| 规格 | 价格 | |
|---|---|---|
| 5mg | ||
| 10mg | ||
| 25mg | ||
| 50mg | ||
| 100mg | ||
| Other Sizes |
| 靶点 |
DA1 receptor ( EC50 = 55.5 nM )
|
|---|---|
| 药代性质 (ADME/PK) |
吸收、分布和排泄
放射性标记研究表明,输注的芬诺多泮约90%经尿液排出,10%经粪便排出。其主要通过结合反应排出,不涉及细胞色素P-450酶。仅有4%的给药剂量以原形排出。 代谢/代谢物 其主要通过结合反应排出,不涉及细胞色素P-450酶。甲基化、葡萄糖醛酸化和硫酸化是主要的结合途径。 生物半衰期 在轻度至中度高血压患者中,其消除半衰期约为5分钟,R(活性)异构体和S异构体之间的差异很小。 |
| 毒性/毒理 (Toxicokinetics/TK) |
妊娠期和哺乳期影响
◉ 哺乳期用药概述 目前尚无关于芬诺多泮在哺乳期使用的信息。生产商建议在服用芬诺多泮期间避免哺乳;然而,由于其口服生物利用度低且半衰期短,乳汁中的芬诺多泮不太可能对母乳喂养的婴儿产生不良影响。此外,芬诺多泮可以静脉注射给婴儿。与多巴胺不同,它不会降低血清催乳素浓度,因此可能不会干扰哺乳。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对哺乳和母乳的影响 截至修订日期,未找到关于哺乳期母亲的相关已发表信息。与多巴胺不同,芬诺多泮输注不会影响正常女性的血清催乳素浓度。对于已经建立泌乳的母亲来说,催乳素水平可能不会影响其母乳喂养能力。 |
| 参考文献 | |
| 其他信息 |
芬诺多泮是一种苯并氮杂卓类药物。它具有多巴胺能拮抗剂、血管扩张剂、α-肾上腺素能激动剂、多巴胺激动剂和抗高血压药的双重作用。
芬诺多泮是一种多巴胺D1受体激动剂,用作抗高血压药。它通过扩张小动脉血管来降低血压。 芬诺多泮是一种多巴胺能激动剂。芬诺多泮的作用机制是作为多巴胺激动剂。 芬诺多泮是一种具有血管扩张和抗高血压特性的苯并氮杂卓衍生物。芬诺多泮是一种多巴胺(DA)受体激动剂,能特异性地与外周DA1受体结合,并以中等亲和力与α2肾上腺素能受体结合。然而,该药物对DA2受体、其他α肾上腺素能受体、β肾上腺素能受体、毒蕈碱受体或5-羟色胺能受体均无显著亲和力。受体结合调节离子跨膜转运,从而刺激腺苷酸环化酶活性以及催乳素释放。这导致血管舒张,增加肾血流量,进而增强钠排泄和利尿,最终降低舒张压。 一种用作抗高血压药物的多巴胺D1受体激动剂。它通过扩张小动脉来降低血压。 另见:甲磺酸非诺多泮(有盐形式)。 药物适应症 用于住院期间短期(最多 48 小时)治疗严重高血压,尤其适用于临床需要快速但可逆的紧急降压的情况,包括伴有终末器官功能恶化的恶性高血压。 FDA 标签 作用机制 非诺多泮是一种速效血管扩张剂。它是 D1 样多巴胺受体的激动剂,并以中等亲和力与 α2 肾上腺素受体结合。它对D2样受体、α1和β肾上腺素能受体、5HT1和5HT2受体或毒蕈碱受体均无显著亲和力。芬诺多泮是外消旋混合物,其中R-异构体具有生物活性。R-异构体对D1样受体的亲和力比S-异构体高约250倍。在非临床研究中,芬诺多泮对突触前D2样多巴胺受体、α或β肾上腺素能受体均无激动作用,也不影响血管紧张素转换酶活性。芬诺多泮可能升高血浆去甲肾上腺素浓度。 |
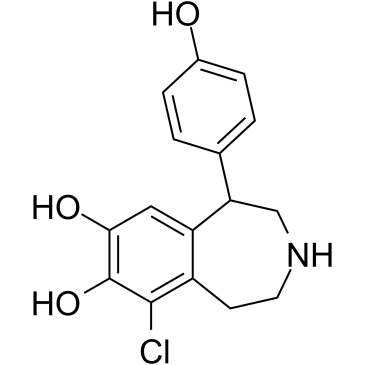
| 分子式 |
C16H16CLNO3
|
|---|---|
| 分子量 |
305.75614
|
| 精确质量 |
305.081
|
| CAS号 |
67227-56-9
|
| 相关CAS号 |
Fenoldopam mesylate; 67227-57-0; Fenoldopam hydrochloride; 181217-39-0
|
| PubChem CID |
3341
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 密度 |
1.4±0.1 g/cm3
|
| 沸点 |
522.6±50.0 °C at 760 mmHg
|
| 闪点 |
269.9±30.1 °C
|
| 蒸汽压 |
0.0±1.4 mmHg at 25°C
|
| 折射率 |
1.656
|
| LogP |
1.72
|
| tPSA |
72.72
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
1
|
| 重原子数目 |
21
|
| 分子复杂度/Complexity |
348
|
| 定义原子立体中心数目 |
0
|
| SMILES |
OC1=C(O)C=C2C(C3=CC=C(O)C=C3)CNCCC2=C1Cl
|
| InChi Key |
TVURRHSHRRELCG-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H16ClNO3/c17-15-11-5-6-18-8-13(9-1-3-10(19)4-2-9)12(11)7-14(20)16(15)21/h1-4,7,13,18-21H,5-6,8H2
|
| 化学名 |
9-chloro-5-(4-hydroxyphenyl)-2,3,4,5-tetrahydro-1H-3-benzazepine-7,8-diol
|
| 别名 |
SKF 82526; Fenoldopam
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~77 mg/mL (~251.8 mM)
Water: ~10 mg/mL |
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2705 mL | 16.3527 mL | 32.7054 mL | |
| 5 mM | 0.6541 mL | 3.2705 mL | 6.5411 mL | |
| 10 mM | 0.3271 mL | 1.6353 mL | 3.2705 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00621790 | Completed | Drug: fenoldopam Drug: placebo |
Acute Renal Failure | Università Vita-Salute San Raffaele |
February 2008 | Phase 3 |
| NCT00982527 | Completed | Drug: Placebo Drug: Fenoldopam |
Kidney Failure, Acute | Bambino Gesù Hospital and Research Institute |
September 2009 | Phase 3 |
| NCT00122018 | Completed | Drug: N-acetylcysteine Drug: fenoldopam |
Kidney Failure, Acute Kidney Failure, Chronic |
Linda F. Barr, M.D. | May 2002 | Phase 2 |
| NCT00747331 | Completed | Drug: Fenoldopam mesilate Drug: Placebo |
Cardiac Complications Cardiopulmonary Bypass |
IRCCS Policlinico S. Donato | September 2008 | Phase 4 |
| September 2008 | Completed | Drug: Fenoldopam | Salt-sensitive Hypertension | Georgetown University | November 2002 | Not Applicable |