| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 100mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Camphor is rapidly absorbed through the mucous membranes and gastrointestinal tract, reaching peak plasma concentrations within 5 to 90 minutes after oral administration. Camphor is excreted via the kidneys. The volume of distribution of camphor is 2 to 4 L/kg. Camphor and its metabolites are relatively lipid-soluble and may accumulate in adipose tissue and other tissues. Camphor ingested by the mother is found in amniotic fluid, umbilical cord blood, fetal blood, and in the fetal brain, liver, and kidneys. Pharmacokinetic data are currently unavailable. Metabolism/Metabolites (S)-camphor is rapidly oxidized to 5-exo-hydroxyfenone, a process primarily mediated by human liver microsomal cytochrome P450. CYP2A6 is the main enzyme involved in the hydroxylation of (-)-camphor by human liver microsomes. Biological Half-Life After oral administration of 200 mg camphor, the half-life is 167 minutes. |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
Protein Binding
No pharmacokinetic data available. |
| 参考文献 | |
| 其他信息 |
L-camphor is a colorless or white crystal. It has a strong aromatic odor, a slightly bitter taste, and a cooling sensation. Odor index at 68°F (20°C): 40. Flash point: 149°F (65°C). It burns with a bright flame and produces smoke. It sublimates significantly at room temperature and pressure; at 176°F (80°C) and 12 mmHg, 14% sublimates within 60 minutes. (NTP, 1992)
(S)-camphor is the S-enantiomer of camphor. It is the enantiomer of (R)-camphor. (S)-camphor, or L(-)-camphor, is a stereoisomer of [DB01744], a bicyclic monoterpene known to enhance the sensation of heat and cold. (S)-Camphor is not a naturally occurring stereoisomer, but it has similar affinity for TRPV channels and current-inhibiting effects. [DB01744] is isolated from the wood of the camphor tree (Cinnamomum camphora) and has a long history of medicinal use. It has been used as a nasal decongestant and cough suppressant, and is also used topically due to its antipruritic, analgesic, and anti-irritant properties. Camphor is the main active ingredient in many over-the-counter ointments and liniments, and as a topical analgesic, it relieves mild muscle and joint pain by inducing sensitivity to hot and cold. (-)-Camphor has been reported to be found in sage, camphor thyme, and several other organisms with relevant data. Pharmacological Indications: Suitable for the temporary relief of mild muscle and joint pain as a topical analgesic. Mechanism of Action: TRPV3 cation channels are molecular sensors that function in nociception and temperature sensation by inducing thermal sensation and thermo-induced hyperalgesia. Camphor interacts with TRPV3 channels via cysteine residues in the pore region, leading to channel activation and increased intracellular calcium levels. Camphor also activates TRPV1 and TRPV1-like currents in dorsal root ganglion (DRG) neurons, but inhibits the ankyrin repeat TRP1 (TRPA1) channel, which is responsible for temperature sensing, expressed in most nociceptive DRG neurons. The exact mechanism by which TRPA1 current inhibition contributes to the analgesic effect of camphor is unclear. Repetitive stimulation of camphor leads to sensitization of TRPV1 and TRPV3 channels, resulting in channel desensitization or reduced response, which may explain the analgesic effect of camphor. Camphor also activates the cold-sensitive transient receptor potential melanin-inhibin 8 (TRPM8) and enhances cold-induced calcium transients, explaining the cooling effect after transdermal administration of camphor. Furthermore, studies have shown that camphor can inhibit the response of menthol to TRPM8 receptors. Pharmacodynamics Topical application of camphor produces a warming sensation, thereby exerting an analgesic effect. It excites and desensitizes sensory nerves by activating thermosensitive TRP vanillic acid receptor type 1 (TRPV1) and TRPV3 receptors. (S)-camphor has been reported to have a weaker effect on TRPV1 channels, which is thought to be due to rapid tolerance, i.e., a diminished response to repeated stimulation. |
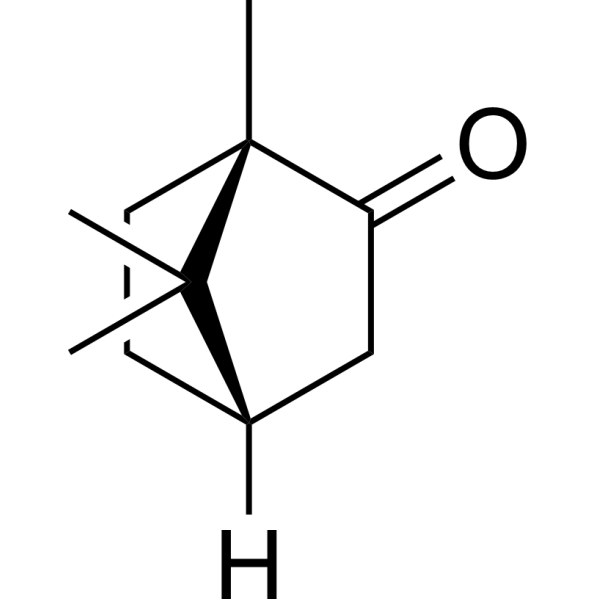
| 分子式 |
C10H16O
|
|---|---|
| 分子量 |
152.24
|
| 精确质量 |
152.12
|
| CAS号 |
464-48-2
|
| PubChem CID |
444294
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 密度 |
1.0±0.1 g/cm3
|
| 沸点 |
207.4±0.0 °C at 760 mmHg
|
| 熔点 |
178-180ºC
|
| 闪点 |
64.4±0.0 °C
|
| 蒸汽压 |
0.2±0.4 mmHg at 25°C
|
| 折射率 |
1.485
|
| LogP |
2.13
|
| tPSA |
17.07
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
1
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
11
|
| 分子复杂度/Complexity |
217
|
| 定义原子立体中心数目 |
2
|
| SMILES |
O=C1C([H])([H])C2([H])C([H])([H])C([H])([H])C1(C([H])([H])[H])C2(C([H])([H])[H])C([H])([H])[H]
|
| InChi Key |
DSSYKIVIOFKYAU-OIBJUYFYSA-N
|
| InChi Code |
InChI=1S/C10H16O/c1-9(2)7-4-5-10(9,3)8(11)6-7/h7H,4-6H2,1-3H3/t7-,10+/m0/s1
|
| 化学名 |
(1S,4S)-1,7,7-trimethylbicyclo[2.2.1]heptan-2-one
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.5686 mL | 32.8429 mL | 65.6858 mL | |
| 5 mM | 1.3137 mL | 6.5686 mL | 13.1372 mL | |
| 10 mM | 0.6569 mL | 3.2843 mL | 6.5686 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。