cancer (CRPC) Previously Treated With Docetaxel
CTID: NCT01469338
Phase: Phase 2 Status: Terminated
Date: 2014-11-24
Everolimus and Octreotide in Patients With Advanced Carcinoid Tumor
CTID: NCT00412061
Phase: Phase 3 Status: Completed
Date: 2014-11-21
Single Dose Pharmacodynamic and Pharmacokinetic Study of DG3173
CTID: NCT02217826
Phase: Phase 1 Status: Completed
Date: 2014-08-19
Multiple Ascending Dose Tolerability, Pharmacokinetic and Pharmacodynamic Study of DG3173
CTID: NCT02217839
Phase: Phase 1 Status: Completed
Date: 2014-08-15
Octreotide Therapy in Children and Young Adults With Prader-Willi Syndrome (PWS)
CTID: NCT00399893
Phase: N/A Status: Terminated
Date: 2014-07-24
Preoperative Octreotide Treatment of Acromegaly
CTID: NCT00521300
Phase: Phase 4 Status: Completed
Date: 2014-06-02
Octreotide and Doxorubicin in Treating Patients With Advanced Cancer
CTID: NCT00008073
Phase: Phase 1 Status: Completed
Date: 2013-12-19
Sandostatin for Patients With Androgen Independent Prostate Cancer
CTID: NCT00510224
Phase: Phase 2 Status: Terminated
Date: 2013-12-11
Long-term Safety and Efficacy Study of Octreotide Implant in Patients With Acromegaly
CTID: NCT01295060
Phase: Phase 3 Status: Terminated
Date: 2013-09-20
Pharmacokinetics, Efficacy and Safety of an Octreotide Implant in Patients With Carcinoid Syndrome
CTID: NCT0088
Hemodynamic profile of terlipressin and octreotide in patients with cirrhosis and portal hypertension. A randomised, single blinded clinical trial.
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2020-04-13
A Phase 3, randomized, double-blind, placebo-controlled, multi-center trial to assess efficacy and safety of octreotide subcutaneous depot (CAM2029) in patients with acromegaly
CTID: null
Phase: Phase 3 Status: Ongoing, GB - no longer in EU/EEA, Completed
Date: 2020-02-13
This is a multicenter, stratified, randomized, open-label comparator-controlled, Phase III study in patients with somatostatin receptor positive, well-differentiated G2 and G3, advanced GEP NETs, diagnosed within 6 months prior to screening, comparing treatment with Lutathera (7.4GBq/200 mCi x 4 administrations every 8± 1 weeks; cumulative dose: 29.6 GBq/800mCi) plus octreotide long-acting (30 mg every 8 weeks during Lutathera treatment and every 4 weeks after last Lutathera treatment) and high dose octreotide long-acting (60 mg every 4 weeks).
CTID: null
Phase: Phase 3 Status: Trial now transitioned, GB - no longer in EU/EEA, Ongoing
Date: 2019-11-15
A Phase 3, open-label, single-arm, multi-center trial to assess the long term safety of octreotide subcutaneous depot (CAM2029) in patients with acromegaly
CTID: null
Phase: Phase 3 Status: Trial now transitioned, Ongoing, GB - no longer in EU/EEA
Date: 2019-07-12
Effectiveness of Somatostatin Analogues in Patients with hereditary hemorrhagic telangiectasia and symptomatic gastrointestinal bleeding, the SAIPAN-trial: a multicenter, randomized, open-label, parallelgroup, superiority trial.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2019-06-03
A phase 3, randomized, double-blind, placebo-controlled, multicenter study to evaluate efficacy and safety of octreotide capsules in patients who previously tolerated and demonstrated biochemical control on injectable somatostatin receptor ligands (SRL) treatment
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA, Completed
Date: 2017-10-27
Octreotide LAR in the induction of immunologic response in patient with neuroendocrine tumors: an interventional pharmacological study
CTID: null
Phase: Phase 2 Status: Completed
Date: 2017-07-11
Evaluation of the impact of a Sandostatin injection before axillary node dissection on lymphorrhea in patients operated for breast cancer
CTID: null
Phase: Phase 2 Status: Completed
Date: 2016-05-09
A PHASE 3, RANDOMIZED, OPEN-LABEL, ACTIVE CONTROLLED, MULTICENTER STUDY TO EVALUATE MAINTENANCE OF RESPONSE, SAFETY AND PATIENT REPORTED OUTCOMES IN ACROMEGALY PATIENTS TREATED WITH OCTREOTIDE CAPSULES, AND IN PATIENTS TREATED WITH STANDARD OF CARE PARENTERAL SOMATOSTATIN RECEPTOR LIGANDS WHO PREVIOUSLY TOLERATED AND DEMONSTRATED A BIOCHEMICAL CONTROL ON BOTH TREATMENTS
CTID: null
Phase: Phase 3 Status: Prematurely Ended, Completed
Date: 2016-04-18
The Effect of Satiety Gut Hormone Modulation on Appetitive Drive After Upper Gastrointestinal Surgery
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2015-12-11
A randomised, open-label clinical trial assessing the efficacy of octreotide to decrease iron infusion and blood transfusion requirements in patients with refractory anaemia due to gastrointestinal bleeding from angiodysplasias.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2015-09-09
Follow-Up Study in Patients with Acromegaly Previously Participating in Chiasma Study CH-ACM-01
CTID: null
Phase: Phase 3 Status: Completed
Date: 2015-06-30
A Randomised, Multicentre, Open Label, Phase II study of Prophylactic Octreotide to Prevent or Reduce the Frequency and Severity of Diarrhoea in Subjects Receiving Lapatinib with Capecitabine for the Treatment of Metastatic Breast Cancer.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2014-08-21
Evaluation of a standardized treatment regimen for acromegaly: A multi centre intervention study
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2014-08-05
A Phase II, Open-label, Multicentre, Randomised Study of the Pharmacokinetics, Pharmacodynamics, Efficacy, and Safety of CAM2029 in Two Patient Groups with Acromegaly and Neuroendocrine Tumours (NET) Previously Treated with Sandostatin® LAR®
CTID: null
Phase: Phase 2 Status: Completed
Date: 2014-07-02
Sandostatin therapy in sarcoidosis
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2014-06-20
Activity and safety of Everolimus in combination with octreotide LAR and Metformin in patients with advanced pancreatic well-differentiated Neuroendocrine Tumors (pWDNETs): a Phase II, open, monocentric, prospective study.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2014-06-05
A Randomized, Multicenter, Phase II study to Investigate Efficacy and Safety of ITF2984 in Acromegalic patients.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2014-02-25
Impact of 68Ga-DOTAOTC PET for diagnosis of newly diagnosed or recurrent meningiomas
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2014-02-12
AN OPEN, MONOCENTRIC, NOT CONTROLLED CLINICAL TRIAL TO DEVELOP AND EVALUATE THE CLINICAL APPLICATION OF A NEW BETA PROBE FOR RADIOGUIDED SURGERY IN MENINGIOMA TUMOR
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2013-12-13
Imaging with 111In-Octreotide SPECT-CT compared to 68Ga-DOTATATE PET-CT in patients admitted for evaluation of GastroEnteroPancreatic NeuroEndocrine Tumours, GEP-NETs
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2013-08-22
68Ga-DOTATOC-PET/CT for diagnosis of neuroendocrina tumours
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2012-10-29
A Randomized, Double-Blind, Double Dummy, Placebo-Controlled, Parallel Group Study to Evaluate the Efficacy of LF-PB 10 mg, 20 mg, and 30 mg to Treat Lymphorrhea Post Axillary Dissection in Breast Cancer
CTID: null
Phase: Phase 2 Status: Completed
Date: 2012-10-09
A multi-centre, stratified, open, randomized, comparator-controlled, parallel-group phase III study comparing treatment with 177Lu-DOTA0-Tyr3-Octreotate to Octreotide LAR in patients with inoperable, progressive, somatostatin receptor positive, midgut carcinoid tumours.
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA, Completed
Date: 2012-04-25
Midrodrine, octeotride and albumin for cirrhotic patients with functional renal impairment
CTID: null
Phase: Phase 4 Status: Completed
Date: 2012-04-03
A prospective trial with ketoconazole and octreotide combination therapy for treatment of Cushing’s disease.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2011-10-31
Efficacy and safety of oral Octreolin™ in patients with acromegaly who are currently receiving parenteral somatostatin analogs
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-08-31
Estudio de prueba de concepto diseñado para evaluar los efectos de Octreolin sobre el gradiente de presión venosa hepática en sujetos con cirrosis e hipertensión portal
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2011-04-15
A prospective, randomized, double-blind, placebo controlled clinical trial to assess the effects of long-acting somatostatin (Octreotide LAR)therapy on disease progression in patients with Autosomal Dominant Polycystic Kidney Disease and moderate to severe renal insufficiency
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-04-04
A phase III, multicenter, randomized, parallel-group study to assess the efficacy and safety of double-blind pasireotide LAR 40 mg and pasireotide LAR 60 mg versus open-label octreotide LAR or lanreotide ATG in patients with inadequately controlled acromegaly
CTID: null
Phase: Phase 3 Status: Completed
Date: 2010-06-10
EFFECTS OF LONG-ACTING SOMATOSTATIN ON DISEASE PROGRESSION IN PATIENTS WITH AUTOSOMAL DOMINANT POLYCYSTIC KIDNEY DISEASE AND MODERATE/SEVERE RENAL INSUFFICIENCY(ALADIN II STUDY)
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2009-11-16
An open label, single arm, phase II study of combination RAD001 and octreotide LAR in patients with advanced neuroendocrine tumors as first line treatment
CTID: null
Phase: Phase 2 Status: Completed
Date: 2009-03-12
PHASE III, OPEN-LABEL, MULTICENTER INTERNATIONAL STUDY TO EVALUATE THE EFFICACY AND SAFETY OF AN OCTREOTIDE IMPLANT VERSUS SANDOSTATIN LAR® DEPOT IN PATIENTS WITH ACROMEGALY
CTID: null
Phase: Phase 3 Status: Completed
Date: 2008-10-29
Traitement par Sandostatine LP remplaçant le traitement par Sandostatine SC chez les enfants présentant un hyperinsulinisme congénital résistant au diazoxide.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2008-09-10
A multi-center, randomized, double-blind, placebo-controlled, crossover study in women with irritable bowel syndrome to evaluate feasibility and reproducibility of barostat assessments of colorectal sensation during colorectal distention and its pharmacological modulation using octreotide
CTID: null
Phase: Phase 2 Status: Completed
Date: 2008-02-04
Co-treatment with pegvisomant and a somatostatin analogue (SA) in SA-responsive acromegalic patients: impact on insulin sensitivity, glucose tolerance, and pharmacoeconomics
CTID: null
Phase: Phase 4 Status: Completed
Date: 2008-01-31
A multicenter, randomized, blinded efficacy and safety study of pasireotide LAR vs octreotide LAR in patients with metastatic carcinoid tumors whose disease-related symptoms are inadequately controlled by somatostatin analogues
CTID: null
Phase: Phase 3 Status: Completed, Prematurely Ended
Date: 2008-01-14
Receptor radionuclide therapy with [177Lu-
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2008-01-10
Prospektive, offene Studie zur Prüfung der Wirksamkeit der zusätzlichen Gabe des Somatostatinanalogon Octreotid (Sandostatin) bzw. des Dopaminagonisten Cabergolin (Dostinex) bei Patientin mit Akromegalie unter laufender Therapie mit Pegvisomant (Somavert);
CTID: null
Phase: Phase 4 Status: Completed
Date: 2007-12-20
A multicenter, randomized, blinded study to assess the safety and efficacy of pasireotide LAR vs. octreotide LAR in patients with active acromegaly.
CTID: null
Phase: Phase 3 Status: Prematurely Ended, Completed
Date: 2007-11-15
OPEN LABEL EXTENSION STUDY EVALUATING THE SAFETY AND BIOLOGICAL ACTIVITY OF A NEW PROLONGED RELEASE FORMULATION OF OCTREOTIDE ACETATE, C2L-OCT-01 PR, ADMINISTERED INTRA MUSCULARLY EVERY 6 WEEKS IN ACROMEGALIC PATIENTS
CTID: null
Phase: Phase 3 Status: Completed, Prematurely Ended
Date: 2007-10-29
SAFETY AND BIOLOGICAL ACTIVITY OF A NEW PROLONGED RELEASE FORMULATION OF OCTREOTIDE ACETATE, C2L-OCT-01 PR, ADMINISTERED INTRA MUSCULARLY EVERY 6 WEEKS IN ACROMEGALIC PATIENTS
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2007-10-25
An open-label, two-step, multicenter European study to evaluate the efficacy and safety of Sandostatin LAR at High Dose or in combination either with GH-receptor antagonist or dopamine-agonist in acromegalic patients not adequately controlled by conventional regimen
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-04-20
Phase I study of Somatostatin-Analogues as Second Line Treatment in Adult Patients with Recurrent Medulloblastoma (WHO IV)
CTID: null
Phase: Phase 1, Phase 2 Status: Prematurely Ended
Date: 2007-04-17
Sandostatin® in the treatment of diarrhea in patients with ileal pouch anal anastomosis.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2007-04-10
OPEN LABEL, RANDOMIZED STUDY COMPARING THE BIOLOGICAL EFFICACY AND SAFETY OF A NEW PROLONGED RELEASE FORMULATION OF OCTREOTIDE ACETATE, C2L-OCT-01 PR, 30 MG ADMINISTERED INTRA MUSCULARLY EVERY 42 DAYS FOR 84 DAYS WITH SANDOSTATIN LAR® 30 MG ADMINISTERED INTRA MUSCULARLY EVERY 28 DAYS FOR 84 DAYS TO ACROMEGALIC PATIENTS
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-02-22
A randomized, double-blind, placebo-controlled, multicenter phase III study in patients with advanced carcinoid tumor receiving Sandostatin LAR® and RAD001 10 mg/d or Sandostatin LAR® and placebo
CTID: null
Phase: Phase 3 Status: Prematurely Ended, Completed
Date: 2007-01-22
phase II study of the combination of bevacizumab plus somatostatin analogue and metronomic capecitabine as first-line therapy in patients with advanced inoperable well-differentiated neuroendrocrine tumors
CTID: null
PhasePha
e.querySelector("font strong").in