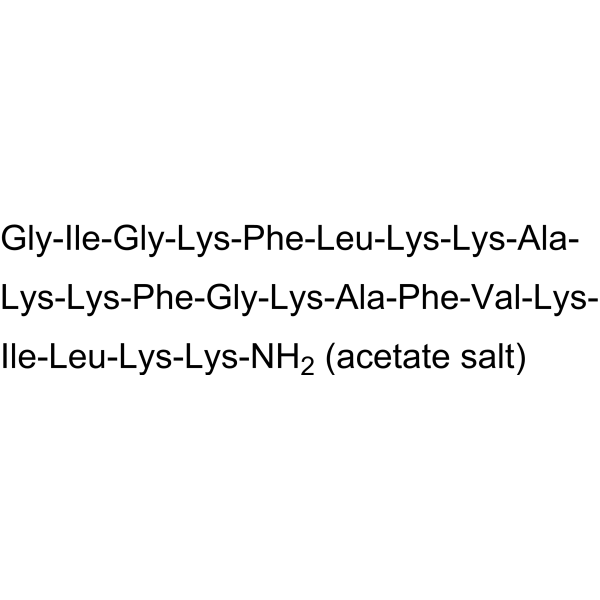
| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
The primary mechanism of action of Pexiganan is believed to be the disruption of microbial cytoplasmic membranes, leading to rapid cell death. It does not act on a specific molecular target like conventional antibiotics but rather interacts with the lipid components of the bacterial membrane. [1]
Pexiganan disrupts bacterial cytoplasmic membranes via electrostatic interactions with negatively charged phospholipids, leading to rapid cell lysis. It lacks specific molecular targets (e.g., enzymes/receptors), and no IC50/Ki/EC50 values for single targets are reported. [1][2][3][4] |
|---|---|
| 体外研究 (In Vitro) |
Pexiganan 对革兰氏阳性/阴性病原体具有广谱抗菌活性。临床分离株的MIC值:大肠杆菌(4–16 µg/mL)、铜绿假单胞菌(8–32 µg/mL)、金黄色葡萄球菌(4–16 µg/mL)、肠球菌属(8–32 µg/mL)、幽门螺杆菌(2–8 µg/mL;MIC₉₀ = 8 µg/mL)。对大肠杆菌、金黄色葡萄球菌和幽门螺杆菌具有杀菌效应(4×MIC下1–4小时内≥3 log₁₀ CFU减少)。酸性环境(pH 5.5)或高浓度二价阳离子(如10 mM Mg²⁺)会降低活性,但50%人血清影响轻微(MIC增加≤2倍)。 [2][3]
Pexiganan(128 µg/mL)处理人胃上皮细胞(GES-1)24小时后细胞毒性低(MTT法检测存活率>80%)。 [3] Pexiganan 在体外对多种与皮肤和软组织感染相关的革兰氏阳性菌和革兰氏阴性菌(包括从糖尿病足溃疡分离的菌株)表现出广谱抗菌活性。它对需氧菌和厌氧菌均有效。 [1] 实验证明其对诸如金黄色葡萄球菌(Staphylococcus aureus,包括耐甲氧西林菌株 - MRSA)、表皮葡萄球菌(Staphylococcus epidermidis)、化脓性链球菌(Streptococcus pyogenes)、粪肠球菌(Enterococcus faecalis,包括万古霉素耐药菌株 - VRE)、大肠杆菌(Escherichia coli)、铜绿假单胞菌(Pseudomonas aeruginosa)、奇异变形杆菌(Proteus mirabilis)、脆弱拟杆菌(Bacteroides fragilis)和消化链球菌(Peptostreptococcus)等微生物有效。 [1] 对大多数敏感微生物的最低抑菌浓度 (MIC) 通常在 16 至 32 µg/mL 范围内。 [1] 其杀菌活性迅速。 [1] |
| 体内研究 (In Vivo) |
小鼠幽门螺杆菌感染模型中,口服pexiganan(20 mg/kg,每日两次,持续7天)使胃部细菌载量降低2 log₁₀ CFU/g;纳米颗粒制剂(PNPs)效果更强(降低3.5 log₁₀)。 [3]
大鼠内毒素休克模型(LPS + D-半乳糖胺)中,静脉注射pexiganan(1 mg/kg)使生存率提高至67%(对照组0%),血浆TNF-α降低75%。与头孢曲松(10 mg/kg)联用生存率达100%。 [4] 局部外用 1% Pexiganan 乳膏在治疗轻度糖尿病足感染的 II 期临床试验中显示出疗效,其临床成功率 (85-90%) 与口服氧氟沙星 (85%) 相当。 [1] 对涉及革兰氏阳性菌和革兰氏阴性菌病原体(包括铜绿假单胞菌)的感染均观察到疗效。 [1] |
| 细胞实验 |
抗菌活性: 采用CLSI推荐的微量肉汤稀释法测定MIC。细菌在阳离子调节MH肉汤(革兰氏阳性/阴性菌)或Brucella肉汤+5% FBS(幽门螺杆菌)中与pexiganan系列稀释液(0.25–128 µg/mL)孵育,35°C培养16–24小时(幽门螺杆菌48小时)。MIC定义为完全抑制可见生长的最低浓度。
时间-杀灭动力学: 细菌(~10⁶ CFU/mL)暴露于pexiganan(1–4×MIC),定时取样(0–24小时),梯度稀释后平板计数。 细胞毒性(GES-1细胞): 细胞接种于96孔板,与pexiganan(4–128 µg/mL)孵育24小时,加入MTT试剂。溶解甲臜晶体后测定570 nm吸光度,以未处理组为基准计算存活率。 [2][3] |
| 动物实验 |
幽门螺杆菌感染模型:雌性C57BL/6小鼠(6-8周龄)经口接种幽门螺杆菌SS1株(5天内接种3剂)。4周后,感染小鼠每日两次口服pexiganan(溶于生理盐水),持续7天。对照组小鼠口服生理盐水。治疗3天后处死小鼠;取胃组织匀浆进行菌落形成单位(CFU)计数。
内毒素休克模型:雄性Sprague-Dawley大鼠(200-250 g)腹腔注射LPS(200 µg/kg)+ D-半乳糖胺(700 mg/kg)。1小时后,静脉注射pexiganan(溶于生理盐水,1 mg/kg),单独注射或与头孢曲松(10 mg/kg)联合注射。监测24小时的存活情况;于 1.5 小时后采集血液样本进行 TNF-α ELISA 检测。[3][4] |
| 药代性质 (ADME/PK) |
在II期临床试验中,局部应用1%的Pexiganan乳膏治疗轻度糖尿病足感染显示出疗效,其临床成功率(85-90%)与口服氧氟沙星(85%)相当。[1]
该乳膏对革兰氏阳性菌和革兰氏阴性菌感染均有效,包括铜绿假单胞菌。[1] 局部应用1%乳膏引起的轻微用药部位反应(灼烧感、红斑)发生率与赋形剂对照组相似。临床上未观察到全身毒性、肾毒性或耳毒性。体外实验表明,在抗菌浓度下,该乳膏对人成纤维细胞的细胞毒性极低。[1][3] Pexiganan(128 µg/mL)在1小时后未显示对人红细胞的溶血活性。[2] |
| 毒性/毒理 (Toxicokinetics/TK) |
在II期临床试验中,局部应用1%的Pexiganan乳膏耐受性良好。[1]
最常见的不良反应是轻微、短暂的用药部位反应(例如灼烧感、疼痛、红斑、皮疹),其发生频率与赋形剂乳膏对照组相似。[1] 未观察到与该药物相关的全身毒性。[1] 未发现耳毒性或肾毒性的证据。[1] 由于全身吸收可忽略不计,因此未报告血浆蛋白结合率和详细的毒代动力学研究。[1] 体外研究表明,在有效对抗细菌的浓度下,该药物对人成纤维细胞的细胞毒性极低。[1] 临床前研究表明该药物缺乏致突变性。[1] |
| 参考文献 | |
| 其他信息 |
Pexiganan是一种合成的22肽氨基酸残基的magainin类似物,专为局部治疗感染性糖尿病足溃疡而开发。其破坏细胞膜的机制可能有助于降低耐药性的产生。II期临床试验表明,该药对包括铜绿假单胞菌阳性伤口在内的轻度感染有效。[1][2]
纳米颗粒包封(PNPs)提高了Pexiganan的稳定性及其体内抗幽门螺杆菌的疗效。[3] 在内毒素休克模型中,Pexiganan抑制了TNF-α的表达,并与β-内酰胺类抗生素具有协同作用,提示其具有免疫调节作用。[4] 醋酸Pexiganan(MSI-78)是一种合成的阳离子抗菌肽类似物,其基于非洲爪蟾(Xenopus laevis)皮肤中发现的magainin肽。 [1] 它最初是作为一种局部抗菌剂开发的,专门用于治疗感染性糖尿病足溃疡。[1] 其作用机制被认为是物理破坏细菌细胞膜,与靶向特定生化途径的传统抗生素相比,这使其产生耐药性的可能性更低。[1] II期临床试验表明,其在治愈或改善轻度糖尿病足感染方面的疗效与口服氧氟沙星相当。[1] 其良好的安全性以及可忽略不计的全身吸收使其成为一种很有前景的局部治疗药物,有望避免全身使用抗生素及其相关的耐药性或副作用。[1] |
| 分子式 |
C122H210N32O22.XC2H4O2
|
|---|---|
| 分子量 |
2477.17 (free base)
|
| 精确质量 |
2535.65
|
| 元素分析 |
C, 58.70; H, 8.50; N, 17.67; O, 15.13
|
| CAS号 |
172820-23-4
|
| 相关CAS号 |
172820-23-4 (acetate); 147664-63-9
|
| PubChem CID |
16129735
|
| 序列 |
H-Gly-Ile-Gly-Lys-Phe-Leu-Lys-Lys-Ala-Lys-Lys-Phe-Gly-Lys-Ala-Phe-Val-Lys-Ile-Leu-Lys-Lys-NH2.CH3CO2H; Gly-Ile-Gly-Lys-Phe-Leu-Lys-Lys-Ala-Lys-Lys-Phe-Gly-Lys-Ala-Phe-Val-Lys-Ile-Leu-Lys-Lys-NH2;
glycyl-L-isoleucyl-glycyl-L-lysyl-L-phenylalanyl-L-leucyl-L-lysyl-L-lysyl-L-alanyl-L-lysyl-L-lysyl-L-phenylalanyl-glycyl-L-lysyl-L-alanyl-L-phenylalanyl-L-valyl-L-lysyl-L-isoleucyl-L-leucyl-L-lysyl-L-lysinamide acetic acid
|
| 短序列 |
GIGKFLKKAKKFGKAFVKILKK; GIGKFLKKAKKFGKAFVKILKK-NH2
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| LogP |
1
|
| tPSA |
914
|
| 氢键供体(HBD)数目 |
32
|
| 氢键受体(HBA)数目 |
32
|
| 可旋转键数目(RBC) |
94
|
| 重原子数目 |
176
|
| 分子复杂度/Complexity |
4940
|
| 定义原子立体中心数目 |
20
|
| SMILES |
CC[C@H](C)[C@@H](C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC2=CC=CC=C2)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CC3=CC=CC=C3)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N)NC(=O)CN
|
| InChi Key |
DNTVNGJSMLPWQF-CVJDLQKLSA-N
|
| InChi Code |
InChI=1S/C122H210N32O22/c1-13-78(10)103(153-99(155)71-132)121(175)135-73-101(157)139-86(49-25-34-58-125)110(164)152-97(69-82-43-19-15-20-44-82)119(173)150-94(66-75(4)5)117(171)145-90(53-29-38-62-129)113(167)142-87(50-26-35-59-126)109(163)137-79(11)105(159)141-88(51-27-36-60-127)112(166)143-91(54-30-39-63-130)114(168)151-96(68-81-41-17-14-18-42-81)107(161)134-72-100(156)138-85(48-24-33-57-124)108(162)136-80(12)106(160)147-98(70-83-45-21-16-22-46-83)120(174)154-102(77(8)9)122(176)146-92(55-31-40-64-131)115(169)148-95(67-76(6)7)118(172)149-93(65-74(2)3)116(170)144-89(52-28-37-61-128)111(165)140-84(104(133)158)47-23-32-56-123/h14-22,41-46,74-80,84-98,102-103H,13,23-40,47-73,123-132H2,1-12H3,(H2,133,158)(H,134,161)(H,135,175)(H,136,162)(H,137,163)(H,138,156)(H,139,157)(H,140,165)(H,141,159)(H,142,167)(H,143,166)(H,144,170)(H,145,171)(H,146,176)(H,147,160)(H,148,169)(H,149,172)(H,150,173)(H,151,168)(H,152,164)(H,153,155)(H,154,174)/t78-,79-,80-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,102-,103-/m0/s1
|
| 化学名 |
(2S)-6-amino-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[2-[[(2S,3S)-2-[(2-aminoacetyl)amino]-3-methylpentanoyl]amino]acetyl]amino]hexanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]amino]hexanoyl]amino]propanoyl]amino]hexanoyl]amino]hexanoyl]amino]-3-phenylpropanoyl]amino]acetyl]amino]hexanoyl]amino]propanoyl]amino]-3-phenylpropanoyl]amino]-3-methylbutanoyl]amino]hexanoyl]amino]-4-methylpentanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]amino]hexanamide
|
| 别名 |
MSI 78; Locilex; 63S35FF5KS; MSI-78; 172820-23-4; Cytolex; Pexiganan Acetate; Pexiganan (acetate); SCHEMBL487091;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Link: https://clinicaltrials.gov/ct2/show/NCT00563394
Conditions:Diabetic Foot UlcersLink: https://clinicaltrials.gov/ct2/show/NCT00563433
Conditions:Diabetic Foot Ulcers