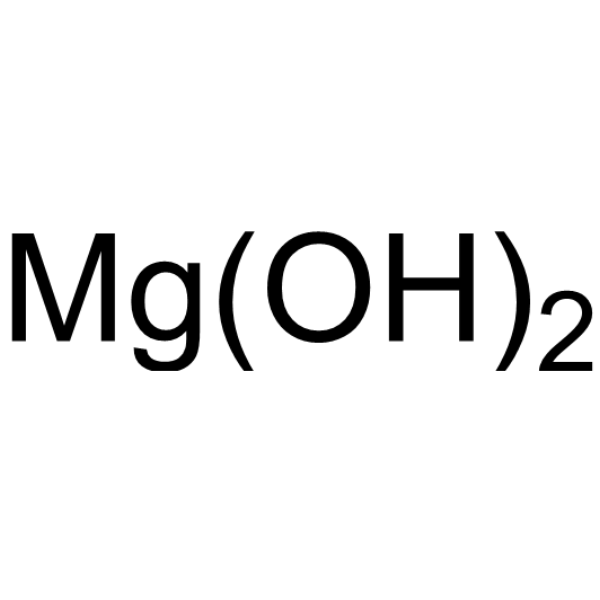
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25g |
|
||
| Other Sizes |
|
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Approximately 15%–50% of magnesium hydroxide is slowly absorbed through the small intestine. After oral administration, up to 50% of the magnesium hydroxide suspension can be absorbed in the small intestine as magnesium ions, and then rapidly excreted in the urine via the kidneys. Unabsorbed drug is primarily excreted in feces and saliva. Peak effect and distribution of magnesium hydroxide vary among individuals. Magnesium hydroxide is primarily excreted in the urine via the kidneys. Because the kidneys play a crucial role in magnesium clearance, patients with renal failure are at risk of developing hypermagnesemia with long-term magnesium supplementation, as adequate magnesium may not be excreted. Although magnesium hydroxide (Mg(OH)2) is classified as a non-systemic antacid, 5% to 10% of the magnesium can still be absorbed… Normally, absorbed magnesium ions are rapidly excreted through the kidneys. In normal individuals, magnesium absorption causes little or no systemic alkalosis, but urine may be alkaline. …Magnesium is slowly excreted from the stomach, thus prolonging its antacid effect. Approximately 5-15% of ingested magnesium is absorbed, and if kidney function is normal, this magnesium is rapidly excreted in the urine. Metabolism/Metabolites: Unless the patient is magnesium deficient, the intestines absorb very little magnesium. Overall, approximately 15%-50% of magnesium hydroxide suspension is absorbed systemically. However, because it is rapidly excreted in the urine, no metabolism occurs. Biological Half-Life: Not applicable. |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
Protein Binding
Magnesium hydroxide does not have any protein binding properties. Interactions …Magnesium hydroxide may interfere with warfarin absorption by alkalizing gastrointestinal contents. …Magnesium hydroxide suspension has been shown to actually enhance the absorption of dicumarol…higher peak serum concentrations… Urinary alkalization can reduce the excretion of many weakly basic drugs (e.g., quinidine). Mg(OH2) can increase the absorption of warfarin in humans and decrease the absorption of barbiturates in rats. Non-Human Toxicity Oral LD50 in rats: 8500 mg/kg Intraperitoneal LD50 in rats: 2780 mg/kg |
| 参考文献 | |
| 其他信息 |
Magnesium hydroxide is a magnesium hydroxide compound in which magnesium is combined with two hydroxide ions. It has anti-acid and flame-retardant properties. Magnesium hydroxide is an inorganic compound that naturally occurs in the mineral brucite. It can be used as an antacid or laxative, available in oral suspensions and chewable tablets. Furthermore, magnesium hydroxide has smoke-suppressing and flame-retardant properties, and is therefore commercially used as a flame retardant. It can also be used topically as a deodorant or to relieve oral ulcers (recurrent aphthous ulcers). Magnesium hydroxide solution is a magnesium hydroxide solution with both anti-acid and laxative effects. At low doses, the emulsion magnesium exerts an anti-acid effect, allowing all hydroxide ions entering the stomach to neutralize stomach acid. At high doses, it acts as a laxative, allowing hydroxide ions to move from the stomach into the intestines, where they absorb and retain water, thus promoting intestinal peristalsis and inducing defecation. Magnesium hydroxide is an inorganic compound that naturally occurs in the mineral brucite. It is an antacid with laxative properties. See also: Magnesium ions (with active portion); Calcium carbonate; Famotidine; Magnesium hydroxide (ingredient); Aluminum hydroxide; Magnesium hydroxide (ingredient)... See more...
Drug Indications Magnesium hydroxide can be used as an antacid or laxative depending on the dosage. As an antacid, it is used to temporarily relieve heartburn, stomach upset, excessive stomach acid, or acidic indigestion. As a laxative, it is used to relieve occasional constipation, promote defecation, and its effects can last from 30 minutes to 6 hours. Mechanism of Action Magnesium hydroxide suspension enters the stomach orally. Depending on the intake, magnesium hydroxide can act as an antacid or a laxative. When adults ingest 0.5-1.5 grams, magnesium hydroxide acts in the stomach through simple acid neutralization. The hydroxide ions in the magnesium hydroxide suspension combine with hydrogen ions in the hydrochloric acid produced by the parietal cells of the stomach. This neutralization reaction produces magnesium chloride and water. When adults ingest 2-5 grams, magnesium hydroxide acts as a laxative in the colon. Most suspensions are not absorbed by the intestines, but rather draw water from surrounding tissues into the intestines through osmosis. As the water content in the intestines increases, the stool softens, and the volume of stool in the intestinal lumen increases. These effects still stimulate intestinal peristalsis and induce the urge to defecate. Magnesium hydroxide also promotes the release of cholecystokinin (CKK) in the intestines, leading to the accumulation of water and electrolytes in the intestinal lumen, further enhancing intestinal peristalsis. COMPD is almost insoluble in water; the solution only dissolves when the hydroxide reacts with hydrochloric acid to form magnesium chloride. …Mg(OH)2 increases the pressure of the lower esophageal sphincter in Heidenham's pouch and gastric acid secretion. Laxatives containing magnesium cations or phosphate anions are generally called saline laxatives. Their laxative effect is thought to be through osmotic pressure-mediated water retention, which in turn stimulates intestinal peristalsis. However, other mechanisms may also contribute to their effect, including the production of inflammatory mediators. Studies have also shown that magnesium-containing laxatives stimulate the release of cholecystokinin, leading to the accumulation of fluid and electrolytes in the intestinal lumen and increasing intestinal peristalsis. /Saline Laxatives, including Magnesium Hydroxide/ Therapeutic Uses Antacids; Laxatives Magnesia Magma can be used as an antacid or a laxative depending on the dosage. 10 ml… can neutralize approximately 270 ml of 0.1N hydrochloric acid. …It is often used in combination with aluminum hydroxide gel because it relieves the latter's mild constipation effect and also has its own antacid effect. When the dosage exceeds the amount required to neutralize stomach acid, the stomach pH may reach 8 or 9. The acid rebound that occurs after taking magnesium hydroxide is not clinically significant. …It is mainly used for…laxative effects, and the dosage and frequency of administration depend on the amount of magnesium that replaces aluminum or calcium salts to achieve normal stool consistency. For more data on the therapeutic uses of magnesium (full version), please refer to the relevant materials. Hydroxides (10 cases in total), please visit the HSDB record page. Drug Warnings One disadvantage of using Mg(OH)2 as an antacid in some patients is its laxative effect. Therefore, magnesium hydroxide is often used in combination with or alternated with antacids that have a laxative effect, such as aluminum hydroxide and calcium carbonate, although the laxative effect of calcium carbonate is currently questionable. Veterinarian: ...renal dysfunction and repeated administration can lead to dangerous levels of magnesium retention, resulting in neurological, neuromuscular, and cardiovascular dysfunction. In patients with kidney disease, long-term use of magnesium salts may lead to magnesium retention and may cause neurological, neuromuscular, and cardiovascular dysfunction. Gastric acid secretion has been studied in ... in patients with duodenal ulcers before and after taking magnesium hydroxide and placebo. Gastric acid secretion increased significantly after taking magnesium hydroxide compared to baseline; no significant difference was found after taking placebo. For more complete data on drug warnings for magnesium hydroxide (7 of 7), please visit the HSDB record page. Pharmacodynamics As an antacid, magnesium hydroxide suspension neutralizes stomach acid by reacting with hydrochloric acid in the stomach to produce magnesium chloride and water. It is almost insoluble in water and has no effect until it reacts with hydrochloric acid in the stomach. After reacting with hydrochloric acid, it reduces the direct irritation of gastric acid and increases the pH of the stomach, thereby inactivating pepsin. Magnesium hydroxide enhances the integrity of the gastric mucosal barrier and improves the tone of the gastric and esophageal sphincters. As a laxative, magnesium hydroxide works by increasing the osmotic pressure of the intestines and absorbing water. This leads to colonic dilation, which in turn promotes intestinal peristalsis, ultimately achieving defecation. |
| 分子式 |
MG(OH)2
|
|---|---|
| 分子量 |
58.32
|
| 精确质量 |
57.99
|
| CAS号 |
1309-42-8
|
| PubChem CID |
73981
|
| 外观&性状 |
Amorphous powder
White, hexagonal crystals Granules |
| 密度 |
2.36g/cm3
|
| 沸点 |
100ºC at 760mmHg
|
| 熔点 |
350ºC ( Decomposes)
|
| 闪点 |
will not burn
|
| tPSA |
40.46
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
3
|
| 分子复杂度/Complexity |
0
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
VTHJTEIRLNZDEV-UHFFFAOYSA-L
|
| InChi Code |
InChI=1S/Mg.2H2O/h;2*1H2/q+2;;/p-2
|
| 化学名 |
magnesium;dihydroxide
|
| 别名 |
Magnesium hydroxide, ultrapure grade, 99%(KT); Magnesium dihydroxide, 99%(KT)
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 17.1468 mL | 85.7339 mL | 171.4678 mL | |
| 5 mM | 3.4294 mL | 17.1468 mL | 34.2936 mL | |
| 10 mM | 1.7147 mL | 8.5734 mL | 17.1468 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。