| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Androgen Receptor
|
|---|---|
| 体外研究 (In Vitro) |
与所有睾酮酯一样,丙酸睾酮在血浆中迅速水解为游离睾酮。睾酮通过两种不同的途径代谢为17酮类固醇。主要的活性代谢产物是雌二醇和二氢睾酮(DHT)。丙酸睾酮被迅速水解成睾酮。睾酮通过两种不同的途径代谢为17酮类固醇。主要的活性代谢产物是雌二醇和二氢睾酮(DHT)。消除途径:肌肉注射约90%的睾酮以睾酮及其代谢产物的葡萄糖醛酸和硫酸结合物的形式在尿液中排出;大约6%的剂量通过粪便排出,大部分以未结合的形式排出。
|
| 体内研究 (In Vivo) |
丙酸睾酮用于小母牛的兽医实践,以刺激最大限度的生长。丙酸睾酮的给药可以诱导与男性性发育相关的蛋白质的产生。临床试验表明,在给予丙酸睾酮后,血浆LH降低。
|
| 细胞实验 |
丙酸睾酮 (TP) 的配制 [13]
将丙酸睾酮 (TP) 溶解于丙酮-水混合液(1:3 体积比)中,于 70°C 加热 15 分钟。冷却后,与完全培养基混合,得到终浓度为 10⁻¹⁰ M、10⁻⁹ M、10⁻⁸ M、10⁻⁷ M 和 10⁻⁶ M 的 TP 溶液。细胞培养物中丙酮的最终体积为 0.3%,添加了 0.3% 丙酮的培养基用作载体对照。将骨髓间充质干细胞 (BM-MSCs) 分别与浓度为 10⁻⁶ M 至 10⁻¹⁰ M 的 TP 或载体对照孵育 24 小时,同时使用未处理的细胞作为空白对照。 MB-MSCs 的细胞增殖测定 [13] 通过羧基荧光素琥珀酰亚胺酯 (CFSE) 染色测定 BM-MSCs 的增殖率,该方法依赖于细胞分裂时荧光染料在两个子细胞间的均等分配。在本研究中,将 2x10⁵ 个 BM-MSC 按一式三份接种于 6 孔组织培养板中,并孵育过夜使其贴壁。为标记细胞,弃去完全培养基,在含有终浓度为 5 µM CFSE 的 1 ml DMEM 中于 37°C 孵育细胞 20 分钟。弃去含有 CFSE 的培养基,然后用浓度为 10⁻⁶ M 至 10⁻¹⁰ M 的 TP 或载体对照处理细胞。24 小时孵育期结束时,通过胰蛋白酶消化收集细胞,用 DPBS 洗涤一次,并使用贝克曼库尔特 FC500 流式细胞仪进行检测。使用 CXP 软件进行分析。 MB-MSCs 的细胞活力和凋亡测定 [13] 使用 ApopNexin Annexin V FITC 凋亡检测试剂盒,通过膜联蛋白 V/碘化丙啶 (PI) 染色,测定不同浓度 TP 对 MSCs 活力和凋亡率的影响。该方法的原理是同时分析在凋亡过程中逐渐丧失的膜不对称性和完整性。为此,将 2x10⁵ 个 BM-MSCs 按一式三份接种于 6 孔组织培养板中,并用浓度为 10⁻⁶ M 至 10⁻¹⁰ M 的 TP 或载体对照处理 24 小时,同时使用未处理的细胞作为对照。孵育结束后,通过胰蛋白酶消化收集细胞,用 DPBS 洗涤一次,然后根据制造商的说明,用 Annexin V-FITC 和 PI 进行标记。简言之,将细胞重悬于 1 ml Annexin V 结合缓冲液中,然后与试剂盒提供的 3 µl Annexin V FITC 和 2 µl PI 在室温避光条件下孵育 15 分钟。孵育结束后将细胞置于冰上,并立即使用贝克曼库尔特 FC500 流式细胞仪进行分析。使用 CXP 软件进行分析。 TP 处理后 BM-MSCs 的表征 [13] 通过检测细胞表面标志物和评估 BM-MSCs 的分化能力,确定浓度为 10⁻⁸ M 的 TP 对 BM-MSCs 特征特性的影响。对于免疫表型分析,将 2x10⁵ 个 BM-MSCs 按一式三份接种于 6 孔组织培养板中,在 10⁻⁸ M TP 存在下孵育 24 小时,然后通过胰蛋白酶消化收集细胞并用上述抗体进行标记。立即使用贝克曼库尔特 DxFLEX 流式细胞仪读取数据,并使用 CytExpert 软件进行分析。对于 BM-MSCs 分化能力的评估,将 1x10⁴ 个 BM-MSCs 接种于 96 孔细胞培养板中,待细胞融合度达 80% 时,使用市售的软骨形成、成骨和成脂分化试剂盒(无论是否添加 10⁻⁸ M TP)诱导分化。每周更换两次分化培养基。分化第 21 天,用 10% 中性缓冲福尔马林溶液固定细胞。分别通过阿利新蓝、茜素红和油红 O 染色评估软骨形成、成骨和成脂分化。 BM-MSCs 与 K562 细胞系的共培养 [13] K562 细胞系 (ATCC #CCL-243) 在补充有 10% 胎牛血清 (FBS) 和 1% 青霉素/链霉素抗生素溶液的 RPMI 培养基中培养。为评估 BM-MSCs 对 K562 细胞系的细胞毒性作用,将 BM-MSCs 接种于 35 mm 细胞培养皿中(1x10⁴ 个细胞/皿),并培养过夜使其贴壁。然后,弃去培养基,BM-MSCs 或先用 10⁻⁸ M TP 处理 24 小时,再与 K562 细胞(1x10⁵ 个细胞/皿)共培养额外的 24 小时;或者,在 10⁻⁸ M TP 存在下将 K562 细胞加到 BM-MSCs 上,并孵育 24 小时。TP 对 K562 细胞的影响通过将其与补充了 10⁻⁸ M TP 的培养基孵育 24 小时来确定,而 BM-MSCs 对 K562 细胞的影响则通过共培养细胞 24 小时来评估。K562 细胞的活力通过用 4',6-二脒基-2-苯基吲哚 (DAPI) 在室温避光条件下孵育细胞 15 分钟进行标记来确定。使用贝克曼库尔特 DxFLEX 流式细胞仪读取细胞,并使用 CytExpert 软件进行分析。 共聚焦显微镜检查 [13] 为评估 TP 处理后 BM-MSC 的极化和细胞形态,将细胞以每孔 2x10⁴ 个细胞的密度接种于 8 孔腔室载玻片中,孵育过夜使其贴壁,然后用 10⁻⁸ M TP 处理 24 小时。如先前文献23所述对载玻片进行固定和透化处理,然后用抗 CXCL9 PE和抗 CXCL5 APC抗体,或用 F-肌动蛋白探针鬼笔环肽 (Alexa Fluor 555 偶联) 于 4°C 孵育过夜进行标记。用 DAPI 复染细胞核,并用 FluoroShield 封片剂封片。使用蔡司 LSM 780 共聚焦显微镜拍摄显微照片。 |
| 动物实验 |
吸收
丙酸睾酮肌内注射后吸收缓慢。这种缓慢吸收是由于其酯基极性较低所致。与庚酸睾酮或环戊丙酸睾酮相比,丙酸睾酮的吸收速度较慢,因此需要更频繁地注射。其吸收参数(AUC)和滞留时间分别为180-210 ng·h/ml和40-60 h。 消除途径 肌内注射的睾酮剂量约90%以葡萄糖醛酸和硫酸结合物的形式经尿液排出,这些结合物是睾酮及其代谢物。剩余剂量中,约 6% 会经粪便排出,主要以非结合型形式排出。 分布容积 丙酸睾酮的登记分布容积范围为 75-120 L/kg。 清除率 与睾酮相比,丙酸睾酮的清除率降低。报告的清除率约为 2000 ml/min。 生物半衰期 与其他睾酮酯相比,丙酸睾酮的半衰期相对较短,约为 4.5 天。 |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
丙酸睾酮肌内注射后吸收缓慢。这种缓慢吸收是由于其酯基极性较低所致。与庚酸睾酮或环戊丙酸睾酮相比,丙酸睾酮的吸收速度较慢,因此需要更频繁地注射。其吸收参数(AUC 和滞留时间)分别为 180-210 ng·h/ml 和 40-60 h。 肌内注射的睾酮剂量约 90% 以葡萄糖醛酸和硫酸结合物的形式经尿液排出,这些结合物是睾酮及其代谢物。剩余剂量中约有 6% 经粪便排出,主要以非结合型形式排出。 丙酸睾酮的分布容积为 75-120 L/kg。 与睾酮相比,丙酸睾酮的清除率降低。报道的清除率约为 2000 ml/min。 代谢/代谢物 与所有睾酮酯一样,丙酸睾酮在血浆中迅速水解为游离睾酮。睾酮通过两条不同的途径代谢为 17-酮类固醇。主要活性代谢物是雌二醇和二氢睾酮 (DHT)。 丙酸睾酮迅速水解为睾酮。睾酮通过两条不同的途径代谢为 17-酮类固醇。主要活性代谢产物是雌二醇和二氢睾酮 (DHT)。 排泄途径:肌注睾酮后,约 90% 的剂量以葡萄糖醛酸和硫酸结合物的形式经尿液排出;约 6% 的剂量经粪便排出,主要以非结合形式排出。 生物半衰期 与其他睾酮酯相比,丙酸睾酮的半衰期相对较短,约为 4.5 天。 |
| 毒性/毒理 (Toxicokinetics/TK) |
毒性概述
睾酮在人类和其他脊椎动物体内的作用主要通过两种机制实现:一是激活雄激素受体(直接激活或以二氢睾酮的形式激活),二是转化为雌二醇并激活某些雌激素受体。游离睾酮 (T) 被转运至靶组织细胞的细胞质中,在那里它可以与雄激素受体结合,或者被细胞质酶 5α-还原酶还原为 5α-二氢睾酮 (DHT)。DHT 与同一雄激素受体的结合力甚至比 T 更强,因此其雄激素效力约为 T 的 2.5 倍。T-受体或 DHT-受体复合物发生结构变化,使其能够进入细胞核并直接与染色体 DNA 的特定核苷酸序列结合。这些结合区域被称为激素反应元件(HRE),它们影响某些基因的转录活性,从而产生雄激素效应。 蛋白质结合 血浆中98%的睾酮与性激素结合球蛋白结合,而2%则保持游离状态或与白蛋白和其他蛋白质结合。 |
| 参考文献 |
[1] Archives of General Psychiatry, 57, 133-140; [2] Personality and Individual Differences, 28, 437-445; [3] Am J Physiol Endocrinol Metab 2003 Jan 7; [4] J Investig Med. 1997 Oct;45(8):441-7; [5] J Clin Endocrinol Metab. 1986 Dec;63(6):1361-4; [6] J Clin Endocrinol Metab. 1997 Feb;82(2):407-13; [7] Am J Physiol Endocrinol Metab. 2002 Mar;282(3):E601-7; [8] Curr Opin Clin Nutr Metab Care. 2004 May;7(3):271-7; [9] Curr Pharm Biotechnol. 2004 Oct;5(5):459-70; [10] Metabolism. 1991 Apr;40(4):368-77; [11]] J Lab Clin Med. 1995 Mar;125(3):326-33; [12] Zhonghua Nan Ke Xue. 2003;9(4):248-51. [13] Balkan Med J. 2023 Mar 8;40(2):117–123. |
| 其他信息 |
丙酸睾酮呈无味白色或淡黄色晶体,或白色或乳白色结晶粉末。(NTP, 1992)
丙酸睾酮是一种类固醇酯。 丙酸睾酮是一种缓释合成代谢类固醇,半衰期短。它是睾酮的合成雄甾烷类固醇衍生物,以17β-丙酸睾酮酯的形式存在。丙酸睾酮最初由Watson实验室研发,并于1974年2月5日获得FDA批准。目前,该药物已停止用于人类,但其兽用产品仍可作为非处方药购买。 丙酸睾酮是一种短效油基注射剂型的睾酮。睾酮抑制垂体分泌促性腺激素,并抑制卵巢产生雌激素,从而降低内源性雌激素水平。此外,该药物可促进男性性特征的维持,适用于性腺功能减退男性的睾酮替代治疗。(NCI04) 睾酮酯,其17β位被丙酸酯取代。 睾酮酯,其17β位被丙酸酯取代。 另见:睾酮(含活性部分);苯甲酸雌二醇;丙酸睾酮(成分)。 药物适应症 丙酸睾酮在兽医实践中用于小母牛,以促进其最大程度的生长。 作用机制 睾酮在人类和其他脊椎动物中的作用主要通过两种机制实现:激活雄激素受体(直接或以二氢睾酮的形式),以及转化为雌二醇并激活某些雌激素受体。游离睾酮 (T) 被转运至靶组织细胞的细胞质中,在那里它可以与雄激素受体结合,或者被细胞质酶 5α-还原酶还原为 5α-二氢睾酮 (DHT)。结合位点称为激素反应元件 (HRE),它们影响某些基因的转录活性,从而产生雄激素效应。 药效学 丙酸睾酮的给药可诱导与男性性发育相关的蛋白质的产生。临床试验表明,给予丙酸睾酮后,血浆促黄体生成素 (LH) 水平会降低。 |
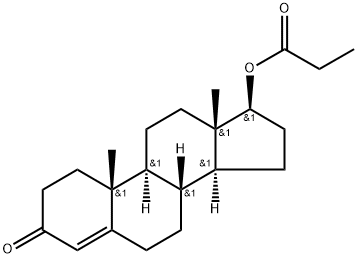
| 分子式 |
C22H32O3
|
|---|---|
| 分子量 |
344.495
|
| 精确质量 |
344.235
|
| CAS号 |
57-85-2
|
| PubChem CID |
5995
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 密度 |
1.1±0.1 g/cm3
|
| 沸点 |
454.6±45.0 °C at 760 mmHg
|
| 熔点 |
118-123 °C
|
| 闪点 |
196.3±28.8 °C
|
| 蒸汽压 |
0.0±1.1 mmHg at 25°C
|
| 折射率 |
1.538
|
| LogP |
4.9
|
| tPSA |
43.37
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
25
|
| 分子复杂度/Complexity |
621
|
| 定义原子立体中心数目 |
6
|
| SMILES |
O(C(C([H])([H])C([H])([H])[H])=O)[C@@]1([H])C([H])([H])C([H])([H])[C@@]2([H])[C@]3([H])C([H])([H])C([H])([H])C4=C([H])C(C([H])([H])C([H])([H])[C@]4(C([H])([H])[H])[C@@]3([H])C([H])([H])C([H])([H])[C@@]21C([H])([H])[H])=O
|
| InChi Key |
PDMMFKSKQVNJMI-BLQWBTBKSA-N
|
| InChi Code |
InChI=1S/C22H32O3/c1-4-20(24)25-19-8-7-17-16-6-5-14-13-15(23)9-11-21(14,2)18(16)10-12-22(17,19)3/h13,16-19H,4-12H2,1-3H3/t16-,17-,18-,19-,21-,22-/m0/s1
|
| 化学名 |
[(8R,9S,10R,13S,14S,17S)-10,13-dimethyl-3-oxo-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl] propanoate
|
| 别名 |
Enarmon Androlon Testosterone propionate
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~30 mg/ml
DMF: ~30 mg/ml Ethanol: ~2 mg/ml |
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9028 mL | 14.5138 mL | 29.0276 mL | |
| 5 mM | 0.5806 mL | 2.9028 mL | 5.8055 mL | |
| 10 mM | 0.2903 mL | 1.4514 mL | 2.9028 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。