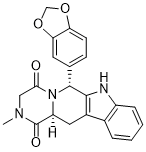
他达拉非 (6R ,12aS)- 是他达拉非的顺式构象异构体,是有效的 PDE5(磷酸二酯酶 5)抑制剂,主要用于治疗勃起功能障碍、良性前列腺增生和原发性肺动脉高压。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Tadalafil and Pembrolizumab in Recurrent or Metastatic Head and Neck Cancer
CTID: NCT03993353
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-18
Study of Tadalafil Vs. Placebo for Improving Hemodynamics and End-Organ Dysfunction in Fontan Physiology
CTID: NCT05206955
Phase: Phase 3 Status: Recruiting
Date: 2024-11-12
Tadalafil as Adjuvant Therapy for DMD
CTID: NCT05195775
Phase: Phase 2/Phase 3 Status: Active, not recruiting
Date: 2024-11-06
Clinical Study to Compare the Efficacy and Safety of Macitentan and Tadalafil Monotherapies With the Corresponding Fixed-dose Combination Therapy in Subjects With Pulmonary Arterial Hypertension (PAH)
CTID: NCT03904693
Phase: Phase 3 Status: Completed
Date: 2024-11-06
The Effects of Mirabegron and Tadalafil on Glucose Tolerance in Prediabetics
CTID: NCT05051436
Phase: Phase 4 Status: Recruiting
Date: 2024-11-04
View More
Use of a Phosphodiesterase Type 5 Inhibitor to Improve Anabolic Resistance in Older Adults
CTID: NCT05458232
Phase: Status: Withdrawn
Date: 2024-11-04
Efficacy of Tadalafil (5mg) For Treatment of Early Storage Symptoms and Erectile Dysfunction After Endoscopic Enucleation of Prostate
CTID: NCT05955001
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-10-30
Safety and Efficacy Evaluation of BZ371A Topically Applied on Prostatectomized Patients
CTID: NCT05558007
Phase: Phase 2 Status: Recruiting
Date: 2024-10-22
A Trial for Prevention of Recurrent Ischemic Priapism in Men With Sickle Cell Disease: A Pilot Study
CTID: NCT05142254
Phase: Phase 2 Status: Completed
Date: 2024-09-20
Evaluation of Efficacy of Botulinum Toxin A Plus Oral Tadalafil 5 mg in Diabetic Men With Erectile Dysfunction
CTID: NCT06583590
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-09-04
Administration of Tadalafil in Patients With Benign Prostatic Hyperplasia
CTID: NCT06466369
Phase: Phase 3 Status: Completed
Date: 2024-08-23
Evaluation of Tadalafil Combined With LIPUS for Treating Erectile Dysfunction
CTID: NCT06543628
Phase: N/A Status: Not yet recruiting
Date: 2024-08-09
Genetic Factors of Erectile Dysfunction Degree and Response to Tadalafil Treatment in Patients With Diabetes
CTID: NCT06520839
Phase: Phase 3 Status: Recruiting
Date: 2024-07-25
Breathe Easier With Tadalafil Therapy for Dyspnea in COPD-PH
CTID: NCT05937854
Phase: Phase 2 Status: Recruiting
Date: 2024-06-27
Vasodilator and Exercise Study for DMD (VASO-REx)
CTID: NCT06290713
Phase: Phase 2 Status: Recruiting
Date: 2024-06-25
Tadalafil for Severe Pulmonary Hypertension Due to Chronic Obstructive Pulmonary Disease
CTID: NCT05844462
Phase: Phase 3 Status: Recruiting
Date: 2024-06-21
A Single Dose Randomized Five-Way Crossover Pharmacokinetics (PK) Study of Tadalafil Semi-Chewable (Gummy) Formulations in Healthy Volunteers
CTID: NCT04762082
Phase: Phase 1 Status: Not yet recruiting
Date: 2024-06-13
A Study of Fixed Dose Combination of Macitentan/Tadalafil (10 mg/20 mg) Compared to the Reference Free Combination of Macitentan and Tadalafil in Healthy Adult Participants
CTID: NCT05236231
Phase: Phase 1 Status: Completed
Date: 2024-05-22
Investigational and Comparative Study in the Management of Diabetic Nephropathy
CTID: NCT05487755
Phase: Phase 3 Status: Completed
Date: 2024-05-14
Effect of PDE5 Inhibition on Adipose Metabolism in Humans
CTID: NCT04684589
Phase: Phase 2 Status: Recruiting
Date: 2024-04-16
Study of CRS-207, Pembrolizumab, Ipilimumab, and Tadalafil in Metastatic Pancreatic Cancer
CTID: NCT05014776
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-04-09
Management of OAB in Female Patients .
CTID: NCT06184334
Phase: N/A Status: Completed
Date: 2024-04-02
Tadalafil Effect + Chemotherapy in Resectable Gastric/GEJ Cancer
CTID: NCT05709574
Phase: Phase 2 Status: Recruiting
Date: 2024-03-18
Improving Cerebral Blood Flow and Cognition in Patients With Cerebral Small Vessel Disease. The ETLAS-2 Trial
CTID: NCT05173896
Phase: Phase 2 Status: Recruiting
Date: 2024-02-26
The Effects of a Nitrate Supplementation on Erectile Function
CTID: NCT06213077
Phase: N/A Status: Recruiting
Date: 2024-01-19
The Phosphodiesterase Inhibitor Tadalafil as an Adjunct to Antidepressants in Major Depressive Disorder Patients
CTID: NCT05030623
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2023-11-21
Efficacy and Safety of DKF-313 in Patients With Benign Prostatic Hyperplasia
CTID: NCT04947631
Phase: Phase 3 Status: Completed
Date: 2023-09-21
Clinical Investigation Using MED3000 Gel or Tadalafil Tablets in the Treatment of Erectile Dysfunction
CTID: NCT04984993
Phase: Phase 3 Status: Completed
Date: 2023-09-21
Effect of Tadalafil, Sildenafil and Pentoxyfylline on Frozen Embryo Transfer Outcomes
CTID: NCT05971667
Phase: Phase 2/Phase 3 Status: Not yet recruiting
Date: 2023-08-03
Nivolumab (Anti-PD1), Tadalafil and Oral Vancomycin in People With Refractory Primary Hepatocellular Carcinoma or Liver Dominant Metastatic Cancer From Colorectal or Pancreatic Cancers
CTID: NCT03785210
Phase: Phase 2 Status: Completed
Date: 2023-07-11
Effect of Phosphodiesterase-5 Inhibition With Tadalafil on SystEmic Right VEntricular Size and Function
CTID: NCT03049540
Phase: Phase 3 Status: Completed
Date: 2023-06-23
Tadalafil to Overcome Immunosuppression During Chemoradiotherapy for IDH-wildtype Grade III-IV Astrocytoma
CTID: NCT04757662
Phase: Phase 1 Status: Completed
Date: 2023-06-13
Endocrine Cardiomyopathy in Cushing Syndrome: Response to Cyclic GMP PDE5 inhibitOrs
CTID: NCT02611258
Phase: Phase 2 Status: Completed
Date: 2023-05-16
Endocrine Cardiomyopathy: Response to Cyclic GMP PDE5 Inhibitors in Acromegaly Cardiomyopathy
CTID: NCT02611336
Phase: Phase 2 Status: Completed
Date: 2023-05-12
Comparison Between Tamsulosin and Tadalafil in Management of Benign Prostatic Hyperplasia Long Term Study
CTID: NCT05818670
Phase: Phase 4 Status: Completed
Date: 2023-04-19
Efficacy and Safety of Toronto Association in the Treatment of Erectile Dysfunction and Premature Ejaculation
CTID: NCT05052879
Phase: Phase 3 Status: Not yet recruiting
Date: 2023-04-12
Silodosin, Tadalafil Alone vs. Silodosin Plus Tadalafil as MET for Lower Ureteric Stones
CTID: NCT05789732
Phase: N/A Status: Completed
Date: 2023-03-31
The Efficacy of Tamsulosin and Tadalafil Compared to Placebo in the Treatment and Prevention of Urinary Disorders After Transperineal Prostate Biopsy
CTID: NCT05537272
Phase: Phase 4 Status: Enrolling by invitation
Date: 2023-02-22
Trial of Perioperative Tadalafil and Influenza Vaccination in Cancer Patients Undergoing Major Surgical Resection of a Primary Abdominal Malignancy
CTID: NCT02998736
Phase: Phase 1 Status: Completed
Date: 2022-12-28
Perfusion by Arterial Spin Labelling Following Single Dose Tadalafil in Small Vessel Disease (PASTIS) Trial
CTID: NCT02450253
Phase: Phase 2 Status: Completed
Date: 2022-10-27
Window of Opportunity Trial of Nivolumab and Tadalafil in Patients With Squamous Cell Carcinoma of the Head and Neck
CTID: NCT03238365
PhaseEarly Phase 1 Status: Completed
Date: 2022-10-05
A Study of Macitentan/Tadalafil Combination Administered a Fixed-dose Combination Formulation Compared to the Reference Free Combination of Macitentan and Tadalafil
CTID: NCT04540744
Phase: Phase 1 Status: Completed
Date: 2022-09-14
Low Dose Tadalafil for Treatment of Female OAB Syndrome: Short Term Follow up.
CTID: NCT04500860
Phase: Phase 1 Status: Completed
Date: 2022-08-15
Efficacy of Tadalafil/Solifenacin VS Tamsulosin/Solifenacin Combination Therapy for BPH/OAB
CTID: NCT05494567
Phase: Phase 4 Status: Unknown status
Date: 2022-08-10
Marrow Infiltrating Lymphocytes - Non-Small Cell Lung Cancer (MILs™ - NSCLC) Alone or in Combination With Nivolumab With or Without Tadalafil in Locally Advanced and Unresectable or Metastatic NSCLC
CTID: NCT04069936
Phase: Phase 2 Status: Terminated
Date: 2022-08-02
Serum YKL-40 Level and Platelets Indices Among Patients With Diabetic Erectile Dysfunction
CTID: NCT05446493
Phase: Phase 4 Status: Completed
Date: 2022-07-06
Acute Haemodynamic Study of TPN171H in Patients With Pulmonary Arterial Hypertension
CTID: NCT04483115
Phase: Phase 2 Status: Completed
Date: 2022-07-01
PDE5 Inhibition for Obesity-Related Cardiometabolic Dysfunction
CTID: NCT02819440
Phase: Phase 2 Status: Completed
Date: 2022-05-27
Effects of Single Dose Tadalafil on Urethral and Anal Closure Function
CTID: NCT05095077
Phase: Phase 1 Status: Completed
Date: 2022-04-06
Efficiency of Tadalafil for Management of Female Sexual Dysfunction
CTID: NCT05266651
Phase: Phase 2/Phase 3 Status: Unknown status
Date: 2022-03-04
Shock Wave vs. On-demand Tadalafil for Erectile Dysfunction
CTID: NCT05199727
Phase: N/A Status: Unknown status
Date: 2022-01-20
Impact of Tadalafil 5 mg on Post-micturition Dribble in Young-age Men With no/Mild Lower Urinary Tract Symptoms
CTID: NCT05146674
Phase: N/A Status: Unknown status
Date: 2022-01-11
A Study of Tadalafil in Pediatric Participants With Pulmonary Arterial Hypertension (PAH)
CTID: NCT01824290
Phase: Phase 3 Status: Completed
Date: 2021-11-05
Endocan Level in Patients With Erectile Dysfunction and Relationship With Tadalafil Treatment
CTID: NCT05109377
Phase: N/A Status: Unknown status
Date: 2021-11-05
Study of Retinal and Choriocapillary Vascular Changes in Patients Undergoing Tadalafil 20mg
CTID: NCT04164355
Phase: Status: Completed
Date: 2021-09-24
The Effect of Combination Therapy Using Li-ESWT and PDE-5 Inhibitor in Patients With Erectile Dysfunction
CTID: NCT05043896
Phase: N/A Status: Completed
Date: 2021-09-14
Tadalafil for Erectile Dysfunction in Patients With Cirrhosis
CTID: NCT03566914
Phase: N/A Status: Completed
Date: 2021-07-06
Safety and Pharmacokinetic Characteristics of DKF-313
CTID: NCT02352311
Phase: Phase 1 Status: Completed
Date: 2021-07-06
PDE5 Inhibition Via Tadalafil to Enhance Anti-Tumor Mucin 1 (MUC1) Vaccine Efficacy in Patients With HNSCC
CTID: NCT02544880
Phase: Phase 1 Status: Completed
Date: 2021-06-15
Study to Determine How Cialis Effects the Renal Function in Response to Volume Expansion in Preclinical Systolic Cardiomyopathy (Aim2)
CTID: NCT01970176
Phase: Phase 1/Phase 2 Status: Completed
Date: 2021-06-11
Eff
Early intervention and recovery of sexual function in men and women after treatment of rectal cancer–a randomized controlled study with tadalafil compared to standard care
CTID: null
Phase: Phase 2 Status: Trial now transitioned
Date: 2019-07-02
Phosphodiesterase-5 inhibition in patients with heart failure with preserved ejection fraction and combined post- and pre-capillary pulmonary hypertension
CTID: null
Phase: Phase 3 Status: Completed
Date: 2018-07-06
cGMP Enhancing Therapeutic Strategy for HFpEF: The cGETS Study
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2018-02-28
Effect of phosphodiesterase-5 inhibition with Tadalafil on SystEmic Right
CTID: null
Phase: Phase 3 Status: Completed
Date: 2018-01-15
Study on New Insights in Remodeling of Endocrine Cardiomyopathies: Intramyocardial, Molecular and Neuroendocrine Assessment in Response to Chronic Inhibition of Cyclic GMP Phosphodiesterase 5A in Cushing’s Syndrome-Endocrine cardiomyopathy in cushing syndrome: Response to cyclic GMP PDE5 inhibitOrs
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2016-12-12
Study on New Insights in Remodeling of Endocrine Cardiomyopathies: ASsessmentt of Intramyocardial, Molecular and NeUroendocrine Parameters in Response to Chronic Inhibition of Cyclic GMP Phosphodiesterase 5A in AcroMegaly - Endocrine cardiomyophaty: Response to cyclic GMP PDE5 inhibitors in Acromegaly cardiomyopathy
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2016-12-12
A prospective, randomized, international, multicenter, double-arm, controlled, open-label study of Riociguat in patients with pulmonary arterial hypertension (PAH) who are on a stable dose of phosphodiesterase-5 inhibitors (PDE-5i) with or without endothelin receptor antagonist (ERA), but not at treatment goal
CTID: null
Phase: Phase 4 Status: Completed
Date: 2016-11-21
A prospesctive, open-label 12-weeks treatment study to determine the effect of tadalafil 5 mg on clitoral blood flow in menopausal and hipertensive women with sexual interest and arousal disorder
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2016-06-01
Effect of Tadalafil on cerebral large arteries in stroke patients.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2016-05-19
The efficacy and safety of initial triple versus initial dual oral combination
CTID: null
Phase: Phase 3 Status: Completed
Date: 2016-03-04
Prospective, multicenter, open-label study evaluating the effects of first-line oral combination therapy of macitentan and tadalafil in patients with newly diagnosed pulmonary arterial hypertension.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-10-14
Perfusion by Arterial Spin labelling following Single dose Tadalafil In Small vessel disease
CTID: null
Phase: Phase 2 Status: Completed
Date: 2015-06-05
Double-blinded placebo-controlled study on men with lower urinary tract symptoms secondary to prostatic hyperplasia (LUTS-BPH) to assess changes in pressure flow study (PFS) and in molecular profile of prostatic tissue and to correlate this parameters with modifications of symptoms scores (IPSS) after 12 weeks treatment with tadalafil
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2015-04-14
A Randomized, Double-Blind, Placebo-Controlled, Phase 3 Trial of Tadalafil for Duchenne Muscular Dystrophy
CTID: null
Phase: Phase 3 Status: Completed, Prematurely Ended
Date: 2013-09-25
A multiple ascending dose study of Tadalafil to assess the pharmacokinetics and safety in a pediatric population with Pulmonary Arterial Hypertension
CTID: null
Phase: Phase 1, Phase 2 Status: Completed
Date: 2012-01-13
Treatment of patients with metastatic melanoma (AJCC stage IV or III unresectable) with the PDE-inhibitor Tadalafil:
CTID: null
Phase: Phase 2 Status: Completed
Date: 2012-01-10
A double-blind, randomized, placebo-controlled, proof of concept study to investigate the safety and efficacy of the combined administration of 0.5 mg sublingual testosterone and 10 mg tadalafil in women with hypoactive sexual desire disorder
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2011-07-04
A Randomized, Double-Blind, Placebo Controlled Study to Evaluate the Effect of Tadalafil Once Daily for 8 Weeks on Prostatic Blood Flow and Perfusion Parameters in Men with Signs and Symptoms of Benign Prostatic Hyperplasia
CTID: null
Phase: Phase 3 Status: Completed
Date: 2010-12-21
A Phase 3b,randomized,double-blind,placebo-controlled parallel-design study to evaluate the efficacy and safety of tadalafil coadministered with finasteride for 6 months in men with lower urinary tract symptoms(LUTS) and prostatic enlargement secondary to benign prostatic hyperplasia(BPH)
CTID: null
Phase: Phase 3 Status: Completed
Date: 2010-11-05
PHASE CONTRAST MAGNETIC RESONANCE (MR) IMAGING IN MONITORING THE EFFECTS OF TADALAFIL IN PATIENTS WITH OUT-OF-PROPORTION PULMONARY HYPERTENSION AND LEFT VENTRICULAR DYSFUNCTION
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2010-10-14
AMBITION: A Randomised, Multicenter Study of First-Line Ambrisentan and Tadalafil Combination Therapy in Subjects with Pulmonary Arterial Hypertension
CTID: null
Phase: Phase 4 Status: Completed
Date: 2010-08-26
Do Phosphodiesterase 5 Inhibitors Improve Exercise Capacity in COPD Patients with Pulmonary Hypertension?
CTID: null
Phase: Phase 2 Status: Completed
Date: 2010-07-14
Impact of Tadalafil (LY450190) Once a Day or Tadalafil On Demand Compared to Sildenafil Citrate On Demand on Treatment Discontinuation in Patients with Erectile Dysfunction who are Naïve to PDE5 Inhibitors
CTID: null
Phase: Phase 4 Status: Completed
Date: 2010-05-07
A Randomised, Double-Blind, Placebo-Controlled Study to Evaluate the Effect on Unassisted Erectile Function of the Early Use of Tadalafil 5 mg Once a Day and Tadalafil 20 mg On Demand Treatment for 9 Months in Subjects Undergoing Bilateral Nerve-Sparing Radical Prostatectomy
CTID: null
Phase: Phase 4 Status: Completed
Date: 2009-10-23
A Phase 3, Randomized, Double-Blind, Placebo-Controlled, Parallel-Design, Global Multicenter Study to Evaluate the Efficacy and Safety of Tadalafil Once Daily Dosing for 12 Weeks in Men with Signs and Symptoms of Benign Prostatic Hyperplasia.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2009-10-01
The effect of selective PDE-5 inhibition on capillary recruitment, glucose uptake and endothelial function following a mixed meal in patients with type 2 diabetes
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2009-07-27
A Randomized, Double-Blind, PLacebo-Controlled, Parallel Study to Assess the Efficacy and Safety of Tadalafil (LY450190) Once a Day in Subjects With Erectile Dysfunction Who Are Naive to PDE5 Inhibitors
CTID: null
Phase: Phase 3 Status: Completed
Date: 2009-01-30
A Randomized, Double-Blind, Placebo-Controlled, Parallel Design, Multinational Study to Evaluate the Efficacy and Safety of Tadalafil 2.5 and 5 mg Once Daily Dosing for 12 Weeks for the Treatment of Erectile Dysfunction and Signs and Symptoms of Benign Prostatic Hyperplasia in Men With Both Erectile Dysfunction and Benign Prostatic Hyperplasia.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2009-01-19
A Phase 3, Randomized, Double-Blind, Placebo-Controlled, Parallel-Design, Multinational Study to Evaluate the Efficacy and Safety of Daily Tadalafil for 12 Weeks in Men With Signs and Symptoms of Benign Prostatic Hyperplasia
CTID: null
Phase: Phase 3 Status: Completed
Date: 2008-10-28
A Comparison of Psychosocial Outcomes Following Tadalafil Once a Day or PDE5 Inhibitor As Needed in Men With Erectile Dysfunction.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2008-07-30
'Evaluation of erectyle disfonction in patients suffering from testosterone deficiency, before and after tretament by Testopatch'
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2008-06-23
A Randomized, Double-Blind, Placebo-Controlled, Parallel-Design, Multicenter Study to Evaluate the Urodynamic Effects of Tadalafil Once a Day for 12 Weeks in Men With Signs and Symptoms of Benign Prostatic Hyperplasia
CTID: null
Phase: Phase 2 Status: Completed
Date: 2007-12-14
EFFICACY EVALUATION OF TADALAFIL 20 MG ON DEMAND VS TADALAFIL 5 MG DAILY AS TREATMENT MODALITY FOR ERECTILE DYSFUNCTION FOLLOWING RADICAL RADIOTHERAPY IN PROSTATIC CANCER
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2007-07-12
Tadalafil 5 mg Once a Day Compared to Placebo in Improving Erectile Dysfunction
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-10-16
Phase IIIB, Double Blind, Placebo Controlled, International, Multicenter, Parallel Group Study, to Assess the Efficacy and Safety of Testim Gel in Combination with a Phosphodiesterase V Inhibitor (Tadalafil), in Male Patients with Low or Baseline Serum Testosterone Levels and Erectile Dysfunction
CTID: null
Phase: Phase 3 Status: Completed, Prematurely Ended
Date: 2006-09-01
A Randomized, Double-Blind, Placebo-Controlled, Parallel-Design, 5-Group, Multinational Study to Evaluate the Efficacy, Dose Response, and Safety of Tadalafil Once-a-Day Dosing for 12 Weeks in Men With Signs and Symptoms of Benign Prostatic Hyperplasia
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-07-13
Double-blind, placebo controlled randomized study of the effects of co-administering testosterone with PDE V inhibitor in ED patients non responders to PDE V inhibitors alone.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-06-14
A Double-Blind, Extension Study to Evaluate the Long-Term Safety and Efficacy of the Phosphodiesterase Type 5 (PDE5) Inhibitor Tadalafil in the Treatment of Patients with Pulmonary Arterial Hypertension
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-04-10
A Randomized, Double-Blind, Placebo-Controlled Phase 3 Study of the Phosphodiesterase Type 5 (PDE5) Inhibitor Tadalafil in the Treatment of Patients with Pulmonary Arterial Hypertension
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-04-10
A Randomized, Double-Blind, Parallel-Design, Placebo-Controlled Study to Evluate the Efficacy and Safety of Tadalafil (2.5 mg e 5 mg) administered once daily to men with diabetes mellitus and erectile dysfunction
CTID: null
Phase: Phase 3 Status: Completed
Date: 2004-09-22
Effect of Tadalafil on the Quality of Life and Sexual Life Satisfaction in Erectile Dysfunction (ED) Patients Previously Treated with other Oral ED therapy
CTID: null
Phase: Phase 4 Status: Completed
Date: 2004-08-03
A Double-Blind Efficacy and Safety Study of the Phosphodiesterase Type 5 Inhibitor Tadalafil in Pediatric Patients with Pulmonary Arterial Hypertension
CTID: null
Phase: Phase 3 Status: Ongoing, Prematurely Ended, Completed
Date:
A randomized controlled study of the efficacy of Tadalafil monotherapy versus combination of Tadalafil and mirabegron for the treatment of overactive bladder (OAB) associated with benign prostatic hyperplasia (BPH)
CTID: UMIN000025282
Phase: Status: Complete: follow-up complete
Date: 2016-12-15
A multicenter phase II trial of the efficacy and safety of tadalafil with pre-eclampsia.
CTID: UMIN000024042
Phase: Phase II Status: Complete: follow-up complete
Date: 2016-09-15
A multicenter phase II trial of the efficacy and safety of tadalafil in fetus with early-onset growth restriction.
CTID: UMIN000023778
Phase: Phase II Status: Complete: follow-up complete
Date: 2016-08-26
Study on the effect of tadalafil on bladder blood flow in patients with lower urinary tract symptoms due to benign prostatic hyperplasia
CTID: UMIN000020658
PhaseNot applicable Status: Complete: follow-up complete
Date: 2016-02-01
A Randomized, Open-Label, Multicenter study to evaluate effect of tadalafil on lower urinary tract symptoms in patients with prostate cancer treated after radiotherapy (T-addon-RT trial)
CTID: UMIN000020674
Phase: Status: Complete: follow-up complete
Date: 2016-01-21
A Randomized, Open-Label, Multicenter study evaluating efficacy of switch from dutasteride to tadalafil in benign prostatic hyperplasia patient with lower urinary tract symptoms
CTID: UMIN000020369
PhaseNot applicable Status: Complete: follow-up complete
Date: 2016-01-20
Open, parallel, prospective, randomized study between tamsulosin and tadalafil for male LUTS patients.
CTID: UMIN000020362
Phase: Status: Complete: follow-up complete
Date: 2016-01-01
Effect of tadalafil on bladder blood flow in patients with lower urinary tract symptoms secondary to benign prostate hyperplasia
CTID: UMIN000016034
Phase: Status: Complete: follow-up complete
Date: 2015-12-22
Tadalafil treatment for Fetus with Early onset growth Restriction
CTID: UMIN000020044
Phase: Phase I Status: Complete: follow-up complete
Date: 2015-12-03
The impact of the associated symptoms of other LUTS by tadalafil administration to patients with BPH
CTID: UMIN000020040
Phase: Status: Complete: follow-up complete
Date: 2015-12-03
Evaluation of silodosin versus tadalafil in patients with urination disorders associated with benign prostatic hyperplasia
CTID: UMIN000018743
Phase: Status: Complete: follow-up complete
Date: 2015-08-21
The study for evaluating the additional effect of tadalafil in patients with the residual lower urinary tract storage symptoms even after treatment with alpha-1-adrenoreceptor antagonists
CTID: UMIN000017896
Phase: Status: Pending
Date: 2015-07-01
Efficacy of tamsulosin or tadalafil for voiding disorders in I-125 prostate seed implant patients
CTID: UMIN000017825
Phase: Status: Complete: follow-up complete
Date: 2015-06-06
Tadalafil monotherapy versus tadalafil and solifenacin combined therapy for patients with lower urinary tract symptoms suggestive of benign prostate hyperplasia and overactive bladder: randomized controlled trial
CTID: UMIN000016805
Phase: Status: Complete: follow-up complete
Date: 2015-03-15
The role of tadalafil for lower urinary tract symptoms after brachytherapy
CTID: UMIN000015767
PhaseNot applicable Status: Pending
Date: 2014-12-01
Add-on effects of tadalafil for tamsulosin treated patients with benign prostatic hyperplasia suffering from residual lower urinary tract symptoms: a randomized, placebo-controlled, double-blind, crossover study
CTID: UMIN000014769
PhaseNot applicable Status: Complete: follow-up complete
Date: 2014-10-01
None
CTID: jRCT2080222487
Phase: Status:
Date: 2014-05-12
H6D-MC-LVHV
CTID: jRCT2080222307
Phase: Status: completed
Date: 2013-11-22
H6D-MC-LVHV
CTID: jRCT2080222307
Phase: Status: completed
Date: 2013-11-22
Effectiveness for endothelial function after administration of tadarafil for men with urinary and sexual symptoms
CTID: UMIN000009580
Phase: Status: Complete: follow-up complete
Date: 2013-01-01
None
CTID: jRCT2080221678
Phase: Status:
Date: 2011-12-21
Tadalafil study for pediatric PAH on efficacy, safety and pharmacodynamics
CTID: UMIN000005973
PhaseNot applicable Status: Complete: follow-up complete
Date: 2011-08-01
Drug interactions and effects of combination therapy for pulmonary arterial hypertension
CTID: UMIN000005464
PhaseNot applicable Status: Recruiting
Date: 2011-04-19