| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Three male Alpk:APfSD (Wistar-derived) rats per group received (methoxyphenyl-U-(14)C)NOA446510/mandipropamide/ at a dose of 3 mg/kg/day for 14 days. The three male rats were sacrificed 24 hours after administration for 3, 7, 10, and 14 consecutive days, and again 1, 4, 7, 14, 21, 28, and 49 days after the last consecutive administration. Radioactive analysis (LSC) was performed on samples from the adrenal glands, brain, heart, kidneys, liver, lungs, pancreas, spleen, thymus, thyroid gland, testes, as well as blood, plasma, bone (femur), fat (abdomen), and muscle. Urine, feces, and cage cleaning fluid were collected from the three male animals 24 hours after the first administration and 24 hours after the 14th consecutive administration, and subjected to LSC. Tissue radioactivity concentrations reached steady state within 4 days after the first dose, then declined rapidly after discontinuation of dosing. After 14 consecutive days of dosing, the highest mean tissue C concentrations were observed in the liver (0.727 μg Equivalent/g) and kidneys (0.234 μg Equivalent/g). Elimination in the liver was biphasic, with a terminal elimination half-life of 4.2 days and a distribution half-life of 2.3 days. The elimination half-life in the kidneys was 8.7 days. The elimination half-life in bone was 12.8 days. Concentrations in other tissues at steady state were generally low (0.1 μg Equivalent/g or lower), and radioactivity decreased below the detection limit within several days after the last dose. Most radioactive material was excreted in feces (79% of the dose after a single dose and 66% after 14 consecutive doses). The urinary radioactivity level was 2.6% after a single dose and 7% after 14 consecutive doses. One or four Alpk:APfSD (Wistar-derived) rats of each sex in each group received a single oral administration of 3 mg/kg and 300 mg/kg of (chlorophenyl-U-(14)C)NOA446510/mandipropamid/ or (methoxyphenyl-U-(14)C)NOA446510/mandipropamid/, respectively. Following administration, exhaled carbon dioxide was collected from one animal of each sex in each group (groups 2, 6, and 7) and collected in a trap containing 2N sodium hydroxide solution; exhaled metabolites were collected at 8 or 10 hours (female), 24, 36, and 48 hours and collected in an activated charcoal trap. At all time points, the residual levels of the radiolabeled (chlorophenyl-U-(14)C)NOA446510 and (methoxyphenyl-U-(14)C)NOA446510 in exhaled carbon dioxide and volatile metabolites were close to or below the detection limit. 48 hours after administration of a 3 mg/kg dose, the total radioactivity in the exhaled breath of both male and female subjects with (chlorophenyl-U-(14)C)NOA446510 was below 0.1% of the dose, while the total radioactivity in the exhaled breath of both male and female subjects with (methoxyphenyl-U-(14)C)NOA446510 was below 0.2% of the dose. No radioactive material (metabolites) was detected in the activated charcoal trap. 48 hours after administration of a 300 mg/kg dose of (methoxyphenyl-U-(14)C)NOA446510, the radioactivity in the exhaled breath of both male and female subjects was below 0.2% of the dose, and no radioactive material was detected in the activated charcoal trap. Following administration to animals in excretion and tissue distribution phases (groups 3, 4, 8, and 9, 4 male and 4 female rats per group), urine was collected at 6 and 10 hours post-administration, and urine and feces were collected at 24, 48, 72, 96, 120, 144, and 168 hours post-administration. Within 168 hours, the mean percentage of total radioactivity excreted in urine and feces for the radiolabeled (chlorophenyl-U-(14)C)NOA446510 and (methoxyphenyl-U-(14)C)NOA446510 was similar. At both dose levels, the primary route of excretion was feces, but the proportion of the administered dose excreted in urine was higher in the low-dose group (300 mg/kg) and in the female (male) groups. At a dose of 300 mg/kg, male animals accounted for an average of 87%-91% of total fecal excretion, while female animals accounted for 81%-84%; total urinary excretion accounted for an average of 2.2%-3.3%, while female animals accounted for 6.4%-11.6%. At a dose of 3 mg/kg, male animals accounted for an average of 76%-81% of total fecal excretion, while female animals accounted for 42%-55%; total urinary excretion accounted for an average of 14%-15%, while female animals accounted for 29%-47%. Seven days after treatment, the tissue distribution of the two radiolabeled substances was similar in both sexes. At a dose of 3 mg/kg, the radioactivity in tissues accounted for 0.06%–0.17% of the total dose, and the radioactivity in the carcass accounted for 0.08%–0.19% of the total dose. The tissues with the highest radioactivity concentrations were the liver (0.033–0.085 μg equivalents/g, accounting for 0.05%–0.16% of the total dose), kidneys (0.007–0.019 μg equivalents/g, <0.01%), and thyroid gland (<0.010–0.030 μg equivalents/g, <0.01%). The radioactivity concentrations in all other tissues were below 0.010 μg equivalents/g. At a dose of 300 mg/kg/day, the radioactivity in tissues accounted for 0.02%–0.04% of the total dose, and the radioactivity in the carcass accounted for 0.01%–0.11% of the total dose. The tissues with the highest radioactivity concentrations were the liver (0.81–1.57 μg Equivalents/g (0.01%–0.03% of the dose)), kidney (0.12–0.35 μg Equivalents/g (<0.01% of the dose)), and thyroid gland (<0.17–1.20 μg Equivalents/g (<0.01% of the dose)). The radioactivity concentrations in whole blood and plasma were <0.08–0.24 μg Equivalents/g and <0.01–0.04 μg Equivalents/g, respectively. The radioactivity concentrations in all other tissues were below 0.10 μg Equivalents/g. In the bile duct cannulated rat phase (groups 10 and 11, 4 male and 4 female rats per group), urine was collected at 10 hours (female) or 12, 24, and 48 hours post-administration, feces were collected at 24 and 48 hours, and bile was collected at 1, 2, 3, 4, 5, 6, 7, 8, and 10 hours (female) or 12, 24, 36, and 48 hours post-administration. In bile duct cannulated rats receiving 3 mg/kg (methoxyphenyl-U-(14)C) NOA446510, the majority of the administered dose was excreted via bile after 48 hours (73% in males and 55% in females); fecal excretion was 14% in males and 22% in females. The urinary excretion rate was 1.4% in male rats and 9.5% in female rats. At a dose of 300 mg/kg, the bile excretion rate was 28% in male rats and 22% in female rats; the drug content in feces was 39% (male) and 26% (female), respectively; after 48 hours, the drug content in urine was 0.8% (male) and 19% (female), respectively. Nine or 15 Alpk:APfSD (Wistar-derived) rats of each sex were given a single oral gavage dose of 3 and 300 mg/kg of (Methoxyphenyl-U-(14)C)NOA446510/mandipropamid/. In the first phase, serial blood samples were collected from the tail vein of each group (3 males and 3 females) at 2, 4.5, 6.5, 8-8.5, 9.5-10, 13, 18, 24, 34, 48, 72 and 96 hours after administration. The administration doses were 3 mg/kg (Group 1) and 300 mg/kg (Group 2). Based on the results of the first phase, the time for animal sacrifice in the second phase was determined. In the second phase, rats in each group (n=3 males and 3 females) were sacrificed at 8, 24, 48, 72, and 96 hours post-administration, at doses of 3 mg/kg (Group 3) and 300 mg/kg (Group 4), respectively. At each time point, radiometric analysis (LSC) was performed on blood samples (separated into plasma and blood), adrenal glands, brain, heart, kidneys, liver, lungs, ovaries, pancreas, spleen, thymus, thyroid gland, testes, uterus, bones (femur), fat (abdomen), and muscle from each animal. In the first phase, following administration of the 3 mg/kg dose, the mean time to peak plasma concentration (Tmax) was 8.5 hours for males and 4.5 hours for females; the elimination half-life (t1/2) was 18.4 hours (males) and 20.2 hours (females), respectively. Following administration of a 300 mg/kg dose, the mean Tmax was 24 hours for males and 10 hours for females; the t1/2 was 32.7 hours for males and 24.8 hours for females. In the second phase, tissue radioactivity concentrations were slightly lower in females than in males at both dose levels. Residual radioactivity concentrations in liver and kidney tissues were the only measurements consistently higher than plasma concentrations (0.13 μg EQ/g for males and 0.10 μg EQ/g for females at a 3 mg/kg dose; 5.12 μg EQ/g for males and 2.65 μg EQ/g for females at a 300 mg/kg dose). All tissue concentrations decreased to low values or below the detection limit within 96 hours post-treatment. The liver had the highest average radioactive concentration among all groups. At a dose of 3 mg/kg, after 8 hours, the concentrations were: 1.25 μg equivalent/g (2.04% of the dose) for men and 0.64 μg equivalent/g (0.94% of the dose) for women; at 300 mg/kg, the concentrations were: 46.4 μg equivalent/g (0.77% of the dose) for men and 27.1 μg equivalent/g (0.39% of the dose) for women. Eight hours after administration, at a dose of 3 mg/kg, the drug concentration in the kidneys was 0.26 μg equivalent/g (0.08% of the dose) for men and 0.25 μg equivalent/g (0.08% of the dose) for women; at a dose of 300 mg/kg, the drug concentration in the kidneys was 10.43 μg equivalent/g (0.04% of the dose) for men and 6.9 μg equivalent/g (0.02% of the dose) for women. At a dose of 3 mg/kg, the mean half-life of liver in men was 23.6 hours and in women 23.1 hours; the mean half-life of kidney in men was 22.2 hours and in women 21.5 hours. At a dose of 300 mg/kg, the mean half-life of liver was 19.1 hours in men and 17.7 hours in women; the mean half-life of kidney was 19.5 hours in men and 15.6 hours in women. The mean half-life of other tissues was generally between 18 and 24 hours (except for the thymus (31.5 hours) and testes (30.0 hours) in men at a dose of 300 mg/kg). In one gavage study, 27 C57BL/10Jf/Alpk mice of each sex were administered a single gavage dose of 10, 50, and 500 mg/kg of (Methoxyphenyl-U-(14)C)NOA446510/mandipropamid/. In a preliminary dietary study, 55 C57BL/10Jf/Alpk mice (half male and half female) were fed a diet containing 800 ppm (Methoxyphenyl-U-(14)C)NOA446510 for two consecutive days. In the main dietary study, 28 C57BL/10Jf/Alpk mice (half male and half female) were fed a diet containing 300, 800, 2000, and 5000 ppm (Methoxyphenyl-U-(14)C)NOA446510 for seven consecutive days. In the intravenous injection study, five C57BL/10Jf/Alpk mice (half male and half female) in each group received a single intravenous injection of 10 mg/kg (Methoxyphenyl-U-(14)C) NOA446510. In the single-dose gavage administration study, most of the radioactive material was excreted within 24 hours after administration, with no sex difference in excretion route or rate. At doses of 10 mg/kg and 50 mg/kg, male animals excreted 29% of the dose in urine and 32% (10 mg/kg) and 27% (50 mg/kg) of the dose in feces within 48 hours after administration, respectively. Female animals excreted 29% and 47% of the dose in urine and 21% and 24% of the dose in feces within the same time period, respectively. At a dose of 500 mg/kg, feces were the main route of excretion, with 54% and 40% of the dose excreted in feces in males and females, respectively. Radioactive material in urine accounted for 16% of the dose in males and 11% in females. No sex or dose-level differences in plasma pharmacokinetics were observed. At each time point and dose level, the radioactivity concentration measured in whole blood was approximately half that measured in plasma. At doses of 10 and 50 mg/kg, absorption was rapid, reaching maximum plasma concentrations (Cmax) within 30 minutes of administration (2 μg/mL for males and 3 μg/mL for females at 10 mg/kg; 11 μg/mL for males and 16 μg/mL for females at 50 mg/kg). The area under the plasma concentration-time curve (AUC, in h·μg equivalents/mL) at the 50 mg/kg dose was approximately five times that at the 10 mg/kg dose (88 vs. 17 for males and 76 vs. 17 for females), indicating a linear increase in absorption. At a dose of 500 mg/kg, the absorption phase was slow, with plasma Cmax of 40 μg/mL (male) and 39 μg/mL (female), reached at 6 hours (male) and 2 hours (female), respectively. Furthermore, at the 500 mg/kg dose, the AUC values for males and females were 6 times (543 h·μg equivalent/mL) and 7 times (535 h·μg equivalent/mL) higher than the 50 mg/kg dose group, respectively, while the expected values were 10 times higher than the 50 mg/kg dose group. Five major metabolites were detected in urine, accounting for more than 90% of the total radioactivity in all samples. At all three dose levels, metabolite 4 (NOA458422 glucuronide, i.e., O-glucuronide of NOA458422 (2-(4-chlorophenyl)-N-(2-(4-hydroxy-3-methoxyphenyl)ethyl)-2-prop-2-alkynyloxyacetamide)) was the major component, and metabolite 2 (illustrated only) was the second component. Unmetabolized NOA446510 was not detected in urine. At a dose of 10 mg/kg, the metabolic profile of fecal samples from 0–24 hours was similar to that of urine, with trace amounts of NOA446510 detected in males but not in females. At doses of 50 and 500 mg/kg, unmetabolized NOA446510 was the major component in feces of both sexes. In plasma, metabolite 4 was the major component in both sexes at all dose levels; metabolite 2 was present in very low amounts at a dose of 10 mg/kg. At a dose of 50 mg/kg, NOA446510 was detected; at a dose of 500 mg/kg, trace amounts of unmetabolized NOA446510 were detected. In a 2-day 800 ppm dietary study, peak whole blood concentrations (Cmax) were reached at 38.5 hours (males) and 41.0 hours (females) after administration. In a 7-day dietary study, similar to the gavage study, radioactivity correlated with plasma composition, with no sex differences, and radioactivity was proportional to dose level (although specific plasma radioactivity concentration data are limited due to a lack of actual dose calculations). Urinary metabolite profiles were qualitatively and quantitatively similar to those of the gavage animals and were independent of sex, dose level, and frequency of administration. Metabolite 4 was the predominant urinary component, and unmetabolized NOA446510 was not detected in any samples. Unmetabolized NOA446510 was detected in all fecal samples. Metabolites 2 and 4 were also present. Unmetabolized NOA446510 was not detected in any plasma samples. In a single intravenous injection study (10 mg/kg), most of the radiolabeled substance was also excreted within 24 hours after administration. Within 24 hours, 58% (male) and 72% (female) of the dose were present in urine, and 25% (male) and 40% (female) of the dose were present in feces, respectively. The metabolite profiles observed in urine samples collected 0–24 hours and 24–48 hours after treatment were identical to those observed after gavage administration, with no sex differences. Loss of propynyl groups and glucuronidation are presumed to be the major biotransformation pathways. For more complete data on absorption, distribution, and excretion of mandipromimethasone (8 types), please visit the HSDB record page. Metabolism/Metabolites Mixed sample analysis was performed on urine and feces from 4 rats of each sex in each group (groups 3, 8, and 9 listed in the table below), which were administered 300 mg/kg of [chlorophenyl-U-14C]NOA446510 and 300 mg/kg of [methoxyphenyl-U-14C]NOA446510 by single gavage, respectively. Additionally, urine, feces, and bile were analyzed from 4 cannulated rats of each sex in each group (groups 10 and 11 listed in the table below), which were also administered 3 mg/kg and 300 mg/kg of [methoxyphenyl-U-14C]NOA446510 by single gavage. In report number CTL/UR0786/REG/REPT (record number 230447), metabolite/component analysis was performed on mixed urine and fecal samples from three male rats 24 hours after administration. [methoxyphenyl-U-14C]NOA446510 was administered by gavage at a single dose of 3 mg/kg, followed by a second gavage 24 hours after 14 consecutive doses of 3 mg/kg. All major metabolites/components comprising more than 5% of the administered dose were identified (analyzed by high-performance liquid chromatography (HPLC) and mass spectrometry (MS)). The hypothesized metabolic pathway is described: loss of one or two propargyl groups, followed by glucuronidation and O-demethylation (illustrated diagram attached). No differences in the metabolism of the two radiolabeled substances were noted. At high doses (300 mg/kg), the metabolic profiles of excrement following administration of the chlorophenyl and methoxyphenyl radiolabeled substances were similar both qualitatively and quantitatively. Following a single administration of 3 mg/kg [methoxyphenyl-U-14C] NOA446510, female animals excreted 46% of the dose in urine and 41% in feces within 96 hours. The major urinary metabolite (40% of the dose) within 96 hours was NOA458422 glucuronide (O-glucuronide of 2-(4-chlorophenyl)-N-[2-(4-hydroxy-3-methoxyphenyl)-ethyl]-2-prop-2-alkynyloxyacetamide). In feces, NOA446510 accounted for 12% of the dose, and NOA458422 accounted for 19%. In male subjects, 14% of the dose was excreted in urine and 72.3% in feces within 96 hours. The major urinary metabolite (accounting for 10% of the dose) was SYN534133 (CGA 380775 (2-4-(chlorophenyl)-2-hydroxy-N-[2-(4-hydroxy-3-methoxyphenyl)-ethyl]acetamide) glucuronide). In feces, NOA458422, the parent compound, and NOA458422 glucuronide accounted for 29.2%, 21.3%, and 12.9% of the total dose, respectively. In male animals that received 3 mg/kg [methoxyphenyl-U-14C] NOA446510 daily for 14 consecutive days, the excretory composition of samples collected 24 hours after the first dose and after 14 consecutive days of administration was similar, consistent with the results of the single-dose study described above. In female animals that received a single dose of 300 mg/kg [chlorophenyl-U-14C] NOA446510, 6% of the dose was excreted in urine and 81% in feces within 96 hours. NOA458422 glucuronide is the main urinary metabolite (3.7% of the dose). NOA446510 accounts for 75% of the dose in female feces. Males excrete 2% and 86% of the dose in urine and feces, respectively, within 96 hours. SYN534133 is the main urinary metabolite, accounting for 1% of the dose. NOA446510 accounts for 79% of the dose in feces. After a single dose of 300 mg/kg [methoxyphenyl-U-14C] NOA446510, females excrete 11% and 82% of the dose in urine and feces, respectively, within 96 hours. NOA458422 glucuronide (7% of the dose) and NOA446510 (71% of the dose) are the main components in urine and feces, respectively. Within 96 hours, male rats excreted 3% of the total dose in urine and 90% in feces. SYN534133 (2% of the total dose) and NOA446510 (73% of the total dose) were the main components in urine and feces, respectively. Following a single administration of 3 mg/kg [methoxyphenyl-U-14C] NOA446510 to cannulated rats, within 48 hours, female rats excreted 15% of the total dose in urine, 46% in bile, and 22% in feces. The major components in urine and bile were NOA458422 glucuronide (10% and 41% of the total dose, respectively). NOA446510 was the major component in feces (22.3% of the total dose). In male rats, within 48 hours, male rats excreted 1%, 73%, and 15% of the total dose in urine, bile, and feces, respectively. NOA458422 glucuronide was the major component in urine (0.7% of the dose) and bile (62.2% of the dose). NOA446510 accounted for 13% of the fecal dose. Following a single injection of 300 mg/kg [methoxyphenyl-U-14C] NOA446510 into bile duct-cannulated rats, female rats excreted 28%, 12%, and 37% of the administered radioactivity in urine, bile, and feces, respectively, within 48 hours. NOA458422 glucuronide was the major component in urine (25% of the dose) and bile (10% of the dose). NOA446510 accounted for 37% of the fecal dose. Male rats excreted 1%, 28%, and 39% of the dose in urine, bile, and feces, respectively, within 48 hours. NOA458422 glucuronide was the major component in urine (0.5% of the dose) and bile (22.5% of the dose). The content of NOA446510 in the feces of male mice was 38.6% of the dose. In one gavage study, 27 C57BL/10Jf/Alpk mice (half male and half female) were administered 10, 50, and 500 mg/kg of (Methoxyphenyl-U-(14)C)NOA446510/mandipropamid/ via single gavage. In a preliminary dietary study, 55 C57BL/10Jf/Alpk mice (half male and half female) were given 800 ppm of (Methoxyphenyl-U-(14)C)NOA446510 in their diet for two consecutive days. In the primary dietary study, 28 C57BL/10Jf/Alpk mice (half male and half female) were given 300, 800, 2000, and 5000 ppm of (Methoxyphenyl-U-(14)C)NOA446510 in their diet for seven consecutive days. In the intravenous injection study, five C57BL/10Jf/Alpk mice (half male and half female) were administered a single intravenous injection of 10 mg/kg of (methoxyphenyl-U-(14)C)NOA446510. …In the urine, five major metabolites were detected, accounting for over 90% of the total radioactivity in all samples. At all three dose levels, metabolite 4 (NOA458422 glucuronide, i.e., O-glucuronide of NOA458422 (2-(4-chlorophenyl)-N-(2-(4-hydroxy-3-methoxyphenyl)ethyl)-2-prop-2-alkynyloxyacetamide)) was the major component, and metabolite 2 (illustrated only) was the second component. Unmetabolized NOA446510 was not detected in the urine. At a dose of 10 mg/kg, the metabolic profile of fecal samples from 0–24 hours was similar to that of urine. A small amount of NOA446510 was detected in males but not in females. At doses of 50 and 500 mg/kg, unmetabolized NOA446510 was the dominant component in the feces of both sexes. In plasma, metabolite 4 was the dominant component in both sexes at all dose levels; metabolite 2 was present in very low amounts at a dose of 10 mg/kg. Concentrations of NOA446510 were detected at a dose of 50 mg/kg; a small amount of unmetabolized NOA446510 was detected at a dose of 500 mg/kg. In a 2-day dietary study at 800 ppm, peak whole blood concentrations (Cmax) were reached at 38.5 hours (males) and 41.0 hours (females) after the start of administration. In the 7-day dietary study, similar to the gavage study, radioactivity correlated with plasma composition, with no sex differences, and radioactivity was proportional to dose level (although specific plasma radioactivity concentration data are limited due to a lack of actual dose calculations). Urinary metabolite profiles were qualitatively and quantitatively similar to those of the gavage animals and were independent of sex, dose level, and frequency of administration. Metabolite 4 was the predominant urinary component, and unmetabolized NOA446510 was not detected in any samples. Unmetabolized NOA446510 was detected in all fecal samples. Metabolites 2 and 4 were also present. Unmetabolized NOA446510 was not detected in any plasma samples. In the single intravenous injection study (10 mg/kg), most of the radiolabeled radioactive material was also excreted within 24 hours of administration. Within 24 hours, urine contained 58% (males) and 72% (females) of the dose, respectively, and feces contained 25% (males) and 40% (females), respectively. The urinary metabolite profiles collected 0–24 hours and 24–48 hours after administration were identical to those observed after gavage administration, with no sex differences. Loss of propynyl groups and glucuronidation are presumed to be the major biotransformation pathways. In the Phase 1 clinical trial, three Alpk:APfSD (Wistar-derived) rats of each sex in each group (groups 1, 2, and 3) received unlabeled NOA446510 (/mandipropamid/). In the first phase, the rats were fed radiolabeled diets at concentrations of 100, 500, and 5000 ppm for 11 consecutive days, followed by the corresponding concentrations of radiolabeled diets on day 12. In addition, in the first phase, three male and three female rats in each group (groups 4, 5, and 6) were administered unlabeled NOA446510 by gavage for 11 consecutive days at doses of 10, 50, and 500 mg/kg/day, respectively, followed by a single gavage administration of the same dose of (methoxyphenyl-U-(14)C)NOA446510 on day 12. In the second phase, six male and six female rats in each group (groups 7, 8, and 9) were fed unlabeled NOA446510 for 11 consecutive days, followed by a diet containing (14)C NOA446510 on day 12. ... Metabolites were identified by high-performance liquid chromatography-mass spectrometry (HPLC-MS). Metabolites in urine and plasma were analyzed. The urinary metabolite profiles differ between men and women, primarily because the major metabolite in women's urine, NOA458422 glucuronide (formed by glucuronidation after the loss of one propargyl group from NOA446510), is present in extremely low amounts (or even absent) in male samples (this may indicate differences in clearance pathways and mechanisms). The proportions of all other urinary metabolites are similar in both men and women. The plasma metabolite profiles are identical in both men and women. Most metabolites are glucuronides. For proposed structures, schematic diagrams, descriptions, mass spectra, HPLC chromatograms, etc., of the metabolites/components, please refer to the study. The concentration of SYN 534133 (glucuronide formed by the loss of two propargyl groups from NOA446510) is similar in both male and female urine. The major metabolite in male samples is NOA458422 glucuronide. NOA458422 glucuronide is the major metabolite in both male and female plasma. In the second phase, the area under the plasma concentration-time exposure curve (AUC0-48(ug·hg-1)) increased linearly with dietary dose levels over 48 hours, with similar values in both males and females (9.5, 31.9, and 344.0 ug·hg-1 at 100, 500, and 5000 ppm doses, respectively, and 8.0, 34.9, and 328.9 ug·hg-1 for females). In the first phase, one Aldley Park Beagle of each sex in each group was given a single oral dose of 100 and 800 mg/kg of (chlorophenyl-U-(14)C)NOA446510/mandipropamid/ on days 1 and 15, respectively. Unlabeled NOA446510 was also administered at doses of 100 and 800 mg/kg/day on days 5 through 14. In the second phase, one dog of each sex in each group received a single intravenous injection of 3 mg/kg of (14)C NOA446510 on day 1, followed by a single oral administration of 3 mg/kg of radiolabeled NOA446510 on day 15. After each administration of radiolabeled NOA446510, the animals were placed in metabolic cages, and urine and feces (at room temperature) were collected at 6, 12, 24, 36, 48, and 72 hours post-administration. During the first phase, blood samples (5 mL) were collected at 1, 2, 3, 4, 6, 8, 12, 24, 36, 48, and 72 hours post-administration, and at 1, 2, 3, 4, 5, 6, 7, and 8 hours post-administration. On day 15, blood samples were collected at 10, 12, and 24 hours post-administration. In Phase 2, blood samples were taken on Day 1 at 2, 5, 10, 20, and 30 minutes and 1, 2, 3, 4, 6, 12, and 24 hours after administration; on Day 15, blood samples were taken at 1, 2, 3, 4, 5, 6, 7, 8, 10, 12, and 24 hours after administration. Phase 1: In Phase 1, following a single oral administration of 100 mg/kg of the radiolabeled drug (gelatin capsules), 7% of the dose was excreted in urine in males and 22% in females within 72 hours. Feces contained 95% (males) and 63% (females) of the dose, respectively. The maximum plasma concentration (Cmax) was 6.7 and 4.6 μg equivalents/g, respectively. Peak plasma concentrations (Tmax) were reached at 4 and 8 hours after administration, respectively, with areas under the plasma concentration-time curve (AUC0-24 (h·μg/g)) of 67 and 51 for males and females, respectively. Unmetabolized NOA446510 accounted for 73% (male) and 54% (female) of the administered dose in feces (three other minor metabolites were also found in feces). Five major metabolites were found in 6-hour urine samples (unmetabolized NOA446510 was not detected). Unmetabolized NOA446510 and five other metabolites were found in mixed male and female plasma. …Following a single dose of 800 mg/kg, 4% of the radioactive dose was excreted in urine, and 71% (male) and 18% (female) of the dose were excreted in feces within 72 hours, respectively. Plasma Cmax concentrations were 25.0 and 17.5 μg equivalents/g, respectively. Tmax was reached at 6 and 10 hours post-administration in male and female dogs, respectively. The resulting total exposure/time (AUC0-24) values were 98 h·μg/g (male) and 152 h·μg/g (female), respectively. In feces, the majority of the recovered dose was unmetabolized NOA446510, at 67% (males) and 12% (females). Five major metabolites were found in 6-hour urine samples (four of which were identical to those found in the 100 mg/kg single-dose administration), but unmetabolized NOA446510 was not detected. Unmetabolized NOA446510 was detected in male and female plasma samples from 0–48 hours, along with three metabolites found in the 100 mg/kg single-dose sample. Following repeated administration of 100 mg/kg of NOA446510, 6% and 5% of the dose were excreted in urine in males and females, respectively, with an excretion rate of 77% in males. Within 72 hours, NOA446510 was detected in the feces of 79% (females). Cmax values were reached in men and women at 4 and 10 hours post-dose, respectively, at 6.0 and 4.3 μg equivalents/g (Tmax), with corresponding AUC0-24 values of 72 and 48 h·μg/g. The main component in feces was unmetabolized NOA446510, accounting for 59% in men and 70% in women (three minor metabolites were also detected in feces). Urine samples at 6 hours post-dose contained the same five metabolites as the single 100 mg/kg dose, plus two additional metabolites; unmetabolized NOA446510 was not detected. Plasma samples from both men and women contained unmetabolized NOA446510, the same three metabolites as the single 800 mg/kg dose sample, and one unidentified minor metabolite. Following repeated oral administration of an 800 mg/kg radiolabeled dose, 3% (males) and 4% (females) of the dose were detected in urine within 72 hours, while 91% (males) and 94% (females) of the dose were detected in feces, respectively. At 6 hours post-administration, the Cmax concentrations were 49.7 and 28.6 μg equivalents/g (Tmax for both sexes). The AUC0-24 values for total exposure/time were 379 h·μg/g (males) and 410 h·μg/g (females), respectively. Unmetabolized NOA446510 accounted for 52% (males) and 93% (females) of the dose in feces, respectively. Urine samples at 6 hours contained the same five metabolites as the 800 mg/kg single-dose group. Plasma samples from 0–48 hours contained NOA446510 and three metabolites identical to those in the 800 mg/kg single-dose group (a fourth metabolite was also identified in female plasma). Phase 2: Male dogs excreted 24% of the dose. Within 72 hours following a single intravenous injection of 3 mg/kg of radiolabeled drug, the drug concentration in urine was 31% in females, 45% in males, and 47% in females. The major urinary metabolites were the same as the five metabolites found in the Phase 1 single oral 100 mg/kg group, plus one more. In male plasma samples from 0-24 hours, unmetabolized NOA446510 and the five metabolites identified in the Phase 1 single oral 100 mg/kg group, plus one more, were detected. On day 15, following a single oral 3 mg/kg radiolabeled drug (gelatin capsule), within 72 hours, the drug concentration in urine was 14% in males and 21% in females, and the drug concentration in feces was 72% in males and 66% in females, respectively. The major urinary metabolites for each sex were the same as the five metabolites detected in the Phase 1 single oral group. A sixth metabolite was also found in the 100 mg/kg dose samples (female samples also contained this sixth metabolite). Two of the three metabolites found in a single oral dose of 800 mg/kg in male and female plasma pools from 0–24 hours were identified, along with another metabolite (other metabolites were detected, but at levels too low to be identified). A possible metabolic pathway was proposed: loss of one or two propargyl groups followed by conjugation with glucuronic acid or sulfate and O-demethylation without cleavage. Biological half-life Three male Alpk:APfSD (Wistar-derived) rats per group received (methoxyphenyl-U-(14)C)NOA446510/mandipromide/ at a dose of 3 mg/kg/day for 14 days. Three male rats per group were sacrificed 24 hours after administration on days 3, 7, 10, and 14. Tests were performed on days 1, 4, 7, 14, 21, 28, and 49 after 14 days of daily administration. ...Liver elimination was biphasic, with a terminal eli |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
Non-Human Toxicity Values
Rat inhalation LC50 > 5.19 +/- 0.55 mg/L/4 hours; Rat dermal LD50 > 5,050 mg/kg; Rat oral LD50 > 5,000 mg/kg |
| 其他信息 |
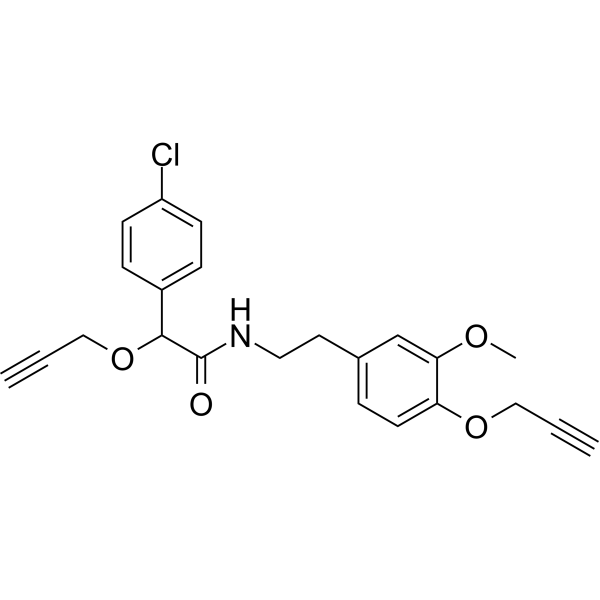
2-(4-Chlorophenyl)-N-{2-[3-methoxy-4-(prop-2-yn-1-oxy)phenyl]ethyl}-2-(prop-2-yn-1-oxy)acetamide is a monocarboxylic acid amide formed by the condensation of the carboxyl group of p-chloromandelic acid propargyl ether with the amino group of 2-[3-methoxy-4-(prop-2-yn-1-oxy)phenyl]ethylamine. It is a monocarboxylic acid amide, a terminal alkyne compound, an aromatic ether, and also a monochlorobenzene compound.
Mechanism of Action …This study describes…the molecular mechanism of resistance to carboxylic acid amide (CAA) fungicides. …A family of four cellulase (CesA) genes containing conserved domains exists in all continuous glycosyltransferases. Phylogenetic analysis shows that they are closely related to cellulase in Phytophthora sp. (a mandeprothylamine target species). Sequencing of the CesA gene in CAA-resistant and susceptible field isolates revealed five single nucleotide polymorphisms (SNPs) affecting the protein's amino acid structure. SNP inheritance in F1, F2, and F3 progeny confirmed that resistance was associated with a single SNP located in PvCesA3. Only when this SNP was present in two alleles did it lead to the substitution of glycine residue at position 1105 of the deduced amino acid sequence with serine (G1105S), thus conferring CAA resistance. Our data suggest that the identified gene is the putative cellulase synthase, and that a recessive mutation in PvCesA3 results in genetic resistance to the carboxylic amide fungicide mandepromine. Mandepromine (MPD) is a carboxylic amide (CAA) effective against downy mildew, such as Plasmopara viticola, the causal agent of grape downy mildew, and late blight of potato caused by Phytophthora infestans. …Here, …we combined biochemical and genetic techniques to identify the molecular target of MPD in Phytophthora infestans. Treatment of germinated sporangia of Phytophthora infestans with MPD resulted in swelling symptoms typical of cell wall synthesis inhibitors, which were reversible upon washing with water. Absorption studies using MPD labeled with 14C indicated that this oomycete controller acts on the cell wall rather than entering the cell interior. Furthermore, the presence of MPD interfered with the incorporation of (14)C glucose into cellulose, suggesting that inhibition of cellulose synthesis is the primary function of MPD. We obtained MPD-insensitive laboratory mutants using ethyl methanesulfonate (EMS) mutagenesis. Sequence analysis of the cellulose synthase gene in these mutants revealed two point mutations in the PiCesA3 gene, which is known to be involved in cellulose synthesis. Both mutations in the PiCesA3 gene resulted in an alteration of the same amino acid (glycine-1105) in the protein. To demonstrate that the mutation in PiCesA3 is the cause of the MPD insensitivity phenotype, we transformed and expressed the mutated PiCesA3 allele in susceptible wild-type strains. |
| 分子式 |
C23H22NO4CL
|
|---|---|
| 分子量 |
411.87808
|
| 精确质量 |
411.123
|
| CAS号 |
374726-62-2
|
| PubChem CID |
11292824
|
| 外观&性状 |
Light beige powder
|
| 密度 |
1.2±0.1 g/cm3
|
| 沸点 |
608.6±55.0 °C at 760 mmHg
|
| 熔点 |
96-97ºC
|
| 闪点 |
321.9±31.5 °C
|
| 蒸汽压 |
0.0±1.7 mmHg at 25°C
|
| 折射率 |
1.575
|
| LogP |
4.16
|
| tPSA |
60.28
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
10
|
| 重原子数目 |
29
|
| 分子复杂度/Complexity |
599
|
| 定义原子立体中心数目 |
0
|
| SMILES |
C#CCOC(C1C=CC(Cl)=CC=1)C(NCCC1C=CC(OCC#C)=C(OC)C=1)=O
|
| InChi Key |
KWLVWJPJKJMCSH-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C23H22ClNO4/c1-4-14-28-20-11-6-17(16-21(20)27-3)12-13-25-23(26)22(29-15-5-2)18-7-9-19(24)10-8-18/h1-2,6-11,16,22H,12-15H2,3H3,(H,25,26)
|
| 化学名 |
2-(4-chlorophenyl)-N-[2-(3-methoxy-4-prop-2-ynoxyphenyl)ethyl]-2-prop-2-ynoxyacetamide
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4279 mL | 12.1395 mL | 24.2789 mL | |
| 5 mM | 0.4856 mL | 2.4279 mL | 4.8558 mL | |
| 10 mM | 0.2428 mL | 1.2139 mL | 2.4279 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。