| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Atinimole has been reported to have an oral bioavailability of 45% in healthy adults. The observed time to peak concentration (Tmax) is 1–2 hours. Prolonged Tmax is known in patients with malaria infection, possibly due to reduced hepatic metabolism or drug accumulation in infected erythrocytes. Atinimole exhibits flipped absorption kinetics, with a total absorption half-life of 1.04 hours. Co-administration with food increases the AUC of atinimole by 144%. An increase in Cmax of 129% was observed, but this was not statistically significant. Food can delay the time to peak concentration by 1 hour. Atinimole is eliminated by metabolism to glucuronide conjugates. Data on artemisinin elimination are scarce, but the elimination of unmetabolized artemisinin compounds in feces and urine has been reported to be negligible. In adult patients infected with P. falciparum, the mean apparent volume of distribution of artemisinin was 0.801 L/kg, while in pediatric patients infected with P. falciparum it was 0.705 L/kg. In adult patients infected with P. falciparum, the mean apparent clearance of artemisinin was 1.340 L/h/kg, while in pediatric patients infected with P. falciparum it was 1.450 L/h/kg. Metabolism/MetabolitesThe major metabolite of artemisinin is a glucuronide conjugate, α-artemisinin-β-glucuronide. It is primarily metabolized by UGT1A9, with UGT2B7 also involved in some metabolism. Biological Half-LifeThe elimination half-life of artemisinin has been reported to be approximately 1 hour. |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
Protein Binding
Artemisinin is reported to bind to plasma proteins at a rate of 44-93%. However, the identities of these proteins have not yet been disclosed. |
| 参考文献 | |
| 其他信息 |
Dihydroartemisinin (DHA) is a derivative of artemisinin. Atinibol is also a derivative of artemisinin and an antimalarial drug used to treat uncomplicated Plasmodium falciparum infection. It was first approved by the European Medicines Agency in October 2011 for use in combination with [DB13941], marketed as Eurotramesim. Artemisinin combination therapy is highly effective against malaria and is strongly recommended by the World Health Organization. Alpha-dihydroartemisinin is an antimalarial drug. Atinibol is the active metabolite of artemether, possessing antimalarial activity and potentially exhibiting insulin-modifying, anti-inflammatory, immunomodulatory, and antitumor activities. After administration of atinibol, heme released from parasite-infected erythrocytes hydrolyzes its active internal peroxide bridge, generating reactive oxygen species (ROS) and carbon-centered free radicals, thereby damaging and killing the parasite. Atinibol may also improve insulin sensitivity and alleviate insulin resistance. In addition, atinib induces 26S proteasome-mediated androgen receptor (AR) degradation, thereby reducing AR expression, which may inhibit the proliferation of androgen-responsive cells. It also reduces luteinizing hormone (LH) and testosterone levels and may improve polycystic ovary syndrome (PCOS). Furthermore, artemisinin may modulate the immune system and inhibit tumor cell proliferation through multiple apoptotic and non-apoptotic pathways.
See also: Artemisinin (note moved to). Drug Indications For the treatment of uncomplicated Plasmodium falciparum infection in adults, children, and infants aged 6 months and older weighing more than 5 kg. In combination with [DB13941]. FDA Label Mechanism of Action Artemisinin-class drugs, including artemisinin (the main active metabolite of many artemisinin-class drugs), are thought to act through a common mechanism of action. While the exact mechanism of action is not fully understood, there are many theories about how artemisinin produces its antimalarial effects. Artemisinin is thought to bind to heme within Plasmodium falciparum. The source of this heme varies depending on the life stage of the parasite. In the early cyclic stage, artemisinin is thought to bind to heme produced by the parasite's own heme biosynthesis pathway. In later stages, artemisinin may bind to heme released from hemoglobin digestion. Once bound to heme, artemisinin is thought to undergo an activation process involving the reduction cleavage of ferrous ions, thereby breaking internal peroxide bridges and generating reactive oxygen species (ROS). This ROS is thought to undergo subsequent intramolecular hydrogen extraction, generating reactive carbon radicals. These carbon radicals are considered the source of the drug's potent activity against Plasmodium falciparum, achieving this effect through alkylation of various protein targets. The nature and extent of the effect of this alkylation on the function of specific proteins are unclear. One key target studied is the sarcoplasmic reticulum/endoplasmic reticulum Ca2+ ATPase pump in Plasmodium falciparum. Artemisinin has been found to irreversibly bind to this protein and inhibit its activity, with a binding site similar to that of carotenoids. Its mechanism of action may be the same as other proteins, namely alkylation via a carbon radical intermediate. Artemisinin appears to preferentially accumulate in infected red blood cells, resulting in concentrations hundreds of times higher than in uninfected cells. This may explain why alkylation is barely observed in uninfected red blood cells. Pharmacodynamics Artemisinin is thought to form a reactive carbon radical intermediate that kills Plasmodium falciparum by alkylating various proteins. |
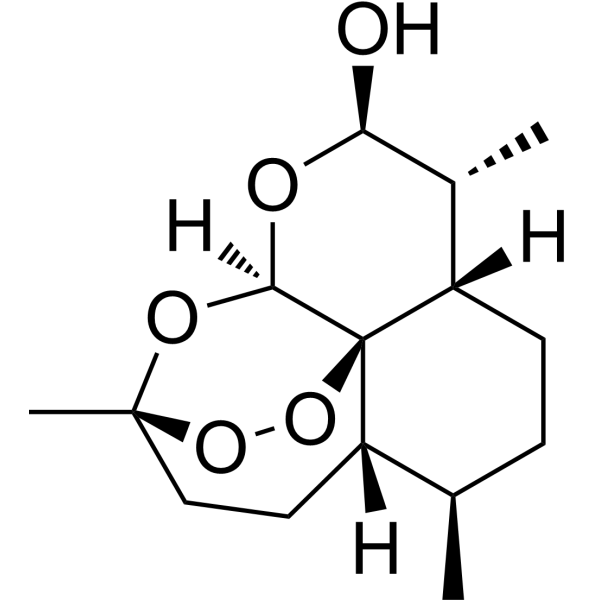
| 分子式 |
C15H24O5
|
|---|---|
| 分子量 |
284.35
|
| 精确质量 |
284.162
|
| CAS号 |
81496-81-3
|
| PubChem CID |
11358077
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 密度 |
1.3±0.1 g/cm3
|
| 沸点 |
375.6±42.0 °C at 760 mmHg
|
| 熔点 |
164-165
|
| 闪点 |
181.0±27.9 °C
|
| 蒸汽压 |
0.0±1.9 mmHg at 25°C
|
| 折射率 |
1.543
|
| LogP |
2.6
|
| tPSA |
57.15
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
20
|
| 分子复杂度/Complexity |
415
|
| 定义原子立体中心数目 |
8
|
| SMILES |
C[C@@H]1CC[C@H]2[C@H]([C@@H](O[C@H]3[C@@]24[C@H]1CC[C@](O3)(OO4)C)O)C
|
| InChi Key |
BJDCWCLMFKKGEE-KDTBHNEXSA-N
|
| InChi Code |
InChI=1S/C15H24O5/c1-8-4-5-11-9(2)12(16)17-13-15(11)10(8)6-7-14(3,18-13)19-20-15/h8-13,16H,4-7H2,1-3H3/t8-,9-,10+,11+,12-,13-,14-,15-/m1/s1
|
| 化学名 |
(1R,4S,5R,8S,9R,10R,12R,13R)-1,5,9-trimethyl-11,14,15,16-tetraoxatetracyclo[10.3.1.04,13.08,13]hexadecan-10-ol
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
Typically soluble in DMSO (e.g. 10 mM)
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5168 mL | 17.5840 mL | 35.1679 mL | |
| 5 mM | 0.7034 mL | 3.5168 mL | 7.0336 mL | |
| 10 mM | 0.3517 mL | 1.7584 mL | 3.5168 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。