| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
GABAB receptor
|
|---|---|
| 体外研究 (In Vitro) |
3-氨基丙基次膦酸(10 μM)可作为皮肤抗衰老药物[4]。它还会诱导电刺激回肠中胆碱能抽搐收缩的浓度依赖性抑制(IC50=1.84-0.23 μM)[2]。
测量了GABA、β-丙氨酸和甘氨酸的膦类似物对豚鼠回肠纵肌的影响。3-氨基丙基次膦酸(AMPh)和2-氨基乙基膦酸(2-AEPh)在非刺激制剂和电刺激制剂中均无任何作用。GABA的膦类似物3-Aminopropylphosphinic acid/3-氨基丙基膦酸(3-APPh)在10(-3)M的剂量下具有GABAB激动作用(放松和抑制抽搐反应)。没有观察到对GABAA受体的激动作用。3-APPh在测试剂量(2 X 10(-4)M和10(-3)M)下也对GABAB激动剂的作用显示出拮抗作用,导致GABA和(-)-巴氯芬抑制抽搐反应的对数剂量-效应曲线平行偏移。相比之下,3-APPh没有拮抗吗啡和去甲肾上腺素的抑制作用。通过GABAA受体介导的GABA的收缩作用不受3-APPh(10(-3)M)的影响。结论3-APPh是豚鼠回肠GABAB位点的部分激动剂。[1] 1.测试了γ-氨基丁酸(GABA)类似物3-氨基丙基次膦酸对豚鼠离体回肠和大鼠离体肛尾肌制剂的活性。比较了3-Aminopropylphosphinic acid/3-氨基丙基次膦酸与GABA和巴氯芬的作用。2.在电刺激的回肠中,3-氨基丙基次膦酸,如GABA和巴氯芬,对胆碱能抽搐收缩产生浓度依赖性抑制,IC50值为1.84+/-0.23 microM(n=12)。与GABA不同,但与巴氯芬一样,3-氨基丙基次膦酸不会产生初始收缩。3.3-氨基丙基次膦酸和巴氯芬在豚鼠回肠中的抑制作用未被荷包牡丹碱(10微M)、酚妥拉明加普萘洛尔(均为1微M),育亨宾(1微M。然而,3-氨基丙基次膦酸的抑制作用,而巴氯芬的抑制作用则没有,被phaclofen(500微M)拮抗。此外,巴氯芬脱敏可消除豚鼠回肠中3-氨基丙基次膦酸的影响。4.3-氨基丙基次膦酸、GABA和巴氯芬可减少电场刺激引起的大鼠尾骨肌抽搐收缩。3-氨基丙基次膦酸抑制尾骨收缩的IC50为0.89+/-0.15微M(n=8)。5.结论是,3-氨基丙基次膦酸是一种强效、选择性的GABAB激动剂,在豚鼠回肠中的效力是巴氯芬的七倍,在大鼠肛门括约肌制剂中的效力比巴氯芬强五倍。[2] |
| 体内研究 (In Vivo) |
3-氨基丙基次膦酸(5 mg/kg;静脉注射)通过阻断 GABA 的作用来抑制豚鼠的迷走神经支气管痉挛[3]。
GABA是中枢神经系统中一种已知的抑制性神经递质。最近的研究还表明,GABA存在于包括肺在内的外周组织中。为了阐明GABA在肺中的作用,研究了GABA和选择性GABA激动剂和拮抗剂对豚鼠神经元诱导的气道收缩的影响。在体外,河豚毒素和阿托品抑制了电场刺激(EFS)诱导的气管收缩,表明收缩是由乙酰胆碱的神经元释放介导的。由EFS引起的收缩,而不是由外源性乙酰胆碱引起的收缩被GABA(EC50=4.5微M)和选择性GABA-B激动剂巴氯芬(EC50=9微摩)抑制,但不被GABA-A激动剂麝香醇抑制。巴氯芬的抑制作用不受GABA-A拮抗剂荷包牡丹碱的影响,但被GABA-B拮抗剂3-Aminopropylphosphinic acid/3-氨基丙基次膦酸(3-APPA)(pA2=4.5)和2-羟基乙酰氯芬(pA2=4.1)显著逆转。在体内,麻醉、机械通气豚鼠的迷走神经刺激(5 V,20 Hz,0.5 ms,5 s)引起胆碱能依赖性支气管痉挛,静脉注射GABA(3和10 mg/kg)和巴氯芬(1-10mg/kg)可抑制该痉挛,但麝香醇不能抑制。GABA和巴氯芬对迷走神经支气管痉挛的抑制作用被3-APPA(5mg/kg,静脉注射)阻断,但未被荷包牡丹碱阻断。分别用酚妥拉明或普萘洛尔阻断α肾上腺素能受体和β肾上腺素能受体治疗动物后,对GABA-B激动剂的反应没有改变。GABA和巴氯芬也不会改变静脉注射乙酰甲胆碱引起的支气管痉挛[3]。 |
| 细胞实验 |
“体外”制剂[1]
实验在雄性豚鼠(体重范围300-500g)中进行;动物们被头部的一击所伤;快速去除末端回肠的片段,并将其放置在以下成分(mM)的改良克雷布斯溶液中:KHzP041.3,KCI 3.4,NaCl134.7,CaC12 2.8,MgSO4 0.6,NaHC03 16.3,葡萄糖7.7。通过Paton和Zar(1968)的方法获得了附着有肌间神经丛的回肠纵向肌条。将管段安装在器官浴中,用5%COZ和95%02的混合物起泡,并保持在37°C。必要时,按照Paton(1963)和Paton&VLzi(1969)描述的方法进行电刺激。使用MARB刺激器的两个同轴铂电极施加刺激(持续时间为1msec的最大矩形脉冲的1.5倍,频率为每分钟6次)。在0.5g的静张力下将回肠连接到等距换能器,并在MARB多导生理记录仪上记录反应。给药前,让制剂平衡60分钟。给药量不得超过总浴量的1%(4毫升)。通过在两次给药之间间隔20分钟,可以防止GABA脱敏的发生。事实上,正如我们之前观察到的(Giotti等人,1983a;b),在非刺激和电刺激制剂中,以15-30min的间隔重复亚最大剂量的GABA都会引起相同的效果。 |
| 动物实验 |
动物/疾病模型:豚鼠[3]
剂量:5 mg/kg 给药途径:静脉注射 实验结果:阻断了GABA对迷走神经支气管痉挛的抑制作用。 豚鼠回肠[2] 雄性豚鼠(300-450克)头部被击打致死并放血。从回盲瓣近端10-15厘米处切取约3厘米长的回肠段。将标本立即置于改良的克氏液(BUlbring,1953)中,并持续通入95% O₂和5% CO₂的混合气体。将肠段从肠系膜上剥离,悬挂于盛有10 ml Krebs溶液的器官浴槽中,并施加1 g的等长张力。采用应变计传感器记录经壁刺激引起的等长收缩(Paton,1954),并在Ormed Multitrace笔式记录仪上显示。通过铂电极,利用Grass SD11刺激器向标本施加矩形脉冲(脉冲宽度0.5 ms;频率0.1 Hz;超最大电压25-35 V),从而实现电刺激。在向器官浴槽中加入化合物之前,标本平衡1小时。 大鼠肛尾肌[2] 雄性Wistar大鼠(200-300 g)被击打头部处死,放血后取出肛尾肌,方法如前所述(Gillespie,1972)。将肌肉组织置于盛有改良克氏液(10 ml)的器官浴槽中,并持续通入95%氧气和5%二氧化碳的混合气体。施加0.5 g的静息张力,并使用Grass SD11电刺激器通过铂环电极对标本进行电场刺激,刺激参数如下:脉冲持续时间1 ms;频率10 Hz;持续1 s。使用应变式传感器测量等长肌肉反应,并在Ormed Multitrace笔式记录仪上显示。两种标本经电刺激产生的典型张力反应在2至4 g之间。张力反应低于此值的标本被剔除。在两种标本中,均构建了激动剂浓度-反应曲线,每次添加激动剂之间间隔30分钟,以最大程度地减少快速耐受性。当使用拮抗剂时,在拮抗剂存在下,先建立初始平衡,然后绘制3-氨丙基膦酸的浓度-反应曲线。使用的药物包括:γ-氨基丁酸、(±)-巴氯芬、(±)-普萘洛尔(ICI)、甲磺酸酚妥拉明、盐酸育亨宾、盐酸纳洛酮、8-苯基茶碱、草酸异丙咪嗪、碘化比库啉、法氯芬和3-氨丙基膦酸(采用Dingwall等人1987a, b的方法制备)。除8-苯基茶碱外,所有化合物均溶于蒸馏水,后续稀释也用蒸馏水进行,并将化合物加入器官浴槽中,加入体积不超过总体积的1%。8-苯基茶碱用 80% 甲醇/2 M NaOH 配制溶液,后续稀释均用蒸馏水进行,所有溶剂对照均为阴性。将普萘洛尔和酚妥拉明直接加入克氏液中。 |
| 参考文献 |
[1]. GABA-related activities of amino phosphonic acids on guinea-pig ileum longitudinal muscle. J Auton Pharmacol. 1986 Sep;6(3):163-9.
[2]. 3-Aminopropylphosphinic acid--a potent, selective GABAB receptor agonist in the guinea-pig ileum and rat anococcygeus muscle. Br J Pharmacol. 1989 Aug;97(4):1292-6. [3]. Prejunctional GABA-B inhibition of cholinergic, neurally-mediated airway contractions in guinea-pigs. Pulm Pharmacol. 1991;4(4):218-24. [4]. 3-Aminopropyl dihydrogen phosphate (3-APPA; 3-aminopropane phosphoric acid); a novel anti-aging substance. Journal of Investigative Dermatology, vol. 4, no. 106, 2015, p. 895. |
| 其他信息 |
Bioulac、De Tinguy-Moreaud、Vincent 和 Neuzil (1979) 曾描述过 3-APPh 对中枢神经元放电的抑制作用,该作用对双环戊烯不敏感(可能是 GABAB 受体);此外,Cates、Li、Yakashe 等人 (1984) 近期也证实该化合物对 GABA 结合位点具有一定的亲和力。关于 GABAA 受体,所测试的膦酸类药物均未显示对该亚型受体的激动活性:只有 3-APPh 能使回肠收缩,但这种收缩作用对双环戊烯和苦味素不敏感,且 GABA 不能使这种收缩脱敏。 3-APPh 对 GABAE 受体的无效性也与先前在中枢水平进行的结合研究结果一致(Galli、Zilletti、Scotton、Adembri 和 Giotti,1980;Cates 等,1984)。然而,我们主要关注的是研究这些药物对 GABA 受体的拮抗作用:它们均未显示对 GABAA 受体介导的收缩有干扰作用;相反,高剂量的 3-APPh 对 GABAB 介导的抑制作用表现出显著的拮抗作用;3-APPh 的拮抗作用是可逆的,并且对 GABA 能药物具有特异性。 2-AEPh 和 3-APPh 均无效。由此产生的一个问题是,3-APPh 对 GABAB 受体的激动作用是否会通过脱敏现象干扰其对同一受体的拮抗作用。3-APPh 能够迅速逆转 GABAA 对肌肉抽搐反应的影响(图 6),这一事实支持其具有直接拮抗作用的观点。此外,在较低测试剂量(2 × 10⁻⁵ M)下,3-APPh 对 GABAB 效应的拮抗作用似乎呈竞争性:剂量-效应曲线平行移动,最大效应几乎没有变化。另一方面,在最高剂量(10⁻³ M)下,脱敏现象可能发挥作用;事实上,在此浓度下,剂量-效应曲线趋于平缓,这可能是由于脱敏作用所致。总之,GABA的膦酸类似物3-氨丙基膦酸(3-APPh)表现出弱GABAB受体拮抗剂和弱GABAB受体激动剂的特性,而对GABAA受体没有任何作用(激动剂或拮抗剂)。这种特性可能被解释为部分激动剂,并且与其他被认为是GABAA受体拮抗剂的药物(如3-APS,其具有强GABAA受体激动剂和弱GABAB受体拮抗剂的特性不同)(表2)。因此,目前似乎尚未发现有效的GABAB受体拮抗剂。然而,准确了解作用于GABAB受体的药物之间的差异,有助于将其用作实验工具,并开发更具选择性的GABAB拮抗剂。[1]
已有若干研究探讨了GABAB受体活性的要求,结果表明,即使是巴氯芬分子的少量改变也会导致其活性完全丧失(Olpe等人,1980;Krogsgard-Larsen,1988)。迄今为止,尚未发现任何GABAB激动剂在本文所述的体外系统中比巴氯芬更有效。3-氨丙基膦酸在豚鼠回肠中的效力是消旋巴氯芬的7倍,在大鼠肛尾肌中的效力是消旋巴氯芬的5倍。Dingwall等人报道的研究表明, (1987a,b)据报道,3-氨基丙基膦酸对GABAB受体的亲和力是巴氯芬的20倍。有趣的是,将本文中描述为强效激动剂的GABA膦酸类似物与被描述为弱效部分激动剂/拮抗剂的膦酸类似物(Luzzi等人,1986)进行比较,尽管两者结构上的唯一区别在于酸性部分。3-氨基丙基膦酸的磷原子周围呈扭曲的四面体排列,只有一个酸性质子,负电荷分布在两个氧原子上,因此它在许多方面与GABA相似。 3-氨丙基膦酸的磷原子周围呈近四面体排列,含有两个酸性质子(取决于pH值),负电荷分布在三个氧原子上。在豚鼠回肠中,3-氨丙基膦酸似乎与位于胆碱能神经末梢突触前GABA受体相互作用。由于3-氨丙基膦酸的Sid值不显著,且未引起初始收缩,因此它可能不具有GABAA受体激动剂活性。尽管对这一假设的最终统计学验证尚需更具体的O值,但使用已知特异性受体拮抗剂Iic acidm(l)的曲线结果表明,3-氨基丙基膦酸不与其他任何类型的受体相互作用。此外,对巴氯芬脱敏的组织不再对3-氨基丙基膦酸产生反应。可乐定(其平均值高于平均值,p<0.05)被用于测试GABAB受体的特异性。在GABAB受体脱敏过程中,可乐定对巴氯芬的效力相当,且在GABAB受体脱敏前后均具有显著性差异。法氯芬被认为是一种弱但选择性的GABAB拮抗剂(Kerr等人,1987;Dutar和Nicholl,1988)。我们能够证明,在豚鼠回肠中,3-氨基丙基膦酸的反应受到抑制,但巴氯芬的反应则没有受到抑制。目前尚不清楚原因,尽管最近有研究声称,法氯芬在多种测试系统中表现出GABA拮抗活性(Karlsson等人,1988;Soltesz等人,1988),但其结构确实支持我们的假设,即丙基膦酸的3-氨基化与GABA受体相互作用。[2] 在豚鼠回肠和大鼠肛尾肌中,3-氨基丙基膦酸的反应时间进程与GABA和巴氯芬的反应时间进程相似。在豚鼠回肠中,GABAB受体激动剂的反应通常表现为短暂性,而在大鼠肛尾肌中,这种反应则更为持久(Bowery等,1981;Muhyaddin等,1982)。我们的药理学研究支持Dingwall等(1987a,b)的观点,即3-氨基丙基膦酸是一种高效、选择性的GABAB受体激动剂。对该化合物的进一步研究可能有助于阐明GABAB受体在哺乳动物肠道中的生理作用。[2] |
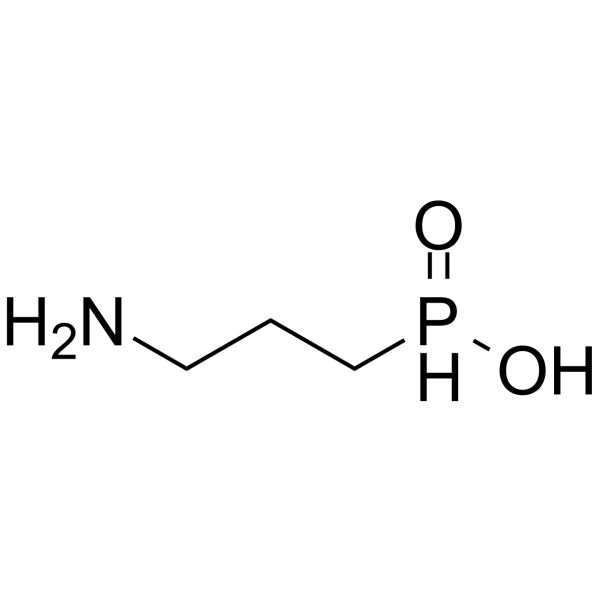
| 分子式 |
C3H10NO2P
|
|---|---|
| 分子量 |
122.08286
|
| 精确质量 |
122.037
|
| CAS号 |
103680-47-3
|
| PubChem CID |
6335948
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 蒸汽压 |
0.001mmHg at 25°C
|
| LogP |
-1.4
|
| tPSA |
95.92
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
7
|
| 分子复杂度/Complexity |
66
|
| 定义原子立体中心数目 |
0
|
| SMILES |
NCCCP(=O)O
|
| InChi Key |
MQIWYGZSHIXQIU-UHFFFAOYSA-O
|
| InChi Code |
InChI=1S/C3H8NO2P/c4-2-1-3-7(5)6/h1-4H2/p+1
|
| 化学名 |
3-aminopropyl-hydroxy-oxophosphanium
|
| 别名 |
3-aminopropylphosphinic acid; 103680-47-3; 3-aminopropyl-hydroxy-oxophosphanium; 3-Aminopropanephosphinic acid; (3-aminopropyl)phosphinic acid; CGP-27492; 3-APPA; Cgp 27492;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 8.1913 mL | 40.9567 mL | 81.9135 mL | |
| 5 mM | 1.6383 mL | 8.1913 mL | 16.3827 mL | |
| 10 mM | 0.8191 mL | 4.0957 mL | 8.1913 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。