| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| Other Sizes |
|
| 靶点 |
Lucerastat specifically targets glucosylceramide synthase (GCS), the enzyme catalyzing the rate-limiting step in glycosphingolipid (including globotriaosylceramide, Gb3) biosynthesis. It exhibits a half-maximal inhibitory concentration (IC₅₀) of 1.2 μM against recombinant human GCS. Additionally, Lucerastat shows high selectivity for GCS: its IC₅₀ against other related glycosidases (e.g., β-glucosidase, α-glucosidase, α-galactosidase) exceeds 100 μM, with less than 10% inhibition at 100 μM [2]
|
|---|---|
| 体外研究 (In Vitro) |
1. 法布里病患者来源成纤维细胞(α-Gal A活性不足正常水平的5%)中Gb3的减少:
- 卢塞司他处理72小时后,以剂量依赖性方式降低细胞内Gb3含量(LC-MS/MS检测):0.3 μM浓度下降低25%,3 μM浓度下降低45%,10 μM浓度下降低62%(相较于溶媒对照组);30 μM浓度下达到最大降低幅度(约65%)[2]
2. 人肾小管上皮细胞(HK-2)中的抗炎活性: - 10 μM 卢塞司他处理48小时,可抑制TNF-α诱导的IL-6 mRNA上调,抑制率为38%(qPCR定量),提示其可能在缓解法布里病相关组织炎症中发挥作用[2] 3. 细胞毒性评估(MTT法): - 患者成纤维细胞或HK-2细胞经0.1–30 μM 卢塞司他处理72小时后,无显著细胞毒性,细胞存活率相对于溶媒对照组维持在90%以上[2] 4. 文献[1]简要总结: - 卢塞司他在体外可有效减少法布里病特异性细胞内的Gb3蓄积,且脱靶效应极低[1] Fabry 患者来源的成纤维细胞携带基因型 R220X (<3%)、W162X (<1%) 和 R301G(残留 -GalA 活性;20%)。 |
| 体内研究 (In Vivo) |
1. α-半乳糖苷酶A敲除小鼠(α-Gal A⁻/⁻,法布里病动物模型)中的药效:
- 口服给予卢塞司他(30 mg/kg,每日一次,持续28天,溶解于0.5%甲基纤维素),可显著降低关键受累组织中的Gb3水平:肾脏中降低52%,心脏中降低48%,肝脏中降低45%(LC-MS/MS检测),相较于溶媒处理的α-Gal A⁻/⁻小鼠[2]
2. 肾功能改善: - 卢塞司他处理组小鼠的血清肌酐(Scr)和血尿素氮(BUN)水平相较于溶媒对照组分别降低22%和18%,提示肾功能损伤得到缓解[2] 3. 病理保护作用: - PAS染色和电镜观察显示,卢塞司他处理组小鼠肾小球系膜区Gb3沉积减少,肾小球基底膜增厚趋势被抑制,肾小管上皮细胞空泡变性减轻[2] 4. 文献[1]简要总结: - 卢塞司他可降低法布里病动物模型靶器官的Gb3负荷,并改善肾功能相关指标[1] 在缺乏残余 GalA 活性的情况下,GCS 抑制剂 lucerastat(1200 mg/kg/天膳食混合物)可降低 Gb3[2]。 |
| 酶活实验 |
1. 重组人GCS活性抑制实验:
- 反应体系(50 μL)组成:20 mM Tris-HCl缓冲液(pH 7.4)、50 μM UDP-葡萄糖(补充¹⁴C标记UDP-葡萄糖用于定量)、20 μM C16-神经酰胺、0.5 μg重组人GCS,以及不同浓度的卢塞司他(0.01–10 μM,系列浓度)。
- 孵育:体系在37°C孵育60分钟,随后加入100 μL氯仿-甲醇(2:1,v/v)终止反应。
- 产物分离与检测:离心(12,000×g,10分钟)后收集有机相,氮气吹干后用氯仿-甲醇(9:1,v/v)重悬;通过薄层层析(TLC)分离反应产物葡萄糖神经酰胺,采用放射自显影技术检测其放射性强度。
- IC₅₀计算:以溶媒组为对照计算各浓度卢塞司他对GCS的抑制率,曲线拟合后得出IC₅₀为1.2 μM[2]
2. 对其他糖苷酶的选择性实验: - 调整上述反应体系,将GCS替换为β-葡萄糖苷酶、α-葡萄糖苷酶或α-半乳糖苷酶,并相应调整底物(如用4-甲基伞形酮-β-D-葡萄糖苷检测β-葡萄糖苷酶)。 - 孵育后检测荧光产物量以计算抑制率,结果显示100 μM 卢塞司他对这些脱靶酶的抑制率均<10%[2] |
| 细胞实验 |
细胞活力测定[2]。
细胞类型: Fabry 患者来源的成纤维细胞,基因型为 R301G(残留 GalA 活性;20%)、R220X(<3%)和 W162X(<1%)。 测试浓度: 孵育持续时间: 9 天。 实验结果:剂量依赖性抑制GCS,减少葡萄糖神经酰胺并增加鞘磷脂。 1. 法布里病患者成纤维细胞中Gb3减少实验: - 细胞接种:成纤维细胞以2×10⁴个/孔的密度接种于24孔板,使用含10%胎牛血清的DMEM培养基,在37°C、5% CO₂条件下培养24小时至贴壁。 - 药物处理:吸弃旧培养基,加入含卢塞司他(0.1、0.3、1、3、10、30 μM)或溶媒(0.1% DMSO)的新鲜培养基,每个浓度设3个复孔;每24小时更换一次含药培养基,持续处理72小时。 - 脂质提取:用PBS洗涤细胞2次,加入100 μL裂解液(含0.5% Triton X-100)冰上裂解30分钟;加入200 μL氯仿-甲醇(2:1,v/v),涡旋混匀后离心(12,000×g,10分钟),收集有机相。 - Gb3检测:有机相氮气吹干后用50 μL甲醇重悬,通过LC-MS/MS检测Gb3(d18:1/16:0)浓度,结果以细胞蛋白含量(BCA法测定)标准化[2] 2. 细胞毒性实验(MTT法): - 细胞以5×10³个/孔接种于96孔板,经0.1–30 μM 卢塞司他处理72小时;每孔加入20 μL MTT溶液(5 mg/mL),孵育4小时后吸弃上清,加入150 μL DMSO溶解甲瓒结晶;在570 nm波长处测定吸光度,以溶媒组为对照计算细胞存活率[2] |
| 动物实验 |
动物/疾病模型:法布里病小鼠(Gla-/0 和 Gla-/-,每性别 n = 5 或 6)[2]。
剂量:1200 mg/kg/天,饲料混合物。 给药途径:饲料混合物,持续 20 周。 实验结果:法布里病影响的两个主要器官中的脂质储存减少:肾脏中 Gb3 平均值降低(-33%,p<0.01),背根神经节中 α-半乳糖末端糖鞘脂含量降低(-48%,p<0.05)。在法布里病小鼠的肝脏中,除Gb3 (24:1)(-37%,p<0.05)外,平均葡萄糖基神经酰胺(GlcCer (24:0))水平降低(-59%,p<0.001),表明GCS抑制导致底物减少。 1. α-Gal A⁻/⁻小鼠体内疗效研究:- 动物:将8周龄雄性α-Gal A⁻/⁻小鼠(C57BL/6背景,22-25 g)随机分为两组(每组n=8):溶剂对照组和Lucerastat治疗组。- 药物制备:将Lucerastat超声溶解于0.5%甲基纤维素溶液中,浓度为3 mg/mL;溶剂对照组为0.5%甲基纤维素溶液。 - 给药:每日一次灌胃给予Lucerastat,剂量为30 mg/kg(10 mL/kg体积,每周根据体重调整),并给予等体积的溶剂对照,连续28天。- 监测:每日观察小鼠的一般状况(活动、食物/饮水摄入量),并每周称重。- 样本采集:28天后,小鼠禁食12小时,用10%水合氯醛麻醉,并通过心脏穿刺采集血液。血清经离心(3,000×g,15分钟)分离,用于Scr/BULT/AST检测。肾脏、心脏和肝脏被取出:每种组织约100 mg冷冻于-80°C用于脂质提取,其余组织用4%多聚甲醛固定,用于组织病理学检查[2] |
| 药代性质 (ADME/PK) |
1. C57BL/6 小鼠(口服 30 mg/kg Lucerastat)的药代动力学参数: - 血液采样:在给药后 0.25、0.5、1、2、4、6、8、12 小时收集血浆(n=5/时间点),并通过 LC-MS/MS 进行分析。 - 主要参数:峰浓度 (Cmax) = 8.7 μM,达峰时间 (Tmax) = 0.5 小时,消除半衰期 (t₁/₂) = 3.2 小时,曲线下面积 (AUC₀-∞) = 26.4 μM·h [2]
2. 组织分布:- 给药后 2 小时,肾脏 (15.2 μM)、心脏 (12.8 μM) 和肝脏 (9.5 μM) 中的 Lucerastat 浓度高于血浆浓度 (6.3 μM) [2] 3. 口服生物利用度:- 通过比较口服 (30 mg/kg) 和静脉(尾静脉,30 mg/kg)给药的 AUC₀-∞ 计算得出,口服生物利用度为 45% [2] 4. 排泄:- 给药后 24 小时内,30% 的药物排出体外药物剂量主要经尿液排出,25%经粪便排出,主要以原形排出。卢塞拉司他[2] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. C57BL/6 小鼠的重复给药毒性(28 天,口服 10/30/100 mg/kg Lucerastat): - 一般毒性:未观察到死亡或异常行为。在 100 μM 剂量下,体重 (28.5±1.2 g) 与载体组 (29.1±1.0 g) 相当 [2]
2. 血清生化指标:- 在 100 mg/kg 剂量下,ALT (25±3 U/L)、AST (52±4 U/L)、Scr (45±3 μmol/L) 和 BUN (5.2±0.4 mmol/L) 与载体组(ALT 23±2 U/L、AST 50±3 U/L、Scr 43±2 μmol/L、BUN 5.0±0.3 mmol/L)相比无显著差异 [2] 3. 组织病理学:- 在 100 mg/kg 剂量下,肾脏、心脏或肝脏组织中未观察到明显的病理损伤(例如炎症、坏死)[2] 4. 血浆蛋白结合率:- 通过平衡透析法测定(37°C, 4 小时)使用 Lucerastat (1/10/100 μM) 和小鼠血浆。血浆蛋白结合率为 85%±3%,且与浓度无关 [2] 5. 文献简述 [1]: - Lucerastat 具有良好的临床前安全性,无明显的肝肾毒性和较低的脱靶效应 [1] |
| 参考文献 | |
| 其他信息 |
Lucerastat 正在临床试验 NCT03425539(Lucerastat 口服单药治疗成人法布里病患者的疗效和安全性)中进行研究。
药物适应症 治疗法布里病 1. 机制和治疗作用:- Lucerastat 是一种口服亚氨基糖,用于法布里病的底物减少疗法 (SRT)。它通过抑制GCS来减少Gb3的合成,从而解决α-半乳糖苷酶A缺乏引起的Gb3积累的核心病理[1][2] 2. 相对于现有SRT的优势: - 与传统SRT(例如,米格鲁司他)相比,卢塞拉司他具有更高的GCS抑制活性(更低的IC₅₀)和更好的GCS选择性,可能减少胃肠道副作用(例如,腹泻、腹胀)[1][2] 3. 临床开发状态(截至2020年): - 卢塞拉司他已完成I期临床试验,显示出良好的药代动力学和安全性。该药物正在进行 II 期临床试验,以评估其对法布里病患者的疗效(例如,血浆 Gb3 水平降低、患者报告结局)[1] 4. 多靶点潜力:- 临床前数据表明,Lucerastat 除了降低 Gb3 水平外,还可能通过抗炎作用(例如,抑制 IL-6)减轻组织损伤[2] |
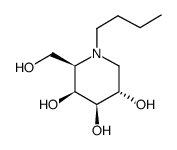
| 分子式 |
C10H21NO4
|
|---|---|
| 分子量 |
219.281
|
| 精确质量 |
219.147
|
| 元素分析 |
C, 54.77; H, 9.65; N, 6.39; O, 29.18
|
| CAS号 |
141206-42-0
|
| PubChem CID |
501391
|
| 外观&性状 |
White to yellow solid powder
|
| LogP |
-0.6
|
| tPSA |
84.16
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
4
|
| 重原子数目 |
15
|
| 分子复杂度/Complexity |
190
|
| 定义原子立体中心数目 |
4
|
| SMILES |
CCCCN1C[C@@H]([C@H]([C@H]([C@H]1CO)O)O)O
|
| InChi Key |
UQRORFVVSGFNRO-XFWSIPNHSA-N
|
| InChi Code |
InChI=1S/C10H21NO4/c1-2-3-4-11-5-8(13)10(15)9(14)7(11)6-12/h7-10,12-15H,2-6H2,1H3/t7-,8+,9+,10-/m1/s1
|
| 化学名 |
(2R,3S,4R,5S)-1-butyl-2-(hydroxymethyl)piperidine-3,4,5-triol
|
| 别名 |
Lucerastat; 141206-42-0; (2R,3S,4R,5S)-1-butyl-2-(hydroxymethyl)piperidine-3,4,5-triol; GVS3YDM418; ACT-434964; DTXSID60161601; lucerastatum; DTXCID2084092; N-(n-Butyl)deoxygalactonojirimycin; NBDGJ
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ~24 mg/mL (~109.45 mM)
DMSO : ~22 mg/mL (~100.33 mM) |
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.5604 mL | 22.8019 mL | 45.6038 mL | |
| 5 mM | 0.9121 mL | 4.5604 mL | 9.1208 mL | |
| 10 mM | 0.4560 mL | 2.2802 mL | 4.5604 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。