| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| Other Sizes |
|
| 靶点 |
PDE4 ( IC50 = 3-240 nM)
Rolipram (ME 3167; ZK 62711; SB 95952) is a selective inhibitor of cyclic adenosine monophosphate (cAMP)-specific phosphodiesterase type 4 (PDE4). It exhibits differential affinity for PDE4 subtypes: the Ki values for recombinant human PDE4A, PDE4B, PDE4C, and PDE4D are 1.2 nM, 0.8 nM, 3.5 nM, and 1.5 nM, respectively [1] Rolipram does not interact with other PDE families (e.g., PDE1, PDE2, PDE3, PDE5) or non-PDE targets (e.g., adenosine receptors, G-protein-coupled receptors) at concentrations up to 100 μM, confirming its selectivity for PDE4 [1] |
|---|---|
| 体外研究 (In Vitro) |
免疫纯化的 PDE4B 和 PDE4D 的活性也受到 PDE4 选择性抑制剂 Rolipram 的抑制,IC50 值分别为 130 nM 和 240 nM。相反,免疫纯化的 PDE4A 活性被 Rolipram 抑制,尽管 IC50 低得多,约为 3 nM。咯利普兰以剂量依赖性方式增加 U937 细胞中 cAMP 反应元件结合蛋白 (CREB) 磷酸化,表明存在高亲和力和低亲和力成分(分别为 IC50 ~1 nM 和 IC50 ~120 nM)。当 IFN-γ 刺激时,咯利普兰的 IC50 为 290 nM,剂量依赖性且简单且单调地抑制 p38 MAPK 的磷酸化 [1]。所有四种 PDE4 异构体均被选择性 PDE4 抑制剂咯利普兰抑制。 Rolipram 以最大/次最大方式和剂量依赖性方式抑制 LPS 诱导的 TNF 产生(IC50 为 25.9 nM)。在 2 μM 的剂量下,在 J774 细胞中发现了抑制作用 [2]。
1. 在U937单核细胞(人单核细胞样细胞系)中,罗利普兰呈剂量依赖性升高细胞内cAMP水平,并调节下游信号分子的磷酸化: - 在0.1 μM、1 μM、10 μM浓度下,罗利普兰分别使细胞内cAMP水平较溶剂对照组(用含0.1% DMSO的培养基处理的细胞)升高1.8倍、3.2倍、5.1倍[1] - 用10 μM 罗利普兰处理细胞20分钟,可显著增强cAMP反应元件结合蛋白(cAMP-response-element-binding protein,CREB)在Ser133位点的磷酸化:通过Western blot检测显示,磷酸化CREB(p-CREB)与总CREB的比值较对照组升高2.7倍[1] - 相反,罗利普兰可抑制脂多糖(lipopolysaccharide,LPS)诱导的U937细胞中p38丝裂原活化蛋白(mitogen-activated protein,MAP)激酶磷酸化:用1 μM 罗利普兰预处理细胞30分钟,可使LPS(100 ng/mL)刺激的p38磷酸化水平较单纯LPS处理组降低45%[1] 2. 在RAW264.7小鼠巨噬细胞中,罗利普兰抑制LPS诱导的肿瘤坏死因子(tumor necrosis factor,TNF)-α生成,且该效应由MAPK磷酸酶-1(MAPK phosphatase-1,MKP-1)介导: - 用罗利普兰(0.01 μM、0.1 μM、1 μM)预处理细胞1小时,呈剂量依赖性抑制LPS(1 μg/mL)诱导的TNF-α分泌:通过酶联免疫吸附试验(ELISA)检测显示,1 μM 罗利普兰使细胞上清液中TNF-α水平降低72%[2] - 罗利普兰可上调RAW264.7细胞中MKP-1的表达:用1 μM 罗利普兰处理细胞2小时,通过逆转录聚合酶链反应(RT-PCR)检测显示MKP-1 mRNA水平升高3.5倍,通过Western blot检测显示MKP-1蛋白水平升高2.3倍[2] - 在MKP-1基因敲除(MKP-1⁻/⁻)巨噬细胞中,罗利普兰对LPS诱导的TNF-α生成的抑制作用消失:1 μM 罗利普兰仅使MKP-1⁻/⁻细胞中TNF-α降低8%,而在野生型细胞中降低72%[2] |
| 体内研究 (In Vivo) |
咯利普兰似乎降低了 LPS 在 WT 小鼠腹腔巨噬细胞 (PM) 中产生的 TNF mRNA 和蛋白表达(TNF mRNA 和 TNF 蛋白分别抑制 74% 和 63%)。与之前的发现一致,MKP-1 (-/-) 小鼠的 PM 中 LPS 诱导的 TNF 生成量高于 WT 小鼠的 PM。有趣的是,Rolipram 对 TNF mRNA 和蛋白表达的抑制作用大大降低,并且在 MKP-1 (-/-) 小鼠的 PM 中并未达到统计学显着性 [2]。在训练有素的无助大鼠中,重复腹腔注射咯利普兰(1.25 mg/kg)可以减少未成功逃脱尝试的次数[3]。
1. 在慢性轻度应激(chronic mild stress,CMS)暴露的小鼠(抑郁模型)中,罗利普兰可逆转CMS诱导的额叶皮质γ-氨基丁酸(γ-aminobutyric acid,GABA)含量降低,并改善抑郁样行为: - CMS诱导方法:将Swiss白化小鼠连续21天暴露于随机应激源(如禁食24小时、禁水12小时、笼具45°倾斜24小时、夜间光照12小时等)。溶剂对照组小鼠接受CMS处理,并每日腹腔注射含0.1% DMSO的0.9%生理盐水;罗利普兰组小鼠接受CMS处理,并每日腹腔注射1 mg/kg或5 mg/kg 罗利普兰,连续21天[3] - GABA含量变化:与非应激对照组小鼠相比,CMS暴露小鼠额叶皮质GABA含量降低32%;5 mg/kg 罗利普兰处理可显著逆转该降低效应:GABA含量较CMS溶剂组升高28%(通过高效液相色谱-HPLC荧光检测法测定)[3] - 抑郁样行为改善:在糖水偏好实验(衡量快感缺乏的指标)中,CMS小鼠糖水偏好度较非应激对照组降低45%;5 mg/kg 罗利普兰可将糖水偏好度恢复至非应激对照组的85%(1 mg/kg剂量无显著效应)[3] |
| 酶活实验 |
免疫沉淀和PDE测定[1]
如前所述,对四种PDE4类酶进行选择性免疫沉淀。如前所述,使用足够的抗血清来确保靶PDE4亚类的所有同工酶被选择性免疫沉淀;然后对这些进行PDE测定。PDE测定是通过修改两步Thompson和Appleman方法完成的。用新鲜细胞裂解物测定总细胞PDE活性和PDE3和PDE4组分的量。如前所述,使用1µM cAMP作为底物和10µM PDE3选择性抑制剂西洛司胺或PDE4选择性抑制剂rolipram/罗利普兰来测定PDE3和PDE4的总活性。 1. PDE4酶活性抑制实验: - 以纯化的重组人PDE4亚型(PDE4A、PDE4B、PDE4C、PDE4D)为酶源。反应体系(总体积200 μL)包含50 mM Tris-HCl(pH 7.5)、10 mM MgCl₂、1 mM EGTA、0.1 mg/mL牛血清白蛋白(BSA)、1 μM [³H]-cAMP(底物)及系列浓度的罗利普兰(0.01 nM至100 nM)。 - 加入酶(每个反应10 ng)启动反应,37°C孵育30分钟;煮沸反应体系2分钟终止反应,随后加入50 μL蛇毒磷酸二酯酶(水解剩余cAMP为腺苷),37°C继续孵育10分钟。 - 加入500 μL Dowex 1×8树脂(Cl⁻型)分离反应产物,离心后收集上清液(含[³H]-腺苷);采用液体闪烁计数器测定上清液放射性。 - 以无罗利普兰的溶剂对照组生成的[³H]-腺苷量为基准,计算酶活性抑制百分比;通过非线性回归将抑制曲线拟合至米氏方程,确定各PDE4亚型的Ki值[1] |
| 细胞实验 |
J774小鼠巨噬细胞在37°C、5%CO2气氛下在DMEM中培养,DMEM补充了含有10%热灭活FBS、100 U·mL-1青霉素、100μg·mL-1链霉素和250 ng·mL-1两性霉素B的谷氨酸-1。在实验中,细胞以每孔2×105个细胞的密度接种在24孔板上。在实验开始之前,细胞单层生长72小时Rolipram、IBMX和BIRB 796溶解在二甲亚砜(DMSO)中,8-Br-cAMP溶解在HBSS中。将LPS(10 ng·mL-1)或指定浓度的目标化合物或溶剂(DMSO,0.1%v/v)加入含有10%FBS和补充剂的新鲜培养基中的细胞中。将细胞进一步孵育指定时间。
通过细胞增殖试剂盒II(XTT)评估LPS和受试化学物质对细胞存活率的影响。没有观察到LPS或实验中使用的其他化学物质会引起细胞毒性。[2]
1. U937单核细胞信号实验: - 细胞培养:U937细胞在含10%胎牛血清(FBS)、100 U/mL青霉素、100 μg/mL链霉素的RPMI 1640培养基中,于37°C、5% CO₂培养箱中培养;将细胞以1×10⁶个/孔的密度接种于6孔板,贴壁培养24小时。 - cAMP检测:用罗利普兰(0.1 μM、1 μM、10 μM)或溶剂(0.1% DMSO)处理细胞15分钟;用5%三氯乙酸(TCA)提取细胞内cAMP,乙醚去除TCA后,采用竞争性放射免疫试剂盒定量cAMP水平,结果以总细胞蛋白(考马斯亮蓝法测定)归一化[1] - CREB和p38磷酸化Western blot分析:用罗利普兰(0.1 μM、1 μM、10 μM)预处理细胞20分钟,随后用LPS(100 ng/mL)刺激10分钟(检测p38)或不刺激(检测CREB);用含蛋白酶和磷酸酶抑制剂的RIPA缓冲液裂解细胞,12,000×g、4°C离心15分钟收集上清液;BCA法测定蛋白浓度,取30 μg蛋白进行10% SDS-PAGE电泳,转印至PVDF膜,用5%脱脂奶封闭1小时;一抗(抗p-CREB Ser133、抗总CREB、抗p-p38 Thr180/Tyr182、抗总p38)4°C孵育过夜,辣根过氧化物酶(HRP)标记二抗室温孵育1小时;ECL化学发光法显影,ImageJ软件定量条带强度,结果以磷酸化蛋白与总蛋白的比值表示[1] 2. RAW264.7巨噬细胞TNF-α和MKP-1实验: - 细胞培养:RAW264.7细胞在含10% FBS、100 U/mL青霉素、100 μg/mL链霉素的DMEM培养基中,于37°C、5% CO₂培养箱中培养;分别以5×10⁵个/孔(检测TNF-α)和1×10⁶个/孔(检测MKP-1)的密度接种于24孔板或6孔板。 - TNF-α检测:用罗利普兰(0.01 μM、0.1 μM、1 μM)或溶剂(0.1% DMSO)预处理细胞1小时,LPS(1 μg/mL)刺激6小时;收集细胞上清液,采用小鼠TNF-α ELISA试剂盒定量TNF-α水平,结果以活细胞数(台盼蓝排斥法测定)归一化[2] - MKP-1 RT-PCR:用1 μM 罗利普兰或溶剂处理细胞1小时、2小时或4小时;TRIzol试剂提取总RNA,逆转录酶和随机引物合成cDNA;采用MKP-1特异性引物(正向:5’-GCTGCTGATGGAGAAGATGG-3’;反向:5’-GGCTTGTCCTTGATGTCGTC-3’)和GAPDH特异性引物(内参)进行PCR;1.5%琼脂糖凝胶电泳分离PCR产物,溴化乙锭染色,ImageJ定量条带强度,MKP-1 mRNA水平以MKP-1与GAPDH的比值表示[2] |
| 动物实验 |
溶于100%聚乙二醇(PEG)中,配制成适当浓度;1 mL/kg;静脉注射
\n雄性哈特利豚鼠\n角叉菜胶诱导的爪水肿[2] \nC57BL/6小鼠(20–25 g)分为6只一组,在注射角叉菜胶前2小时,腹腔注射200 μL PBS或罗利普兰(100 mg·kg−1,溶于PBS)。在注射角叉菜胶前,小鼠经腹腔注射0.5 mg·kg−1美托咪定(Domitor® 1 mg·mL−1)和75 mg·kg−1氯胺酮(Ketalar® 10 mg·mL−1)麻醉。小鼠一侧后爪皮内注射30 μL角叉菜胶(1.5%,溶于生理盐水)。对侧后爪注射30 μL生理盐水作为对照。注射角叉菜胶前后3小时,使用体积描记器测量爪体积。水肿程度以爪体积随时间的变化来表示。实验结束后,麻醉小鼠经颈椎脱臼处死。小鼠称重后,分别放入单独的笼子中。为了使小鼠适应蔗糖溶液并获得蔗糖摄入量的基线数据,给小鼠喂食一瓶2%的蔗糖溶液。24小时后,取出溶液瓶并称重以测量液体摄入量。然后重新放入水瓶。再次测量1小时内的蔗糖摄入量。根据体重和蔗糖摄入量(24小时和1小时),将小鼠分为实验组和对照组(每组n=12)。除蔗糖消耗量外,体重也被用于区分动物,以尽量减少因体型差异导致的蔗糖摄入量变化。实验组动物接受为期6周的慢性轻度应激(CMS)。抗抑郁药治疗组动物从CMS第3周开始至第6周结束,每日口服(po)罗利普兰(rolipram)。对照组动物在6周期间未受干扰,仅在最后3周每日按计划口服蒸馏水,以模拟实验组动物的给药方式,此外还进行了清洁、喂食和称重等操作。 [3] \n药物给药和强迫游泳试验[3] \n在指定情况下,小鼠在慢性轻度应激(CMS)暴露的最后3周内,每日一次经口给予蒸馏水(对照组)或溶于蒸馏水的罗利普兰(0.1 mg/kg/天)。注射体积不超过20 ml/kg体重。该剂量是根据实验开始前的预实验确定的,预实验表明,该剂量给药后小鼠的各项指标均发生了变化。 \n1. 慢性轻度应激(CMS)小鼠模型和罗利普兰治疗: \n- 动物:使用雄性瑞士白化小鼠(25-30 g)。小鼠以5只/笼饲养于标准条件下(22±2°C,12小时光照/黑暗循环,早上7:00开灯),除应激处理期间外,可自由摄取食物和水。 \n - 慢性应激(CMS)诱导:将小鼠随机分为三组:非应激对照组、CMS溶剂组和CMS+罗利普兰组(1 mg/kg或5 mg/kg)。CMS每日进行,持续21天,应激源随机选择如下:食物剥夺(24小时)、饮水剥夺(12小时)、笼子倾斜45°(24小时)、夜间光照(12小时)、强迫冷水游泳(4°C,5分钟)和社会隔离(24小时)。每种应激源每周使用不超过两次,以避免小鼠产生适应性[3] \n - 药物制备和给药:将罗利普兰溶解于含0.1% DMSO的0.9%生理盐水(溶剂)中。 CMS + Rolipram 组的小鼠在每日应激程序开始前 30 分钟,每天腹腔注射一次 Rolipram,剂量为 1 mg/kg 或 5 mg/kg(注射体积:10 μL/g 体重)。CMS 载体组仅腹腔注射载体,非应激对照组未接受任何应激处理和注射[3] \n- 行为学测试(蔗糖偏好测试):在第 22 天(最后一次 CMS 处理后 1 天),进行蔗糖偏好测试。小鼠单独饲养,并放置两个瓶子:一个装有 1% 蔗糖溶液,另一个装有自来水。禁水 12 小时后,称量瓶子重量,然后允许小鼠自由取用瓶子 2 小时。蔗糖偏好性计算公式为(蔗糖摄入量/总液体摄入量)× 100% [3] \n - 组织采集和 GABA 测定:行为学测试结束后,小鼠通过颈椎脱臼处死。在冰上解剖前额皮质,称重,并在 0.1 M 高氯酸(1:10 w/v)中匀浆。匀浆液在 4°C 下以 12,000×g 离心 15 分钟,上清液经 0.22 μm 滤膜过滤。采用高效液相色谱-荧光检测法(激发波长:330 nm,发射波长:450 nm)分析 GABA 含量,以邻苯二甲醛为衍生化剂。GABA 水平根据组织重量进行标准化 [3] |
| 毒性/毒理 (Toxicokinetics/TK) |
小鼠口服LD50 >300 mg/kg,《生物与药物学报》,17(498),1994 [PMID:8069256]
1. 在CMS小鼠研究中,罗利普兰 以1 mg/kg和5 mg/kg的剂量(腹腔注射,每日一次,持续21天)未引起明显的毒性: - 体重:所有组别的小鼠体重均正常增加(21天内约增加2-3 g),罗利普兰治疗组与CMS对照组之间无显著差异[3] - 死亡率和临床症状:所有组均未观察到死亡。罗利普兰治疗组小鼠在整个治疗期间未出现异常行为(例如,共济失调、嗜睡、多动)或体征(例如,毛发蓬乱、腹泻)[3] |
| 参考文献 |
|
| 其他信息 |
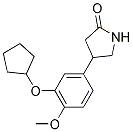
罗利普兰属于吡咯烷-2-酮类化合物,其结构为在4位带有3-(环戊氧基)-4-甲氧基苯基取代基的吡咯烷-2-酮。它是一种IV型特异性磷酸二酯酶(PDE4)抑制剂。它具有抗抑郁作用,并且是EC 3.1.4(磷酸二酯水解酶)抑制剂。
一种具有抗抑郁特性的磷酸二酯酶抑制剂。 一种具有抗抑郁特性的磷酸二酯酶4抑制剂。 此处显示U937单核细胞表达一系列PDE4,即cAMP特异性磷酸二酯酶(PDE)同工酶:长链同工酶PDE4A4、PDE4D5和PDE4D3,以及短链同工酶PDE4B2。这些同工酶提供了U937细胞总cAMP PDE活性的约76%。总PDE4A、PDE4B和PDE4D的比活性分别为0.63±0.09、8.8±0.2和34.4±2.9 pmol/min/mg蛋白。PDE4选择性抑制剂罗利普兰对免疫纯化的PDE4B和PDE4D活性的抑制作用相似,IC50值分别约为130 nM和240 nM。相比之下,罗利普兰对免疫纯化的PDE4A活性的抑制作用显著降低,IC50值约为3 nM。罗利普兰以剂量依赖的方式增加U937细胞中cAMP反应元件结合蛋白(CREB)的磷酸化水平,这表明CREB中同时存在高亲和力(IC50值约为1 nM)和低亲和力(IC50值约为120 nM)的组分。罗利普兰以剂量依赖的方式单调抑制干扰素-γ(IFN-γ)刺激的p38丝裂原活化蛋白(MAP)激酶的磷酸化,其IC50值约为290 nM。基于此,我们推测罗利普兰对PDE4A4的抑制作用参与了CREB磷酸化的调控,但并不影响IFN-γ刺激的p38 MAP激酶的磷酸化。 PDE4A4在用细菌脂多糖(LPS)或IFN-γ刺激U937细胞后被选择性激活,而沃特曼宁和雷帕霉素均可减弱这一过程。据推测,PDE4A4亚型参与了U937单核细胞中区室化的cAMP信号传导反应。[1] 背景和目的:3',5'-环核苷酸PDE4在多种炎症和免疫细胞中表达,PDE4催化cAMP水解为5'AMP,从而下调细胞内的cAMP信号传导。MAPK磷酸酶-1(MKP-1)是一种内源性p38 MAPK信号传导抑制因子,可限制炎症基因的表达和炎症反应。本研究探讨了PDE4抑制剂罗利普兰对MKP-1表达的影响,以及MKP-1是否参与罗利普兰的抗炎作用。实验方法:我们研究了罗利普兰对J774小鼠巨噬细胞系以及野生型(WT)和MKP-1(-/-)小鼠原代腹腔巨噬细胞(PM)中TNF生成的影响。此外,我们还研究了罗利普兰对WT和MKP-1(-/-)小鼠角叉菜胶诱导的爪部炎症的影响。主要结果:在J774细胞和PM中,罗利普兰、非选择性PDE抑制剂IBMX以及cAMP类似物8-Br-cAMP均能增强MKP-1的表达。PKA抑制剂可逆转罗利普兰对MKP-1 mRNA表达的增强作用。罗利普兰、IBMX 和 8-Br-cAMP 也抑制活化巨噬细胞中 TNF 的产生。相应地,罗利普兰抑制了野生型小鼠 PM 中 TNF 的产生,但有趣的是,它并未抑制 MKP-1(-/-) 小鼠 PM 中 TNF 的产生。此外,罗利普兰减轻了野生型小鼠角叉菜胶诱导的爪部炎症,但对 MKP-1(-/-) 小鼠没有这种作用。结论和意义:PDE4 抑制剂罗利普兰被发现能够增强 MKP-1 的表达,并且 MKP-1 至少部分介导了 PDE4 抑制的抗炎作用。结果表明,能够增强 MKP-1 表达和/或 MKP-1 活性的化合物具有作为新型抗炎药物的潜力。[2] 目的:探讨罗利普兰在抑郁症模型中对 GABA 水平的影响。材料与方法:本研究记录了磷酸二酯酶4型抑制剂罗利普兰对雄性小鼠前额皮质(FCx;大脑中负责情绪和认知控制的关键区域)中γ-氨基丁酸(GABA)含量的影响。这些小鼠暴露于慢性轻度应激(CMS)诱导的快感缺失(即对愉悦刺激的敏感性丧失或丧失)。结果:结果表明,以0.1 mg/kg/天的剂量口服罗利普兰(溶于蒸馏水)3周后,CMS诱导的快感缺失得到逆转。此外,罗利普兰显著减少了强迫游泳试验(FST)记录的长期行为改变中的不动时间。同时,罗利普兰治疗组小鼠在CMS诱导的快感缺失状态下,其前额皮质中的GABA含量显著增加。结论:本研究表明,在抑郁症动物模型中,GABA水平可能降低,而罗利普兰可逆转GABA水平并改善行为,这可能支持GABA能活性改变在情绪障碍中发挥作用的假设。这些作用可能补充罗利普兰的抗抑郁作用,罗利普兰最初是通过抑制4型磷酸二酯酶(PDE4)介导的,PDE4可增加环磷酸腺苷(cAMP)信号通路,从而发挥抑郁症的药物治疗作用。[3] 1. 作用机制:罗利普兰通过抑制PDE4发挥其细胞效应,PDE4可将cAMP水解为AMP。通过阻断PDE4,罗利普兰可增加细胞内cAMP水平,激活蛋白激酶A(PKA)。激活的PKA可磷酸化CREB,CREB是一种转录因子,可调节参与细胞存活和炎症的基因表达。此外,cAMP水平升高可抑制p38 MAPK磷酸化,从而降低促炎信号通路的激活[1] 2. 抗炎机制:罗利普兰的抗炎作用(例如,抑制TNF-α的产生)依赖于MKP-1。罗利普兰诱导的cAMP水平升高可上调MKP-1,MKP-1是一种磷酸酶,可使促炎性MAPK(例如p38、JNK)去磷酸化。这种去磷酸化作用抑制了驱动TNF-α表达的转录因子(例如NF-κB)的激活,从而减轻炎症[2] 3. 抗抑郁样作用:小鼠慢性应激诱导的抑郁样行为与GABA能神经传递减少(额叶皮质GABA含量降低)有关。罗利普兰通过增加 GABA 合成或减少 GABA 降解(本研究未明确测试该机制)来逆转这种缺陷,从而恢复正常的 GABA 能信号传导并改善快感缺失(通过蔗糖偏好性测量)。这表明罗利普兰可能具有作为抗抑郁药的潜力,尤其是在应激相关性抑郁症方面[3]。 4. 选择性说明:罗利普兰对 PDE4 亚型的 Ki 值差异(对 PDE4B 的亲和力最高,对 PDE4C 的亲和力最低)可能导致其组织特异性效应,因为 PDE4 亚型在免疫细胞(PDE4B、PDE4D)和中枢神经系统(PDE4A、PDE4D)中的表达存在差异[1]。 |
| 分子式 |
C16H21NO3
|
|---|---|
| 分子量 |
275.34
|
| 精确质量 |
275.152
|
| 元素分析 |
C, 69.79; H, 7.69; N, 5.09; O, 17.43
|
| CAS号 |
61413-54-5
|
| 相关CAS号 |
(R)-(-)-Rolipram;85416-75-7;(S)-(+)-Rolipram;85416-73-5
|
| PubChem CID |
5092
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.2±0.1 g/cm3
|
| 沸点 |
472.7±45.0 °C at 760 mmHg
|
| 熔点 |
127-133ºC
|
| 闪点 |
239.7±28.7 °C
|
| 蒸汽压 |
0.0±1.2 mmHg at 25°C
|
| 折射率 |
1.552
|
| LogP |
1.43
|
| tPSA |
47.56
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
4
|
| 重原子数目 |
20
|
| 分子复杂度/Complexity |
341
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
HJORMJIFDVBMOB-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H21NO3/c1-19-14-7-6-11(12-9-16(18)17-10-12)8-15(14)20-13-4-2-3-5-13/h6-8,12-13H,2-5,9-10H2,1H3,(H,17,18)
|
| 化学名 |
4-(3-(cyclopentyloxy)-4-methoxyphenyl)pyrrolidin-2-one
|
| 别名 |
ZK-62711; SB 95952; SB95952; rolipram; 61413-54-5; (+/-)-Rolipram; (R,S)-rolipram; ZK 62711; Rolipramum [Latin]; 4-(3-(cyclopentyloxy)-4-methoxyphenyl)pyrrolidin-2-one; Rolipramum; SB-95952; ME-3167; ZK-62711; ME3167; ZK62711; ME 3167; ZK 62711
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (9.08 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (9.08 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (9.08 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 30% PEG400+0.5% Tween80+5% propylene glycol:10 mg/L 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6319 mL | 18.1594 mL | 36.3187 mL | |
| 5 mM | 0.7264 mL | 3.6319 mL | 7.2637 mL | |
| 10 mM | 0.3632 mL | 1.8159 mL | 3.6319 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05522673 | Terminated Has Results | Drug: 11(R)-rolipram | Depression | National Institute of Mental Health (NIMH) |
February 8, 2023 | Phase 1 |
| NCT00011375 | Completed | Drug: Rolipram | Multiple Sclerosis | National Institute of Neurological Disorders and Stroke (NINDS) |
February 2001 | Phase 2 |
| NCT01215552 | Terminated | Drug: HT-0712 | Healthy Elderly Volunteers | Dart NeuroScience, LLC | September 2010 | Phase 1 |
| NCT00250172 | Completed | Drug: [C-11](R)-rolipram | Dosimetry Healthy |
National Institute of Mental Health (NIMH) |
October 31, 2005 | Phase 1 |
|
|---|
|
|