| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
抗生素头孢泊肟(头孢泊肟酸)对革兰氏阴性厌氧菌(Bacteroidetesceae)的最低抑菌浓度(MIC)为0.125-4 mg/L。观察到头孢泊肟抑制小韦荣球菌的 MIC 值为 0.25-8 mg/L。头孢泊肟可抑制糖酵解链球菌、微小消化链球菌和布氏瘤胃球菌,最低抑制浓度 (MIC) 低于每升 2 毫克 [1]。头孢泊肟或头孢泊肟酸可抑制肺炎链球菌和化脓性链球菌的菌群。菌落形成单位[2]。
|
|---|---|
| 体内研究 (In Vivo) |
在治疗方面,小鼠对头孢菌素(2.5 至 50 mg/kg;口服;每 8 小时一次;连续 48 小时)反应良好 [3]。
|
| 动物实验 |
动物/疾病模型:雌性瑞士CD1小鼠[3]
剂量:2.5、5、10、25、40和50 mg/kg 给药途径:口服; 给药途径:口服。每8小时一次;持续48小时 实验结果:疗效值>350。 |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
头孢泊肟酯是一种前药,经胃肠道吸收后脱酯化为活性代谢物头孢泊肟。空腹受试者口服100 mg头孢泊肟酯后,约50%的给药剂量被全身吸收。 在推荐剂量范围(100至400 mg)内,约29%至33%的给药剂量在12小时内以原形经尿液排出。 生物半衰期 2.09至2.84小时 |
| 毒性/毒理 (Toxicokinetics/TK) |
妊娠期和哺乳期用药
◉ 哺乳期用药概述 现有信息有限,表明头孢泊肟在乳汁中的浓度较低,预计不会对母乳喂养的婴儿造成任何不良影响。有报道称,头孢菌素类药物偶尔会扰乱婴儿的胃肠道菌群,导致腹泻或鹅口疮,但这些影响尚未得到充分评估。哺乳期妇女可以安全使用头孢泊肟。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 一名40岁非妊娠女性服用头孢泊肟200毫克,每日两次,持续2天后出现高泌乳素血症和双侧溢乳。停药七天后,溢乳症状消失,血清催乳素水平显著下降至正常范围。一个月后,催乳素水平进一步下降。由于未发现其他病因,作者认为溢乳和高催乳素血症很可能是由头孢泊肟引起的。 一名22岁女性,服用缓释文拉法辛150毫克/天,持续3个月。两周前,她开始服用头孢泊肟200毫克/天,连续14天。之后,她出现双侧乳房胀痛和溢乳症状,持续3天。实验室检查和头部CT结果均正常,仅碱性磷酸酶轻度升高,血清催乳素水平升高。两周内,她的溢乳症状开始减轻,三周后消失,期间文拉法辛的剂量没有改变。她的血清催乳素水平也恢复正常。作者认为她的症状和高催乳素血症可能是由头孢泊肟引起的。 已建立泌乳的母亲的催乳素水平可能不会影响其母乳喂养能力。 血清蛋白结合率 22%至33%,血浆蛋白结合率21%至29%。 |
| 参考文献 |
[1]. Werner H, et, al. Comparative in vitro activity of cefpodoxime against anaerobes other than Bacteroides fragilis. Infection. 1991 Sep-Oct;19(5):377-9.
[2]. Valentini S, et, al. In-vitro evaluation of cefpodoxime. J Antimicrob Chemother. 1994 Mar;33(3):495-508. [3]. Pérez-Trallero E, et, al. Prediction of in-vivo efficacy by in-vitro early bactericidal activity with oral beta-lactams, in a dose-ranging immunocompetent mouse sepsis model, using strains of Streptococcus pneumoniae with decreasing susceptibilities to penicillin. J Chemother. 2001 Apr;13(2):118-25. |
| 其他信息 |
头孢泊肟是一种第三代头孢菌素类抗生素,其头孢烯骨架的3位和7位分别带有甲氧基甲基和(2Z)-2-(2-氨基-1,3-噻唑-4-基)-2-(甲氧基亚氨基)乙酰氨基取代基。它以酯类前药的形式口服,用于治疗急性中耳炎、咽炎和鼻窦炎。它是一种抗菌药物。它属于头孢菌素类和羧酸类化合物。
头孢泊肟是一种口服的第三代头孢菌素类抗生素,对大多数革兰氏阳性菌和革兰氏阴性菌均有效。头孢泊肟酯常用于治疗急性中耳炎、咽炎和鼻窦炎,它是一种前药,经肠黏膜吸收后脱酯化为头孢泊肟。头孢泊肟是一种头孢菌素类抗菌药物。头孢泊肟是第三代半合成头孢菌素,也是一种具有杀菌活性的β-内酰胺类抗生素。头孢泊肟的作用机制是与位于细菌细胞质膜上的青霉素结合蛋白(PBPs)结合。结合后,转肽酶活性受到抑制,从而阻止五甘氨酸桥与五肽第四个氨基酸残基的交联,进而阻断肽聚糖链的合成。因此,头孢泊肟抑制细菌隔膜和细胞壁的合成。 头孢泊肟是一种第三代头孢菌素类抗生素。其头孢菌素核心的C-3位含有一个甲氧基。 另见:头孢泊肟酯(活性成分);头孢泊肟钠(活性成分)。 适应症 适用于治疗由指定微生物的敏感菌株引起的轻度至中度感染。 FDA标签 作用机制 头孢泊肟对多种革兰氏阳性菌和革兰氏阴性菌均有效。头孢泊肟在β-内酰胺酶存在下稳定。因此,许多由于产生β-内酰胺酶而对青霉素和头孢菌素产生耐药性的微生物可能对头孢泊肟敏感。头孢泊肟会被某些超广谱β-内酰胺酶灭活。头孢泊肟的杀菌活性源于其对细胞壁合成的抑制作用。头孢泊肟的活性代谢物优先与青霉素结合蛋白3结合,从而抑制肽聚糖(细菌细胞壁的主要成分)的合成。 药效学 头孢泊肟对大多数革兰氏阳性菌和革兰氏阴性菌均有效,但对铜绿假单胞菌、肠球菌和脆弱拟杆菌无效。 |
| 分子式 |
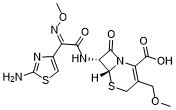
C15H17N5O6S2
|
|---|---|
| 分子量 |
427.46
|
| 精确质量 |
427.062
|
| CAS号 |
80210-62-4
|
| 相关CAS号 |
Cefpodoxime-d3;2477791-28-7
|
| PubChem CID |
6335986
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.8±0.1 g/cm3
|
| 熔点 |
200-202ºC
|
| 折射率 |
1.780
|
| LogP |
0.94
|
| tPSA |
209.98
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
11
|
| 可旋转键数目(RBC) |
7
|
| 重原子数目 |
28
|
| 分子复杂度/Complexity |
744
|
| 定义原子立体中心数目 |
2
|
| SMILES |
C(C1=C(COC)CS[C@@H]2[C@@H](C(N12)=O)NC(=O)/C(/C1=CSC(N)=N1)=N\OC)(=O)O
|
| InChi Key |
WYUSVOMTXWRGEK-HBWVYFAYSA-N
|
| InChi Code |
InChI=1S/C15H17N5O6S2/c1-25-3-6-4-27-13-9(12(22)20(13)10(6)14(23)24)18-11(21)8(19-26-2)7-5-28-15(16)17-7/h5,9,13H,3-4H2,1-2H3,(H2,16,17)(H,18,21)(H,23,24)/b19-8-/t9-,13-/m1/s1
|
| 化学名 |
5-Thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic acid, 7-(((2-amino-4-thiazolyl)(methoxyimino)acetyl)amino)-3-(methoxymethyl)-8-oxo-, (6R-(6alpha,7beta(Z)))-
|
| 别名 |
U76253AR-3746 U-76253AR3746 R 3763 U 76253AR-3763 R3763
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~250 mg/mL (~584.85 mM)
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3394 mL | 11.6970 mL | 23.3940 mL | |
| 5 mM | 0.4679 mL | 2.3394 mL | 4.6788 mL | |
| 10 mM | 0.2339 mL | 1.1697 mL | 2.3394 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。