| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
描述:巴非替尼(Bafetinib,曾用名 INNO406;NS-187)是一种在研抗癌药物,最初由日本新药株式会社(Nippon Shinyaku)研发,后授权给 CytRx 公司。它是一种口服生物利用度高的双重 Bcr-Abl/Lyn 抑制剂,具有潜在的抗肿瘤活性。在无细胞实验中,它对 Bcr-Abl/Lyn 的抑制 IC50 值分别为 5.8 nM 和 19 nM。在 Bcr-Abl 阳性 KU812 小鼠模型中,巴非替尼显著抑制了肿瘤生长,并且在 20mg/kg/天的剂量下完全抑制了肿瘤生长,且未引起不良反应。
NS-187(也称为 CNS-9)是一种强效且选择性的双重 Bcr-Abl/Lyn 酪氨酸激酶抑制剂,旨在克服费城染色体阳性 (Ph+) 白血病对伊马替尼的耐药性。其体外活性是伊马替尼的 25-55 倍,体内抑制 Bcr-Abl 阳性肿瘤生长的效力至少是伊马替尼的 10 倍。NS-187 可抑制 13 种 Bcr-Abl 激酶结构域点突变体中的 12 种(T315I 除外),并且还能抑制 Lyn,而不影响 Src、Blk 或 Yes 的磷酸化。[1]
| 靶点 |
Abl (IC50 5.8 nM in an ELISA-based kinase assay; IC50 72 nM in purified Abl kinase domain assay); Lyn (IC50 19 nM); Src (IC50 1700 nM); Fyn (inhibited at 0.1 μM, exact IC50 not reported); Arg (inhibited at 0.1 μM); wild-type Bcr-Abl (cellular IC50 11 nM in K562, 22 nM in 293T, 63 nM in BaF3/wt for autophosphorylation); Bcr-Abl E255K mutant (cellular IC50 98 nM in 293T, 340 nM in BaF3); Bcr-Abl mutants M244V, G250E, Q252H, Y253F, E255K, E255V, F317L, M351T, E355G, F359V, H396P, F486S (in vitro IC50 ranging from 81 nM to 1400 nM, see Table 2); no inhibition of T315I (>10,000 nM). [1]
|
|---|---|
| 体外研究 (In Vitro) |
体外活性:巴非替尼可阻断野生型 Bcr-Abl 的自磷酸化及其下游激酶活性,在 K562 和 293T 细胞中的 IC50 值分别为 11 nM 和 22 nM。巴非替尼可有效抑制 Bcr-Abl 阳性细胞系(包括 K562、KU812 和 BaF3/wt 细胞)的生长,而对 Bcr-Abl 阴性 U937 细胞系的增殖无影响。此外,巴非替尼对 Bcr-Abl 点突变细胞系(例如 BaF3/E255K 细胞)表现出剂量依赖性的抗增殖作用。在 Bcr-Abl 阳性白血病细胞系中,巴非替尼通过阻断 Bcr-Abl 的磷酸化,诱导 caspase 介导和非 caspase 依赖性细胞死亡。 NS-187 可阻断 K562 细胞 (IC50 11 nM) 和 293T 细胞 (IC50 22 nM) 中野生型 Bcr-Abl 的自磷酸化,其效力分别是伊马替尼的 25 倍和 55 倍。[1] 它在远低于伊马替尼的浓度下即可抑制 K562 和 BaF3/wt 细胞中 CrkL 和 ERK 的磷酸化。 [1] 对于Bcr-Abl E255K突变体,NS-187可抑制BaF3/E255K细胞中的自身磷酸化(IC50 340 nM)并阻断CrkL/ERK磷酸化,而伊马替尼无效。[1] 对于T315I突变体,NS-187对BaF3/T315I细胞中的自身磷酸化或CrkL/ERK均无抑制作用(IC50 >10,000 nM)。 [1] NS-187 可抑制 Bcr-Abl 阳性细胞系 K562(效力强于伊马替尼,但未给出确切的 IC50 值)、KU812(效力强于伊马替尼)、BaF3/wt(效力更强)和 BaF3/E255K(浓度依赖性,效力强于伊马替尼)的增殖,但对 Bcr-Abl 阴性 U937 细胞或 BaF3/T315I 细胞无影响(浓度高达 10 μM)。[1] 它抑制 PDGFR 和 c-Kit 的磷酸化,IC50 值与伊马替尼相似(基于细胞的实验中,PDGFR IC50 为 56 nM,c-Kit IC50 为 840 nM),但在临床相关浓度下不抑制 EGFR(IC50 > 10,000 nM)。[1] |
| 体内研究 (In Vivo) |
在Bcr-Abl阳性KU812小鼠模型中,巴非替尼(0.2 mg/kg/天)可显著抑制肿瘤生长,20 mg/kg/天剂量可完全抑制肿瘤生长且无不良反应。对于Balb/c小鼠,巴非替尼的最大耐受剂量为200 mg/kg/天,生物利用度(BA)为32%。在携带 Ba/F3/wt bcr-ablGFP、Ba/F3/Q252H 或 Ba/F3/M351T 细胞的中枢神经系统 (CNS) 白血病模型中,巴非替尼 (60 mg/kg) 和环孢素 A (CsA) (50 mg/kg) 联合治疗比单独使用巴非替尼或 CsA 更能显著抑制脑内白血病细胞的生长。
在 Balb/c-nu/nu 小鼠的 KU812 皮下异种移植模型中,口服 NS-187,剂量为 0.2 mg/kg/d(每日两次),可显著抑制肿瘤生长;剂量为 20 mg/kg/d 时,可完全抑制肿瘤生长且无不良反应;而伊马替尼需要 200 mg/kg/d 才能完全抑制肿瘤生长。NS-187 的效力至少是伊马替尼的 10 倍。 [1] 在 BaF3/wt 静脉注射白血病模型中,NS-187(6-200 mg/kg/d,每日两次口服,持续 11 天)与载体相比,以剂量依赖的方式延长了生存期;伊马替尼(400 mg/kg/d)几乎没有效果(所有小鼠均在第 25 天死亡)。[1] 在 BaF3/E255K(伊马替尼耐药突变体)静脉注射模型中,NS-187(120 mg/kg/d,每日两次口服,持续 26 天)与载体和伊马替尼(200 mg/kg/d)相比,显著延长了生存期。[1] |
| 酶活实验 |
利用杆状病毒在Sf9细胞中表达,构建了纯化的Abl激酶结构域(野生型和13个点突变体,氨基酸229-515),并经Q-Sepharose和HisTrap柱纯化。激酶活性测定在25 μL反应体系中进行,该体系包含250 μM肽底物、[γ-33P]ATP(740 Bq/μL)和20 μM非标记ATP,采用SignaTECT蛋白酪氨酸激酶测定系统。每种Bcr-Abl激酶的使用浓度均为10 nM。[1] Abl、Src和Lyn的激酶活性测定采用ELISA试剂盒进行。[1] 使用KinaseProfiler检测了NS-187对79种酪氨酸激酶的抑制作用。在 0.1 μM 时,NS-187 抑制 Abl、Arg、Fyn 和 Lyn。[1]
|
| 细胞实验 |
细胞系(K562、BaF3/wt、BaF3/E255K、BaF3/T315I、KU812、U937、NHDF、NCI-H526、A431)在含10%胎牛血清(FBS)、2 mM L-谷氨酰胺和适当补充剂的RPMI 1640或DMEM培养基中,于37°C、5% CO2条件下培养。为抑制细胞内酪氨酸磷酸化,将细胞用系列稀释的化合物处理1.5小时。随后,将血清饥饿的NHDF、NCI-H526和A431细胞用PDGF(50 ng/mL)、SCF(100 ng/mL)或EGF(100 ng/mL)刺激10分钟。用RIPA裂解缓冲液裂解细胞,取等量裂解液蛋白进行Western blotting分析,使用抗磷酸酪氨酸抗体(PY20-HRP)以及抗c-Abl、c-Kit、CrkL、EGFR、ERK1或PDGFR抗体。[1]
细胞增殖实验中,将细胞以三复孔接种于96孔板(1×10^3或5×10^3个细胞/孔),并用系列稀释的化合物孵育3天。采用MTT法检测细胞增殖,并通过逻辑曲线拟合计算IC50值。[1] |
| 动物实验 |
巴非替尼溶于0.5%甲基纤维素溶液中;≤20 mg/kg/天;口服。KU812异种移植瘤模型通过将KU812细胞皮下注射到Balb/c-nu/nu雌性小鼠右侧腹部建立。
皮下异种移植瘤模型:将2.5×10^7个KU812细胞皮下注射到8周龄Balb/c-nu/nu雌性小鼠右侧腹部。接种7天后,将小鼠随机分为5组,分别灌胃给予NS-187、伊马替尼或载体(0.5%甲基纤维素),每日两次,连续10天。每周至少两次用游标卡尺测量肿瘤大小,并计算公式为(d^2 × D)/2/1000 cm^3。 [1] 对于 BaF3/wt 白血病模型:将 1×10^6 个 BaF3/wt 细胞经尾静脉注射到 Balb/c-nu/nu 小鼠体内。次日,将小鼠随机分为 7 组,并连续 11 天,每天两次口服给予化合物或载体。采用 Kaplan-Meier 法和 log-rank 检验分析生存率。[1] 对于 BaF3/E255K 白血病模型:将 5×10^4 个 BaF3/E255K 细胞经尾静脉注射到 Balb/c 小鼠体内。次日,将小鼠随机分为 3 组,并连续 26 天,每天两次口服给予化合物或载体。生存分析方法同上。[1] |
| 药代性质 (ADME/PK) |
在对Balb/c小鼠进行初步药代动力学研究时,以30 mg/kg的剂量口服NS-187,结果如下:Tmax = 2小时;Cmax = 661 ng/mL;AUC0-∞ = 2294 ng·h/mL;T1/2 = 1.0小时;生物利用度(BA) = 32%。[1] 在Balb/c或Balb/c-nu/nu小鼠中,NS-187的最大耐受剂量(MTD)为200 mg/kg/d(100 mg/kg,每日两次)。在此剂量下,估计Cmax为2226 ng/mL (4.0 μM)。[1]
|
| 毒性/毒理 (Toxicokinetics/TK) |
NS-187在小鼠中耐受性良好。剂量高达200 mg/kg/d(100 mg/kg,每日两次)时,接受治疗的荷瘤小鼠的体重与未接受治疗的小鼠的体重没有显著差异[1]。
|
| 参考文献 | |
| 其他信息 |
巴非替尼是一种联芳基化合物。巴非替尼已在临床试验中用于治疗多种癌症,包括成人胶质瘤、成人混合型胶质瘤、成人胶质母细胞瘤、慢性粒细胞白血病和急性淋巴细胞白血病。巴非替尼是一种口服有效的2-苯氨基嘧啶衍生物,具有潜在的抗肿瘤活性。INNO-406 特异性结合并抑制 Bcr/Abl 融合蛋白酪氨酸激酶,该酶是由与慢性粒细胞白血病 (CML) 相关的费城染色体易位产生的异常酶。此外,该药物还抑制 Src 家族成员 Lyn 酪氨酸激酶,该激酶在伊马替尼耐药的 CML 细胞和多种实体瘤细胞中表达上调。INNO-406 对这些特定酪氨酸激酶的抑制作用可减少细胞增殖并诱导细胞凋亡。相当一部分慢性粒细胞白血病 (CML) 患者对伊马替尼耐药,有时是由于 Bcr/Abl 融合蛋白激酶结构域的点突变所致。INNO-406 具有双重抑制活性,已被证实能够克服这种耐药性,是治疗伊马替尼耐药性 CML 的有效药物。
NS-187 是一种 2-苯氨基嘧啶类化合物。它与 Abl 的结合模式与伊马替尼类似,通过与 Met318、Thr315、Glu286 和 Asp381 形成氢键而稳定。该化合物抑制 Lyn,但不抑制 Src 或 Yes,这是因为其含有 Gln252(而 Src/Yes 中为 Cys252),Gln252 促进了氢键的形成和 P 环的诱导契合。 [1] NS-187不抑制T315I Bcr-Abl突变体,因为该突变破坏了一个关键的氢键,并从空间上阻断了结合位点。[1] 它有望成为治疗伊马替尼耐药的费城染色体阳性白血病(CML和Ph+ ALL)的新型药物。[1] |
| 分子式 |
C30H31F3N8O
|
|---|---|
| 分子量 |
576.62
|
| 精确质量 |
576.257
|
| CAS号 |
859212-16-1
|
| 相关CAS号 |
859212-16-1;887650-05-7;
|
| PubChem CID |
11387605
|
| 外观&性状 |
Light yellow to yellow solid powder
|
| 密度 |
1.4±0.1 g/cm3
|
| 熔点 |
166-168°C
|
| 折射率 |
1.640
|
| LogP |
3.03
|
| tPSA |
99.17
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
11
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
42
|
| 分子复杂度/Complexity |
872
|
| 定义原子立体中心数目 |
1
|
| SMILES |
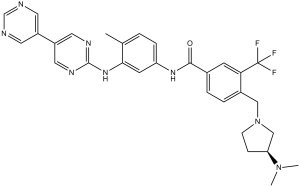
CC1=C(C=C(C=C1)NC(=O)C2=CC(=C(C=C2)CN3CC[C@@H](C3)N(C)C)C(F)(F)F)NC4=NC=CC(=N4)C5=CN=CN=C5
|
| InChi Key |
ZGBAJMQHJDFTQJ-DEOSSOPVSA-N
|
| InChi Code |
InChI=1S/C30H31F3N8O/c1-19-4-7-23(13-27(19)39-29-36-10-8-26(38-29)22-14-34-18-35-15-22)37-28(42)20-5-6-21(25(12-20)30(31,32)33)16-41-11-9-24(17-41)40(2)3/h4-8,10,12-15,18,24H,9,11,16-17H2,1-3H3,(H,37,42)(H,36,38,39)/t24-/m0/s1
|
| 化学名 |
(S)-N-(3-([4,5'-bipyrimidin]-2-ylamino)-4-methylphenyl)-4-((3-(dimethylamino)pyrrolidin-1-yl)methyl)-3-(trifluoromethyl)benzamide
|
| 别名 |
INNO-406; INNO 406; NS187; NS187; INNO406;NS-187; NS 187
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (4.34 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (4.34 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL 澄清 DMSO 储备液添加到 900 μL 玉米油中并混合均匀。 View More
配方 3 中的溶解度: 0.5% methylcellulose+0.2% Tween 80: 30 mg/mL 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7342 mL | 8.6712 mL | 17.3424 mL | |
| 5 mM | 0.3468 mL | 1.7342 mL | 3.4685 mL | |
| 10 mM | 0.1734 mL | 0.8671 mL | 1.7342 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01215799 | Completed | Drug: Bafetinib | Hormone Refractory Prostate Cancer | CytRx | August 2010 | Phase 2 |
| NCT01144260 | Completed | Drug: bafetinib | B-Cell Chronic Lymphocytic Leukemia | CytRx | June 2010 | Phase 2 |
| NCT01234740 | Completed | Drug: bafetinib Procedure: microdialysis |
Adult Anaplastic Astrocytoma Adult Anaplastic Ependymoma |
City of Hope Medical Center | December 2010 | Phase 1 |
| NCT00352677 | Completed | Drug: INNO-406 | Chronic Myeloid Leukemia Acute Lymphocytic Leukemia |
CytRx | July 2006 | Phase 1 |
|
|
|