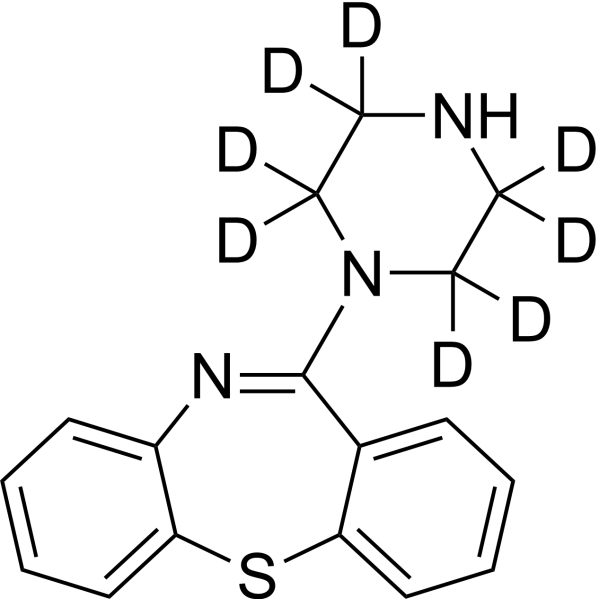
11-(哌嗪-1-基)二苯并[b,f][1,4]硫氮杂卓-d8 是 11-(哌嗪-1-基)二苯并[b,f][1,4]硫氮杂卓的氘标记形式。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Postoperative Pain Management in Rhinoplasty
CTID: NCT03584152
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-27
Can Ibuprofen Delay Ovulation in Natural Cycle-IVF?
CTID: NCT02571543
Phase: Phase 2 Status: Completed
Date: 2024-11-19
Non-Steroidal Anti-Inflammatory Drugs in Acute Myocarditis
CTID: NCT06686862
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-11-13
Prescribing Lorazepam for IUD Insertion: Pilot Feasibility Study
CTID: NCT06496854
Phase: Phase 2 Status: Not yet recruiting
Date: 2024-11-08
Comparing Analgesic Regimen Effectiveness and Safety for Surgery for Kids Trial
CTID: NCT06671002
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-11-04
View More
Pain Control Differences Between Oxycodone and Ibuprofen in Children With Isolated Forearm Injuries
CTID: NCT04523623
Phase: N/A Status: Withdrawn
Date: 2024-10-26
Paracetamol And Ibuprofen/Indomethacin in Closing Patent Ductus Arteriosus
CTID: NCT03648437
Phase: Phase 1 Status: Terminated
Date: 2024-10-18
Effect of Photobiomodulation to Reduce Post-operative Pain After Endodontic Surgery
CTID: NCT05935306
Phase: N/A Status: Recruiting
Date: 2024-10-10
Acute Low Back Pain. Topical Diclofenac and Oral Ibuprofen.
CTID: NCT04611529
Phase: Phase 4 Status: Completed
Date: 2024-10-09
NSAID Use After Robotic Partial Nephrectomy
CTID: NCT05842044
Phase: Phase 2 Status: Recruiting
Date: 2024-10-08
Preemptive Ibuprofen Effects on Pain Perception Following Extraction and Bone Graft
CTID: NCT05919745
Phase: Phase 4 Status: Recruiting
Date: 2024-09-27
Intravenous Ibuprofen Postoperative Analgesia After Abdominal Hysterectomy
CTID: NCT05750264
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-09-19
The Effectiveness of Paracetamol Versus Ibuprofen in Management of Patent Ductus Arteriosus in Preterm Neonates
CTID: NCT06601114
Phase: N/A Status: Active, not recruiting
Date: 2024-09-19
Comparison of IV Nalbuphine Versus Ibuprofen for Postoperative Pain Control in Cesarean Section
CTID: NCT06594224
Phase: N/A Status: Completed
Date: 2024-09-19
Efficacy of Opioid-limiting Pain Management Protocol in Men Undergoing Urethroplasty
CTID: NCT03859024
Phase: Phase 4 Status: Completed
Date: 2024-09-19
Impact of Anti-inflammatory Medications in Patients With Elevated Serum Prostate-specific Antigen
CTID: NCT05512754
Phase: Phase 4 Status: Recruiting
Date: 2024-09-19
Non-Steroidal or Opioid Analgesia Use for Children With Musculoskeletal Injuries
CTID: NCT03767933
Phase: Phase 2 Status: Completed
Date: 2024-09-19
Post-operative Course of Dexamethasone to Reduce Tonsillectomy Morbidity
CTID: NCT04879823
Phase: Phase 3 Status: Terminated
Date: 2024-09-05
Effect of NSAID Use on Pain and Opioid Consumption Following Distal Radius Fracture
CTID: NCT03749616
Phase: Phase 4 Status: Terminated
Date: 2024-09-05
Pain Management Following Dental Treatment Under General Anesthesia
CTID: NCT06574516
Phase: N/A Status: Completed
Date: 2024-09-04
Management Of Pain After Cesarean Trial
CTID: NCT03929640
Phase: Phase 3 Status: Terminated
Date: 2024-09-04
Non-Opioid Pain Medications After Intracapsular Adenotonsillectomy
CTID: NCT04791761
Phase: Phase 1/Phase 2 Status: Terminated
Date: 2024-08-30
Comparing the Effect of Different Intracanal Dressing on Failed Root Canal Treated Cases on Periapical Healing
CTID: NCT06342830
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-08-20
Pain Multidisciplinar Intervention in Major Trauma Patients
CTID: NCT05355441
Phase: Status: Recruiting
Date: 2024-08-20
Approach to Ankle Sprains in the Emergency Department
CTID: NCT06563271
Phase: Phase 4 Status: Completed
Date: 2024-08-20
Variability in Analgesic Response to Ibuprofen
CTID: NCT06539741
Phase: Status: Not yet recruiting
Date: 2024-08-06
An Evaluation of Pain Outcomes of Ketorolac Administration in Children Undergoing Circumcision
CTID: NCT04646967
Phase: Phase 2 Status: Completed
Date: 2024-08-02
Efficacy and Safety in the Combination of Ibuprofen / Loratadine Versus Ibuprofen Versus Loratadine
CTID: NCT06531707
Phase: Phase 3 Status: Recruiting
Date: 2024-08-02
Comparing the Difference in Pain Control in the Pediatric General Surgery Population: to Alternate or Combine Acetaminophen and Ibuprofen?
CTID: NCT06505148
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-07-22
Controlled Trial to Determine Most Effective Post-Operative Analgesia After Third Molar Extraction
CTID: NCT06484439
PhaseEarly Phase 1 Status: Completed
Date: 2024-07-10
A Trial Investigating the Pain-relieving Properties of Lu AG06474 in Healthy Adult Participants
CTID: NCT06077786
Phase: Phase 1 Status: Completed
Date: 2024-06-28
Ibuprofen Use on Post-operative Pain Following Cholecystectomy
CTID: NCT06478758
Phase: Phase 4 Status: Completed
Date: 2024-06-27
Canadian National PDA Treatment Study
CTID: NCT04347720
Phase: Status: Completed
Date: 2024-06-21
Selective Early Medical Treatment of Patent Ductus Arteriosus in Extremely Low Gestational Age Infants: A Pilot RCT
CTID: NCT05011149
Phase: Phase 3 Status: Recruiting
Date: 2024-06-21
Nonopioid Pain Control Regimen After Arthroscopic Hip Procedures
CTID: NCT05076110
Phase: Phase 4 Status: Recruiting
Date: 2024-06-18
Open-Label Placebo for Non-Specific Pain in the ED
CTID: NCT06408519
Phase: N/A Status: Recruiting
Date: 2024-06-14
Reduced Opioid Prescription After Laparoscopic Hysterectomy
CTID: NCT05548582
Phase: N/A Status: Active, not recruiting
Date: 2024-06-04
Multimodal Intervention for Cachexia in Advanced Cancer Patients Undergoing Chemotherapy
CTID: NCT02330926
Phase: Phase 3 Status: Completed
Date: 2024-06-04
Efficacy of 300 mg Ibuprofen Prolonged-Release Tablets for the Treatment of Pain After Surgical Removal of Impacted Third Molars
CTID: NCT03785756
Phase: Phase 3 Status: Completed
Date: 2024-06-03
Opioid Use After Laparoscopic Salpingectomy
CTID: NCT06434233
Phase: N/A Status: Recruiting
Date: 2024-05-30
A Study to Evaluate the Effect of Contraceptive Vaginal Rings on Primary Dysmenorrhea (P08257/MK-8175A/MK-8342B-057)
CTID: NCT01670656
Phase: Phase 2 Status: Completed
Date: 2024-05-28
Efficacy and Safety of Etonogestrel + 17β-Estradiol Vaginal Ring (MK-8342B) in the Treatment of Women With Primary Dysmenorrhea (MK-8342B-060)
CTID: NCT02668822
Phase: Phase 3 Status: Terminated
Date: 2024-05-28
Efficacy and Safety of Etonogestrel + 17β-Estradiol Vaginal Ring (MK-8342B) in Women With Primary Dysmenorrhea (With Optional Extension) (MK-8342B-059)
CTID: NCT02668783
Phase: Phase 3 Status: Terminated
Date: 2024-05-28
Ibuprofen With or Without Dexamethasone for Acute Radicular Low Back Pain.
CTID: NCT05721027
Phase: Phase 4 Status: Recruiting
Date: 2024-05-16
Pentoxifylline and Lumbar Radiculopathy
CTID: NCT03060434
Phase: Phase 4 Status: Active, not recruiting
Date: 2024-05-14
A Study to Investigate the Analgesic Efficacy of Ibuprofen Alone and Ibuprofen Plus Hyoscine-n- Butyl Bromide in Reducing Pain of Outpatient Hysteroscopy
CTID: NCT06398054
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-05-10
Treating Postictal Symptoms Using Ibuprofen and Nifedipine
CTID: NCT03949478
Phase: Phase 2 Status: Recruiting
Date: 2024-05-09
Safety and Efficacy of Pre-incisional Intravenous Ibuprofen to Reduce Postoperative Pain and Opioid Dependence After Posterior Cervical or Lumbar Instrumented Spine Surgery
CTID: NCT02276911
Phase: Phase 2 Status: Terminated
Date: 2024-04-19
Preeclampsia And Nonsteroidal Drugs for Analgesia: a Randomized Non Inferiority Trial
CTID: NCT03978767
Phase: Phase 2 Status: Recruiting
Date: 2024-04-09
Ibuprofen to Decrease Opioid Use and Post-operative Pain Following Unilateral Inguinal Herniorrhaphy
CTID: NCT02929589
Phase: Phase 3 Status: Terminated
Date: 2024-03-26
Adjunctive Acetylsalicylic Acid and Ibuprofen for Tuberculosis
CTID: NCT04575519
Phase: Phase 2 Status: Recruiting
Date: 2024-03-25
Efficacy of Lidocaine Patch in Acute Musculoskeletal Pain in the Emergency Department
CTID: NCT03571737
Phase: Phase 3 Status: Completed
Date: 2024-03-25
The Lacosamide's Effect on Calcitonin Gene-related Peptide in Migraine Patients
CTID: NCT05632133
Phase: Phase 3 Status: Completed
Date: 2024-03-21
Effects of Acute Exercise and Ibuprofen on Symptoms, Immunity, and Neural Circuits in Bipolar Depression
CTID: NCT06088732
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-03-13
Aggressive Antipyretics for Fever Reduction in CNS Malaria
CTID: NCT03399318
Phase: Phase 2 Status: Completed
Date: 2024-03-05
Ibuprofen Versus Ketorolac For Postoperative Pain Relief After Cesarean Section
CTID: NCT05907993
Phase: Phase 4 Status: Completed
Date: 2024-02-28
Ibuprofen With or Without Acetaminophen for Low Back Pain
CTID: NCT03554018
Phase: Phase 3 Status: Completed
Date: 2024-02-28
Opioids Versus Non-Opioids Postoperative After Knee Arthroscopic Surgery
CTID: NCT03858231
Phase: Phase 4 Status: Recruiting
Date: 2024-02-26
Paracetamol / Ibuprofen for Postpartum Pain in the Early Postpartum Period
CTID: NCT04653506
Phase: N/A Status: Completed
Date: 2024-02-20
Ibuprofen and Biomarkers of Acute Kidney Injury After Running in the Heat
CTID: NCT06247462
Phase: Phase 1 Status: Completed
Date: 2024-02-08
Efficacy of a Standardized Centella Asiatica Extract in Patients With Temporomandibular Disorder
CTID: NCT06231212
Phase: Phase 2/Phase 3 Status: Completed
Date: 2024-02-07
Efficacy of Metamizole Versus Ibuprofen and a Short Educational Intervention Versus Standard Care in Acute Low Back Pain
CTID: NCT04111315
Phase: Phase 4 Status: Recruiting
Date: 2024-02-07
Gynecologic Endoscopic Surgery of Female Motion Sickness Patients
CTID: NCT06232785
Phase: Phase 4 Status: Completed
Date: 2024-01-31
PDA Treatment With Ibuprofen and Changes in Tissue Oxygenation.
CTID: NCT05325177
Phase: Phase 4 Status: Recruiting
Date: 2024-01-30
Outcomes Associated With Application of a Normothermia Protocol in Patients With Severe Neurological Insult and Fever
CTID: NCT00890604
Phase: N/A Status: Completed
Date: 2024-01-30
Opioid-Free Pain Protocol After Shoulder Arthroplasty
CTID: NCT05488847
Phase: Phase 4 Status: Recruiting
Date: 2024-01-12
SCHF Post-Op Study Between Opioid and Non-Opioid Pain Management
CTID: NCT06187584
Phase: Phase 4 Status: Enrolling by invitation
Date: 2024-01-10
Comparison of the Efficacy of Paracetamol and Ibuprofen in the Management of Fever in Sepsis Patients
CTID: NCT06061575
Phase: Phase 4 Status: Recruiting
Date: 2024-01-08
Vaginal Postpartum Pain Management Protocol Comparison
CTID: NCT04087317
Phase: N/A Status: Completed
Date: 2024-01-03
Hypertension In Postpartum Preeclampsia Study
CTID: NCT03011567
Phase: N/A Status: Completed
Date: 2024-01-03
Effect of Acetaminophen Versus Ibuprofen in Treating Recurrent Apthous Ulcers in Pediatric Celiac Disease
CTID: NCT06149507
Phase: Phase 4 Status: Not yet recruiting
Date: 2023-12-28
Flotetuzumab for the Treatment of Relapsed or Refractory Advanced CD123-Positive Hematological Malignancies
CTID: NCT04681105
Phase: Phase 1 Status: Active, not recruiting
Date: 2023-12-20
Ibuprofen Versus Indomethacin Following Emergent Cerclage Placement
CTID: NCT04726085
Phase: N/A Status: Enrolling by invitation
Date: 2023-12-05
Comparision Between Paracetamol and Ibuprofen in Closure of Patent Ductus Arteriosus
CTID: NCT06152796
Phase: Phase 2 Status: Not yet recruiting
Date: 2023-12-01
Efficacy of Combined Ibuprofen and Acetaminophen Therapy Versus Ibuprofen Alone Versus Placebo Alone for Pain Management
CTID: NCT04059172
PhaseEarly Phase 1 Status: Recruiting
Date: 2023-11-30
The Effects of NSAIDs on Bone Metabolism Following Exercise
CTID: NCT05512013
Phase: Phase 1 Status: Completed
Date: 2023-11-29
A Multi-surgery Assessment of ZYNRELEF (HTX-011), AMAZE.
CTID: NCT06109415
Phase: Phase 4 Status: Completed
Date: 2023-10-31
A Multi-surgery Assessment of ZYNRELEF (HTX-011), AMAZE
CTID: NCT06109428
Phase: Phase 4 Status: Completed
Date: 2023-10-31
PAIR (Paracetamol and Ibuprofen Research) Pilot Trial
CTID: NCT04986839
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2023-10-30
A Multi-surgery Assessment of ZYNRELEF (HTX-011), AMAZE. Master Protocol HTX-011-401.
CTID: NCT05109312
Phase: Phase 4 Status: Active, not recruiting
Date: 2023-10-26
'Management of Low Back Pain in the Emergency Department With Different Analgesic Dosages'
CTID: NCT06064175
Phase: Phase 4 Status: Completed
Date: 2023-10-03
Dexketoprofen and Ibuprofen in Long Bone Fractures
CTID: NCT06060236
Phase: Phase 4 Status: Recruiting
Date: 2023-10-02
Hydrocodone Compared to Acetaminophen and Ibuprofen for Post-nail Procedure Analgesia
CTID: NCT05544734
Phase: Phase 4 Status: Completed
Date: 2023-09-28
Low-Dose Ibuprofen in Improving Cognitive Impairment in Patients With Cancer
CTID: NCT03186638
Phase: Phase 2 Status: Completed
Date: 2023-09-25
Post-operative Pain Management in Children With Supracondylar Humerus Fractures
CTID: NCT05640674
Phase: Phase 4 Status: Enrolling by invitation
Date: 2023-09-13
Evaluation of Effect of Intravenous Morphine vs Intravenous Ibuprofen and Acetaminophen vs Intravenous Ibuprofen
CTID: NCT05630222
Phase: Phase 3 Status: Completed
Date: 2023-09-07
Pain Management Following Sinus Surgery
CTID: NCT03822962
PhaseEarly Phase 1 Status: Terminated
Date: 2023-09-06
Total Knee Arthroplasty (TKA) Study of HTX-011 in an Multimodal Analgesic Regimen (MMA) Regimen
CTID: NCT03974932
Phase: Phase 3 Status: Completed
Date: 2023-08-31
Efficacy of Intravenous Ibuprofen and Paracetamol on Postoperative Pain and Tramadol Consumption in Shoulder Surgery
CTID: NCT05401916
Phase: N/A Status: Completed
Date: 2023-08-18
Chiropractic Spinal Manipulative Therapy for Acute Neck Pain
CTID: NCT05374057
Phase: Phase 4 Status: Recruiting
Date: 2023-08-08
Post Operative Analgesia and Patient Satisfaction
CTID: NCT04976387
Phase: Phase 3 Status: Completed
Date: 2023-08-08
Delirium in Elderly Patients With Trauma of the Hip
CTID: NCT02689024
Phase: Phase 4 Status: Terminated
Date: 2023-08-04
Dark Chocolate, Coconut Water, and Ibuprofen in Managing Primary Dysmenorrhea
CTID: NCT05971186
Phase: Phase 2 Status: Completed
Date: 2023-08-02
Ibuprofen Versus Ketorolac for Perioperative Pain Control After Open Hysterectomy
CTID: NCT05610384
Phase: N/A Status: Completed
Date: 2023-07-27
Effect of Acetaminophen on Postpartum Blood Pressure Control in Preeclampsia With Severe Features
CTID: NCT02911701
Phase: Phase 4 Status: Completed
Date: 2023-07-27
LIBERATE Trial in COVID-19
CTID: NCT04334629
Phase: Phase 4 Status: Withdrawn
Date: 2023-07-25
Effect of Preoperative Oral Ibuprofen on Anesthetic Efficacy of Inferior Alveolar Nerve Block With Supplemental Buccal and Lingual Infiltrations Using Articaine in Mandibular Molar Teeth With Irreversible Pulpitis
CTID: NCT05927922
Phase: Phase 1 Status: Not yet recruiting
Date: 2023-07-03
Early Treatment Versus Expectative Management of PDA in Preterm Infants
CTID: NCT02884219
Phase: N/A Status: Completed
Date: 2023-07-03
Ibuprofen Liquid Capsules 2 x 200 mg Efficacy and PK/PD Study in Surgical Removal of Impacted Third Molars
CTID: NCT05484401
Phase: Phase 3 Status: Completed
Date: 2023-06-22
Supracondylar Post-Operative Pain Study
CTID: NCT03759028
Phase: Phase 4 Status: Recruiting
Date: 2023-06-13
Effect of Combinations of Paracetamol, Ibuprofen, and Dexamethasone on Patient-Controlled Morphine Consumption in the First 24 Hours After Total Hip Arthroplasty
CTID: NCT04123873
Phase: Phase 4 Status: Completed
Date: 2023-06-01
Safety of Ibuprofen After Major Orthopaedic Surgeries
CTID: NCT05575700
Phase: Phase 4 Status: Recruiting
Date: 2023-04-28
Bariatric Surgery and Pharmacokinetics of Ibuprofen
CTID: NCT03476577
Phase: Status: Recruiting
Date: 2023-04-13
Intravenous Ibuprofen Versus Ketorolac in Bariatric Surgery
CTID: NCT05801900
Phase: N/A Status: Not yet recruiting
Date: 2023-04-06
Effect of Post-operative Ibuprofen After Surgery for Chronic Rhinosinusitis
CTID: NCT03055507
Phase: Phase 2/Phase 3 Status: Completed
Date: 2023-03-22
Ibuprofen Compared to Morphine as a Pediatric Postoperative Pain Management Tool Following Inguinal Surgery
CTID: NCT02603848
Phase: N/A Status: Recruiting
Date: 2023-03-15
The Effect of Combined Oral and Topical Analgesics to Reduce Pain Perception During Electrodiagnostic Testing
CTID: NCT04337814
Phase: N/A Status: Completed
Date: 2023-02-28
Effectiveness of Pre-emptive Analgesics on Post-Operative Pain After Stainless Steel Crown Placement On Primary Molars
CTID: NCT05602064
Phase: Phase 4 Status: Completed
Date: 2023-02-23
Efficacy of the Combination of Ibuprofen and Paracetamol in Acute Non-specific Low Back Pain
CTID: NCT05222724
Phase: Phase 4 Status: Completed
Date: 2023-01-31
Postoperative Analgesic Effects of Ibuprofen Versus Ketorolac in Patients Undergoing in Orthopedic Surgery
CTID: NCT05695664
PhaseEarly Phase 1 Status: Completed
Date: 2023-01-25
Methadone and Medication Abortion
CTID: NCT04941443
Phase: Phase 4 Status: Terminated
Date: 2023-01-23
the Effect of Preoperative Sodium Ibuprofen on Postoperative Endodontic Pain
CTID: NCT04964622
Phase: Phase 1/Phase 2 Status: Completed
Date: 2022-12-07
Efficacy of Acetaminophen-ibuprofen Combination on the Postoperative Pain After Thyroidectomy
CTID: NCT05626010
Phase: N/A Status: Unknown status
Date: 2022-11-25
A RCT of a Combination of Analgesics for Pain Management in Children With a Suspected Fracture
CTID: NCT02985177
Phase: Phase 4 Status: Withdrawn
Date: 2022-11-23
Acute Low Back Pain in the Emergency Department Treated With Osteopathic Manipulative Treatment Versus NSAIDs
CTID: NCT04129437
Phase: Phase 4 Status: Unknown status
Date: 2022-11-07
Combination of Ibuprofen, G-CSF and Plerixafor as Stem Cells Mobilization Regimen in Patients Affected by X-CGD
CTID: NCT03055247
Phase: Phase 2 Status: Unknown status
Date: 2022-10-31
Efficacy of Acetaminophen-ibuprofen Combination on the Postoperative Pain After Laparoscopic Gynecology Surgery
CTID: NCT05509244
Phase: N/A Status: Unknown status
Date: 2022-09-26
Pain Control Without Opioids
CTID: NCT04813991
Phase: Phase 3 Status: Withdrawn
Date: 2022-09-10
Pilot Study to Collect and Evaluate Data on the Use of IV* Ibuprofen in the Treatment of an Acute Migraine Attack
CTID: NCT01230411
Phase: Phase 4 Status: Completed
Date: 2022-09-06
Evaluation of Methylprednisolone or Ibuprofen on Efficacy of Nerve Blocks With Symptomatic Irreversible Pulpitis
CTID: NCT04157036
Phase: Phase 3 Status: Terminated
Date: 2022-08-29
Evaluation the Effect of Using Preoperative Ibuprofen Versus Low Level Laser Therapy on Inferior Alveolar Nerve Block
CTID: NCT05479175
Phase: N/A Status: Unknown status
Date: 2022-07-29
Sanaria PfSPZ Challenge With Pyrimethamine or Chloroquine Chemoprophylaxis Vaccination (PfSPZ-CVac Approach): A Randomized Double Blind Placebo Controlled Phase I/II Trial to Determine Safety and Protective Efficacy Against Natural Plasmodium Falcipa...
CTID: NCT03952650
Phase: Phase 1/Phase 2 Status: Completed
Date: 2022-07-12
Effect of Ibuprofen on Postoperative Opiate Medication Use and Shoulder
CTID: NCT02588027
Phase: N/A Status: Unknown status
Date: 2022-07-07
A Comparison of NSAIDs for Acute, Non-radicular Low Back Pain.
CTID: NCT03861611
Phase: Phase 4 Status: Completed
Date: 2022-05-31
ICE T for Post GYN Surgery Pain
CTID: NCT03987022
Phase: Phase 4 Status: Completed
Date: 2022-05-02
Effect of Cryotherapy Versus Post-operative Ibuprofen Medication on Post-operative Pain in Mandibular Molar Teeth With Symptomatic Irreversible Pulpitis
CTID: NCT05341999
Phase: Phase 1 Status: Unknown status
Date: 2022-04-22
Towards Predicting the Analgesic Response to Ibuprofen Following Third-molar Extraction
CTID: NCT03893175
Phase: Phase 1 Status: Completed
Date: 2022-04-19
To Evaluate the Food Effect and the Absorption Profile of Ibuprofen Modified-Release Tablets 800 mg
CTID: NCT05329454
Phase: Phase 1 Status: Completed
Date: 2022-04-15
Ibuprofen Plus Acetaminophen for Enhanced Pain Reduction
CTID: NCT04630834
Phase: Phase 4 Status: Unknown status
Date: 2022-04-14
Early Treatment Versus Expectant Management of PDA in Preterm Infants
CTID: NCT03860428
Phase: N/A Status: Completed
Date: 2022-04-13
Efficacy and Safety Study of Ibuprofen Gel Compared to Placebo in the Treatment of Acute Musculoskeletal Pain
CTID: NCT05013567
Phase: Phase 3 Status: Unknown status
Date: 2022-03-21
Utility of Pharmacogenomic Testing and Postoperative Dental Pain Outcomes
CTID: NCT02932579
Phase: Phase 4 Status: Terminated
Date: 2022-03-16
Patient Narcotic Requirements After Outpatient Otolaryngology Procedures
CTID: NCT03404518
Randomized clinical trial to evaluate the efficacy of hydroxychloroquine associated or not with azithromycin as a treatment for COVID-19 infection.
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2020-04-07
“Pre-emptive analgesia with Ibuprofen in outpatient laparoscopic cholecystectomy. Recovery Quality ”
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-12-26
Evaluation of efficacy and bioavailability of a new pediatric formulation based on ibuprofen lysinate vs ibuprofen in the management of pain in children
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2019-12-18
Effect of Combinations of Paracetamol, Ibuprofen, and Dexamethasone on Patient-Controlled Morphine Consumption in the First 24 Hours After Total Hip Arthroplasty. The RECIPE Randomized Clinical Trial
CTID: null
Phase: Phase 4 Status: Completed
Date: 2019-11-26
nd
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-10-22
Oral Ibuprofene versus oral kethorolac for children with musculoskeletal injury: a double blind randomized controlled study.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2019-10-02
Efficacy of Non-Steroidal Anti-Inflammatory (Ibuprofen) Chronotherapy in Healing After Mandibular Third Molar Surgical Extraction – A Randomized Clinical Trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-09-05
Efficacy of a new ibuprofen formulation for vaginal application
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2018-07-27
PHASE III CLINICAL TRIAL FOR THE EVALUATION OF THE EFFICACY AND SAFETY OF A PARACETAMOL + IBUPROFEN COMBINATION IN PATIENTS WITH PRIMARY DYSMENORRHOEA
CTID: null
Phase: Phase 3 Status: Completed
Date: 2018-04-25
'The effect of Betamethasone gel and NSAID gel lubricated on the laryngeal mask on pain in the throat, hoarseness and cough after anesthesia. '
CTID: null
Phase: Phase 2 Status: Completed
Date: 2017-07-14
Double-blind, randomized, placebo-controlled, Single-center, Exploratory Clinical Trial to Investigate Safety and Efficacy of COMBOPROFEN for treatment of muscular pain associated with DOMS
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2017-05-16
Phase III clinical trial , multicenter , randomized , double-blind , crossover, active-controlled and placebo to evaluate the analgesic efficacy and safety of paracetamol / ibuprofen 500/200 mg compared with 500 mg paracetamol alone, ibuprofen 200 mg alone and placebo patients with primary dysmenorrhea
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2017-03-08
Prospective study of anti-inflammatory therapy post interventional radiological treatment of HCC in the cirrhotic patient.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2017-03-02
A multicenter randomized trial to evaluate the efficacy of fentanyl pectin nasal spray (FPNS) versus Physician Choice (PC) - Usual Care (UC), in reducing incidental predictable breakthrough pain (IP-BTP) at swallowing in patients with head and neck cancer undergoing radiotherapy
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2017-02-23
Phase III, randomized, multicenter, double-blind clinical trial to evaluate two echo-guided administration regimens of ibuprofen in the treatment of patent ductus arteriosus: impact on intestinal prognosis
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2016-11-10
Multicentre controlled, randomized clinical trial to compare the efficacy and safety of ambulatory treatment of mild acute diverticulitis without antibiotics with the standard treatment with antibiotics
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-10-27
SINCERE: A single-centre, assessor blind, randomised pilot study to evaluate the safety, tolerability and acceptability of RB Lotion compared to standard-of-care for Radiation Induced Skin Reactions (RISR), in subjects undergoing palliative external beam Radiotherapy (RT).
CTID: null
Phase: Phase 2 Status: Completed
Date: 2016-10-19
A randomised, open-label trial of a Multimodal Intervention (Exercise, Nutrition and Anti-inflammatory Medication) plus standard care versus standard care alone to prevent / attenuate cachexia in advanced cancer patients undergoing chemotherapy.
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA, Completed
Date: 2016-05-17
Randomized clinical trial to compare the safety and effectiveness of metamizol, ibuprofen and tramadol added to a fixed dose of paracetamol in the treatment of post- surgical pain in patients ? 80 year after an intervention to repare a fracture of the proximal third of the femur, admitted to a convalescence unit
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-04-25
A Phase 3 Randomized, Double-Blind, Placebo-Controlled Trial to Study the Efficacy and Safety of MK-8342B (ENG-E2 Vaginal Ring) in Women with Moderate to Severe Primary Dysmenorrhea (with Optional Extension).
CTID: null
Phase: Phase 3 Status: Completed
Date: 2016-04-15
Paracetamol versus ibuprofen in preterm infants with a hemodynamically significant patent ductus arteriosus: a randomized clinical trial.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2016-04-06
A single centre, randomised, single-blind, parallel group single dose study to compare the speed of onset of ibuprofen gel,
e.querySelector("font strong").innerText = 'View More'
} else if(up_display === 'none' || up_display === '') {
icon_angle_down.style.display = 'none';
icon_angle_up.style.display = '