| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
The active components of Caterpillar Leaf are absorbed orally via chewing the leaves. Peak plasma concentrations of cathinone, cathinin, and norephedrine were reached at 127 min, 183 min, and 200 min, respectively. Four volunteers chewed a dose of Caterpillar Leaf equivalent to one-quarter of a typical Caterpillar Leaf intake. Blood samples were collected for up to 80 hours, and alkaloids were determined using gas chromatography-mass spectrometry. Data were evaluated using a computed pharmacokinetic compartmental model. Plasma concentration-time data for alkaloids can be described using a two-compartment model with two absorption stages. The oral mucosa is considered the first site of absorption, where most alkaloids are absorbed (mean absorption rate of cathinone was 59 ± 21%, and ofcacin was 84 ± 6%). Chewing the leaves resulted in highly efficient extraction of alkaloids, with only 9.1 ± 4.2% residue. Cathinone was eliminated from the central compartment with a mean half-life of 1.5 ± 0.8 hours. The half-life of cathinone is 5.2 ± 3.4 hours. The metabolism of cathinone to norephedrine significantly affects its plasma concentration profile. Chewing cathinone has virtually no effect on psychophysiological function. Peak plasma concentrations of cathinone are reached 1.5 to 3.5 hours after the start of chewing. One hour after chewing 60 grams of fresh cathinone, the average plasma concentration of cathinone can reach 100 ng/mL. Eight hours later, cathinone is almost undetectable in the blood. …Only 2% of cathinone is excreted unchanged in the urine. A small amount of cathinone is excreted unchanged via the kidneys (0.6% to 3.3%). Metabolites/Metabolites: Cathinone is metabolized via ketone reduction to norephedrine and norpseudoephedrine in a ratio of approximately 9:1. …Up to 52% of the cathinone isomers are recovered in 24-hour urine samples as amino alcohol metabolites after oral administration. Cathinone is primarily excreted in the urine: 40% of the oral dose is excreted within 6 hours, and 84.6% within 24 hours. Following oral administration of synthetic cathinone (isomers and racemic mixtures), 22-52% of cathinone was recovered from urine samples after 24 hours, mainly as amino alcohol metabolites. GC/MS, HPLC, and CD analysis identified R/S-(-)-norephedrine as the major metabolite of S-(-)-cathinone and R/R-(-)-norpseudamine as the major metabolite of R-(+)-cathinone. Both amino alcohols are generated by stereoselective ketone reduction reactions. First-pass metabolism of cathinone in the liver leads to the formation of norephedrine. The elimination half-life of cathinone is 1.5 ± 0.8 hours. The elimination half-life of cathinone (administered in the form of catechu) is approximately 4 hours. Six volunteers who had never taken any medication before received a single dose of catechu, equivalent to 0.8 mg/kg body weight… The terminal elimination half-life was 260 ± 102 minutes. |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
Interactions
This study used a drug discrimination paradigm to evaluate the effect of combined administration of caffeine and nicotemid on the discrimination of low-dose cathinone. In a two-bar food-motivated maneuver task training rats to distinguish 0.8 mg/kg L-cathinone and its solvent, 0.2 mg/kg cathinone elicited a 29.2% response rate on the lever corresponding to cathinone. With administration of 25 mg/kg nicotemid and 20 mg/kg caffeine, the number of trials with the lever selected by rats was 0% and 50%, respectively. Caffeine, nicotemid, or caffeine plus nicotemid in combination with low-dose cathinone all produced significant discrimination abilities similar to those of cathinone. The enhancing effect of caffeine and nicotemid on cathinone reflects combinations of similar components in abused drugs, particularly the anti-lipid drug X-112, which contains all three components and is widely abused in Europe. This study investigated the effects of (-)-cathinone, caffeine, and their combinations on sexual behavior in male rats. In the absence of female rats, male rats' sexual activity was assessed by recording erectile responses (including genital grooming, yawning/stretching, and same-sex mounting). Mating behavior was observed by accommodating male rats with female rats induced into estrus through subcutaneous injections of estradiol benzoate and progesterone. Mating patterns (mounting, insertion, ejaculation, and refractory period) in male rats were recorded. Oral administration of cathinone (5 mg/kg/day), caffeine (50 mg/kg/day), and combinations thereof for 15 consecutive days increased sexual arousal (motivation) in male rats, manifested as increased mating behavior and anorectal exploration. However, erectile and ejaculatory responses measured in this study did not show an aphrodisiac effect. Based on the current results, it can be inferred that cathinone (the psychostimulant component of catechu) altered behavioral patterns in male rats, and that caffeine, when administered concurrently, also altered the effects of cathinone. However, our data do not provide any evidence that cathinone has an aphrodisiac effect. |
| 其他信息 |
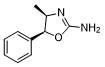
Cathinone is the S-stereoisomer of 2-aminophenylacetone. It is a central nervous system stimulant and psychotropic drug. It is a 2-aminophenylacetone compound and a monoamine alkaloid. Cathinone is a Class I controlled substance under the U.S. Drug Enforcement Administration (DEA). Class I controlled substances currently have no recognized medical use in the United States, lack safety under medical supervision, and have a high potential for abuse. It is a stimulant. Cathinone is a monoamine alkaloid found in the shrub Catha edulis. It is closely associated with ephedrine, cathinone, and other amphetamines and may be the primary source of the stimulant effects of Catha edulis. Cathinone differs from many other amphetamines in that its structure is keto. Other amphetamines with this structure include the antidepressant bupropion and the stimulant methcathinone. Internationally, cathinone is listed as a Class I controlled substance. According to international law, cathinone was added to the List of Class I Controlled Substances under the Controlled Substances Act in 1993. Cathinone has been reported to be found in Catha edulis, Ephedra sinica, and other organisms with relevant data. Mechanism of Action: Cathinone, the active ingredient in Catha edulis, has shown sustained analgesic effects in a rat tail-flick test. Researchers tested the roles of monoamine neurotransmitters, endogenous opioids, and stress responses in this analgesic effect. Reserpine or p-chlorophenylalanine depleted catecholamines or serotonin, respectively, while nomiphenecin inhibited neuronal uptake of biogenic amines and amphetamines; all of these drugs blocked the early (30 minutes) and late (24 hours) analgesic effects of cathinone. High doses (4 mg/kg) of naloxone also produced the same inhibitory effect. However, rats tolerant to morphine retained the early and late analgesic responses to cathinone. In the tail-flip test, cathinone significantly enhanced the increase in plasma ACTH at 30 minutes and 24 hours, and this enhancement could be reversed by naloxone. However, neither dexamethasone nor adrenalectomy could prevent the analgesic response at these time points. ...The sustained analgesia induced by cathinone is mainly due to its amphetamine-like activation of the monoaminergic pathway, but requires the integrity of non-μ-opioid mechanisms. The adrenal-pituitary axis is less likely to be involved in the action of cathinone.
Caterpillar leaf infusion is a traditional African remedy for respiratory diseases. The benefits of catechu are believed to stem from the activity of its main active ingredient, cathinone. Cathinone inhibits electric field-induced acetylcholine release and smooth muscle contraction, which may explain its beneficial effects on airway diseases. The mechanism of action of this natural product appears to be through activation of presynaptic α2-adrenergic receptors and 5-hydroxytryptamine 7 receptors. This new study describes how cathinone modulates airway tone and may, in part, explain the use of catechu as a traditional remedy for relieving symptoms of respiratory diseases. The pharmacological properties of (-)-cathinone, the alkaloid that imparts the stimulating properties to catechu leaves, are very similar to those of (+)-amphetamine. Since amphetamine is known to induce the release of serotonin from storage sites in the central nervous system, experiments were conducted to determine whether (-)-cathinone also possesses this effect. When comparing the effects of (-)-cathinone and (+)-amphetamine on radioactive release in rat striatum tissue pre-labeled with 3H-5-hydroxytryptamine, the release effect of (-)-cathinone was found to be similar to that of (+)-amphetamine, although its potency was only one-third that of the latter. Therefore, the catechu alkaloid (-)-cathinone appears to play an important role in serotonergic neurotransmission, similar to (+)-amphetamine. The effects of (-)-cathinone and (+)-amphetamine, the main psychoactive alkaloids of catechu, in the anterior caudate-putamen and nucleus accumbens were compared. Using in vivo microdialysis, extracellular levels of dopamine and its metabolites were simultaneously measured in two brain regions following intraperitoneal injection of 0.8, 1.6, or 3.2 mg/kg amphetamine or cathinone (doses expressed as salts). Both drugs increased dopamine levels in a dose-dependent manner while decreasing metabolite levels. However, the relative strength of these effects depended on the specific drug, dose, and brain region measured. At the maximum dose, amphetamine had a greater effect on dopamine in both the caudate nucleus and the nucleus accumbens than cathinone. However, at lower doses, this difference was only observed in the nucleus accumbens at a dose of 1.6 mg/kg. The results also indicated that at a dose of 3.2 mg/kg, the effects of the two drugs differed across brain regions, with a greater effect on dopamine in the caudate nucleus than in the nucleus accumbens. These findings suggest functional heterogeneity in the rat striatum, which may be relevant to understanding normal brain function and the nervous system's response to psychoactive drugs. For more complete data on the mechanisms of action of cathinones (6 in total), please visit the HSDB record page. |
| 分子式 |
C9H11NO
|
|---|---|
| 分子量 |
176.219
|
| 精确质量 |
176.095
|
| CAS号 |
71031-15-7
|
| PubChem CID |
62258
|
| 外观&性状 |
Pale yellow leaf from petroleum ether
|
| 熔点 |
46.5 °C
|
| LogP |
1.1
|
| tPSA |
43.1
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
11
|
| 分子复杂度/Complexity |
139
|
| 定义原子立体中心数目 |
1
|
| SMILES |
C[C@@H](C(=O)C1=CC=CC=C1)N
|
| InChi Key |
LJQBMYDFWFGESC-VXNVDRBHSA-N
|
| InChi Code |
InChI=1S/C10H12N2O/c1-7-9(13-10(11)12-7)8-5-3-2-4-6-8/h2-7,9H,1H3,(H2,11,12)/t7-,9-/m1/s1
|
| 化学名 |
((+/-)cis-4,5-dihydro-4-methyl-5-phenyl-2-oxazolamine)
|
| 别名 |
(+/-)cis-4-methylaminorex(4S,5R)-4-Methylaminorex
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6747 mL | 28.3736 mL | 56.7472 mL | |
| 5 mM | 1.1349 mL | 5.6747 mL | 11.3494 mL | |
| 10 mM | 0.5675 mL | 2.8374 mL | 5.6747 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。