| 规格 | 价格 | |
|---|---|---|
| 100mg | ||
| 250mg | ||
| 500mg | ||
| Other Sizes |
| 参考文献 |
1: Vyhlídalová B, Krasulová K, Pečinková P, Poulíková K, Vrzal R, Andrysík Z, Chandran A, Mani S, Dvorak Z. Antimigraine Drug Avitriptan Is a Ligand and Agonist of Human Aryl Hydrocarbon Receptor That Induces CYP1A1 in Hepatic and Intestinal Cells. Int J Mol Sci. 2020 Apr 17;21(8):2799. doi: 10.3390/ijms21082799. PMID: 32316498; PMCID: PMC7216230. 2: Fang W, Bauer F, Dong Y, Breit B. A domino reaction for generating β-aryl aldehydes from alkynes by substrate recognition catalysis. Nat Commun. 2019 Oct 25;10(1):4868. doi: 10.1038/s41467-019-12770-w. PMID: 31653836; PMCID: PMC6814718. 3: Puente J, Jaque M, Carrasco C, Cruz C, Valenzuela M, Wolf M, Mosnaim A. Triptan drugs, natural killer cell cytotoxicity, and neutrophils pro-matrix metalloproteinase-9 secretion. Headache. 2008 Nov-Dec;48(10):1482-9. doi: 10.1111/j.1526-4610.2008.01136.x. Epub 2008 May 9. PMID: 18479418. 4: Schmidt LE, Dalhoff K. Food-drug interactions. Drugs. 2002;62(10):1481-502. doi: 10.2165/00003495-200262100-00005. PMID: 12093316. 5: Bhalla P, Sharma HS, Wurch T, Pauwels PJ, Saxena PR. Molecular cloning and expression of the porcine trigeminal ganglion cDNA encoding a 5-ht(1F) receptor. Eur J Pharmacol. 2002 Feb 1;436(1-2):23-33. doi: 10.1016/s0014-2999(01)01605-3. PMID: 11834243. 6: Maslov LN, Krylatov AV, Lishmanov AIu, Solenkova NV, Bogomaz SA. Tolerantnost' serdtsa krys k aritmogennym vozdeĭstviiam v usloviiakh farmakologicheskoĭ aktivatsii K(ATP)-kanalov [Myocardial tolerance to arrhythmogenic exposure and pharmacological activation of K(ATP)-channels in rats]. Eksp Klin Farmakol. 2001 Mar-Apr;64(2):41-4. Russian. PMID: 11548447. 7: Knight YE, Edvinsson L, Goadsby PJ. Blockade of calcitonin gene-related peptide release after superior sagittal sinus stimulation in cat: a comparison of avitriptan and CP122,288. Neuropeptides. 1999 Feb;33(1):41-6. doi: 10.1054/npep.1999.0009. PMID: 10657470. 8: Roon KI, Maassen Van Den Brink A, Ferrari MD, Saxena PR. Bovine isolated middle cerebral artery contractions to antimigraine drugs. Naunyn Schmiedebergs Arch Pharmacol. 1999 Nov;360(5):591-6. doi: 10.1007/s002109900095. PMID: 10598799. 9: Nilsson T, Longmore J, Shaw D, Olesen IJ, Edvinsson L. Contractile 5-HT1B receptors in human cerebral arteries: pharmacological characterization and localization with immunocytochemistry. Br J Pharmacol. 1999 Nov;128(6):1133-40. doi: 10.1038/sj.bjp.0702773. PMID: 10578124; PMCID: PMC1571736. 10: Sharma A, Jusko WJ, Fulmor IE, Norton J, Uderman HD, Salazar DE. Pharmacokinetics and pharmacodynamics of avitriptan during intravenous administration in healthy subjects. J Clin Pharmacol. 1999 Jul;39(7):685-94. doi: 10.1177/00912709922008326. PMID: 10392323. 11: Jhee SS, Salazar DE, Ford NF, Fulmor IE, Sramek JJ, Cutler NR. A double- blind, randomized, crossover assessment of blood pressure following administration of avitriptan, sumatriptan, or placebo to patients with mild to moderate hypertension. Cephalalgia. 1999 Mar;19(2):95-9. doi: 10.1046/j.1468-2982.1999.019002095.x. PMID: 10214534. 12: Swan L, Hood S, Birnie DH, Muir DF, McCann GP, Hillis WS. The haemodynamic effect of the 5HT1 agonist BMS-180048: a class effect of triptans? Br J Clin Pharmacol. 1999 Feb;47(2):189-94. doi: 10.1046/j.1365-2125.1999.00875.x. PMID: 10190654; PMCID: PMC2014163. 13: Dahlöf CG, Falk L, Risenfors M, Lewis CP. Safety trial with the 5HT1B/1D agonist avitriptan (BMS-180048) in patients with migraine who have experienced pressure, tightness, and/or pain in the chest, neck, and/or throat following sumatriptan. Cephalalgia. 1998 Oct;18(8):546-51. doi: 10.1046/j.1468-2982.1998.1808546.x. PMID: 9827246. 14: Marathe PH, Sandefer EP, Kollia GE, Greene DS, Barbhaiya RH, Lipper RA, Page RC, Doll WJ, Ryo UY, Digenis GA. In vivo evaluation of the absorption and gastrointestinal transit of avitriptan in fed and fasted subjects using gamma scintigraphy. J Pharmacokinet Biopharm. 1998 Feb;26(1):1-20. doi: 10.1023/a:1023236823320. PMID: 9773390. 15: Marathe PH, Greene DS, Kollia GD, Barbhaiya RH. Evaluation of the effect of food on the pharmacokinetics of avitriptan. Biopharm Drug Dispos. 1998 Sep;19(6):381-94. doi: 10.1002/(sici)1099-081x(199809)19:6<381::aid- bdd118>3.0.co;2-j. PMID: 9737819. 16: MaassenVanDenBrink A, Reekers M, Bax WA, Ferrari MD, Saxena PR. Coronary side-effect potential of current and prospective antimigraine drugs. Circulation. 1998 Jul 7;98(1):25-30. doi: 10.1161/01.cir.98.1.25. PMID: 9665056. 17: Cutler NR, Salazar DE, Jhee SS, Fulmor IE, Ford N, Smith RA, Sramek JJ. Pharmacokinetics and pharmacodynamics of avitriptan in patients with migraine after oral dosing. Headache. 1998 Jun;38(6):446-52. doi: 10.1046/j.1526-4610.1998.3806446.x. PMID: 9664749. 18: Marathe PH, Greene DS, Lee JS, Barbhaiya RH. Assessment of effect of food, gender, and intra-subject variability in the pharmacokinetics of avitriptan. Biopharm Drug Dispos. 1998 Apr;19(3):153-7. doi: 10.1002/(sici)1099-081x(199804)19:3<153::aid-bdd90>3.0.co;2-t. PMID: 9569997. 19: Marathe PH, Greene DS, Kollia GD, Barbhaiya RH. A pharmacokinetic interaction study of avitriptan and propranolol. Clin Pharmacol Ther. 1998 Mar;63(3):367-78. doi: 10.1016/S0009-9236(98)90168-0. PMID: 9542480. 20: Marathe PH, Greene DS, Kollia GD, Barbhaiya RH. The effects of age and gender on the single dose pharmacokinetics of avitriptan administered to healthy volunteers. J Clin Pharmacol. 1997 Oct;37(10):937-45. doi: 10.1002/j.1552-4604.1997.tb04268.x. PMID: 9505985.
|
|---|
| 分子式 |
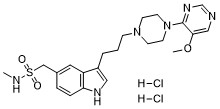
C22H32CL2N6O3S
|
|---|---|
| 分子量 |
531.497
|
| 精确质量 |
458.21
|
| 元素分析 |
C, 49.72; H, 6.07; Cl, 13.34; N, 15.81; O, 9.03; S, 6.03
|
| CAS号 |
170956-82-8
|
| 相关CAS号 |
191588-96-2 (HCl);170956-82-8 (2HCl);151140-97-5 (3HCl);151140-96-4; 171171-42-9 (fumarate); 171171-42-9;
|
| SMILES |
O=S(CC1=CC2=C(NC=C2CCCN3CCN(C4=NC=NC=C4OC)CC3)C=C1)(NC)=O.[H]Cl.[H]Cl
|
| InChi Key |
LXDKLZKTSZKRLF-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C22H30N6O3S.2ClH/c1-23-32(29,30)15-17-5-6-20-19(12-17)18(13-25-20)4-3-7-27-8-10-28(11-9-27)22-21(31-2)14-24-16-26-22;;/h5-6,12-14,16,23,25H,3-4,7-11,15H2,1-2H3;2*1H
|
| 化学名 |
1H-Indole-5-methanesulfonamide, 3-(3-(4-(5-methoxy-4-pyrimidinyl)-1-piperazinyl)propyl)-N-methyl-, dihydrochloride
|
| 别名 |
Avitriptan HCl; Avitriptan hydrochloride; Avitriptan dihydrochloride; Avitriptan; BMS 180048; BMS-180048; BMS180048;
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体内) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8815 mL | 9.4073 mL | 18.8147 mL | |
| 5 mM | 0.3763 mL | 1.8815 mL | 3.7629 mL | |
| 10 mM | 0.1881 mL | 0.9407 mL | 1.8815 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。