| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
VHL E3 ligase and HaloTag7 fusion proteins. [1]
|
|---|---|
| 体外研究 (In Vitro) |
一个配体用于 E3 泛素连接酶,另一个配体用于靶蛋白;这两个配体通过接头连接形成 PROTAC。 PROTAC 利用细胞内泛素-蛋白酶体系统特异性破坏靶蛋白[1]。
在稳定表达GFP-HaloTag7的HEK293细胞中,经24小时处理后,流式细胞术检测显示,HaloPROTAC4可诱导超过70%的GFP-HaloTag7降解,但其效果低于HaloPROTAC3(后者可达90%降解)。 [1] 连接链长度研究表明,较长连接链(如HaloPROTAC4中所用)在高浓度下会导致显著的自抑制现象,而中等长度连接链(如HaloPROTAC3中所用)则未观察到自抑制,且最大降解效率更高。 [1] |
| 细胞实验 |
使用稳定表达GFP-HaloTag7的HEK293 Flp-In细胞。用HaloPROTAC4处理细胞24小时。用胰蛋白酶消化细胞并重悬于DMEM中。使用流式细胞仪在FL1通道检测细胞内EGFP荧光强度。平均荧光强度以溶剂对照(0.1% DMSO)归一化。绘制剂量反应曲线以评估降解效力。 [1]
|
| 参考文献 | |
| 其他信息 |
HaloPROTAC4是HaloPROTAC系列化合物之一,旨在通过募集VHL E3连接酶降解HaloTag7融合蛋白。[1]
它含有酚类降解诱导剂Moisty B,并带有更长的聚乙二醇连接臂。[1] 研究发现连接臂长度对活性至关重要,较长的连接臂在高浓度下会导致自身抑制。[1] HaloPROTAC被认为是一种用于在胞质环境中靶向降解HaloTag7融合蛋白的化学遗传工具。[1] |
| 分子式 |
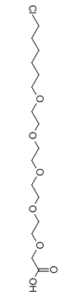
C16H31CLO7
|
|---|---|
| 分子量 |
370.866145372391
|
| 精确质量 |
370.175
|
| CAS号 |
1799506-30-1
|
| PubChem CID |
134694956
|
| 外观&性状 |
Colorless to light yellow ointment
|
| LogP |
1.1
|
| tPSA |
83.4
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
7
|
| 可旋转键数目(RBC) |
20
|
| 重原子数目 |
24
|
| 分子复杂度/Complexity |
267
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
JKRQJQUHILRKGV-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H31ClO7/c17-5-3-1-2-4-6-20-7-8-21-9-10-22-11-12-23-13-14-24-15-16(18)19/h1-15H2,(H,18,19)
|
| 化学名 |
2-[2-[2-[2-[2-(6-chlorohexoxy)ethoxy]ethoxy]ethoxy]ethoxy]acetic acid
|
| 别名 |
PROTAC Linker 4
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
Ethanol : ~100 mg/mL (~269.64 mM)
DMSO : ~100 mg/mL (~269.64 mM) |
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6964 mL | 13.4818 mL | 26.9636 mL | |
| 5 mM | 0.5393 mL | 2.6964 mL | 5.3927 mL | |
| 10 mM | 0.2696 mL | 1.3482 mL | 2.6964 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
 Schematic depiction of a bifunctional HaloPROTAC containing chloroalkane (which binds HaloTag7 fusion proteins) and a hydroxyproline derivative which binds VHL.
A) The enantiomers of HaloPROTACs (containing D-amino acid residues) which do not bind VHL do not induce degradation of GFP-HaloTag7, supporting the necessity of VHL binding for activity. B) Pre-treatment with excessent-HaloPROTAC3 (1 hour) prevents degradation of GFP-HaloTag7 by HaloPROTAC3 after 24 hours. C) Pre-treatment with epoxomicin (4 hours) prevents degradation of GFP-HaloTag7 by HaloPROTAC3 after 20 hours. D)Treatment with VL285 attenuates the ability of HaloPROTAC3 to induce the degradation of GFP-HaloTag7. E) Structure of VL285. All error bars depict SEM.ACS Chem Biol.2015 Aug 21;10(8):1831-7. |
|---|
 The average fluorescence per cell compared to vehicle control was measured by flow cytometry after 24 hour treatment with the indicated compounds and concentrations.
A) Comparison of HaloPROTAC3 (quintuplicate) to Hyt36 (triplicate) shows that HaloPROTAC3 is significantly more potent and efficacious. B) HaloPROTAC3 leads to 50% degradation of GFP-HaloTag7 within 4 to 8 hours. C) Significant recovery from 24 hour treatment with HaloPROTAC3 is observed after a 24 hour washout.ACS Chem Biol.2015 Aug 21;10(8):1831-7. |
 A) A study of linker length with Degradation Inducing Moiety B shows that three ethylene glycol units are optimal for the degradation of GFP-HaloTag7. B) Structures of HaloPROTACs that have weaker affinity for VHL. C) Reducing the affinity for VHL attenuates their ability to induce degradation of GFP-HaloTag7, although the effect is not necessarily linear.
Immunoblotting confirms that nearly complete degradation of A) GFP-HaloTag7 is observed after 24 hour treatment with 500 nM HaloPROTAC3, with significant degradation at 50 nM HaloPROTAC3. HaloPROTAC3 can lead to degradation of other HaloTag7 fusion proteins such as B) HaloTag7-ERK1 and HaloTag7-MEK1.ACS Chem Biol.2015 Aug 21;10(8):1831-7. |