| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 体外研究 (In Vitro) |
采用人肝微粒体(HLM)和受控给药尿液样本研究metizolam代谢。在HLM中,metizolam(10 μM)主要通过CYP3A4/5代谢为α-羟基metizolam和二氢二醇-metizolam。动力学分析显示α-羟基metizolam生成的Km = 18.2 μM,Vmax = 0.41 nmol/min/mg蛋白。[1]
|
|---|---|
| 体内研究 (In Vivo) |
采用人肝微粒体(HLM)和受控给药尿液样本研究metizolam代谢。在HLM中,metizolam(10 μM)主要通过CYP3A4/5代谢为α-羟基metizolam和二氢二醇-metizolam。动力学分析显示α-羟基metizolam生成的Km = 18.2 μM,Vmax = 0.41 nmol/min/mg蛋白。[1]
|
| 药代性质 (ADME/PK) |
The metabolism of methezolam was investigated using human liver microsomes (HLM) and urine samples from a controlled dosing study. In the HLM, methezolam (10 μM) was primarily metabolized by CYP3A4/5 to α-hydroxymethezolam and dihydrodiolmethezolam. Kinetic analysis showed that the Km of α-hydroxymethezolam was 18.2 μM and the Vmax was 0.41 nmol/min/mg protein. [1] After oral administration of 1 mg methezolam, 0.3% of the total excretion of unmethezolam was observed in human urine. The main metabolites were α-hydroxymethezolam glucuronide (23.6%), dihydrodiolmethezolam glucuronide (17.8%), and desmethylmethezolam (12.1%). The urine detection window was 24–48 hours. [1]
After a single recreational use of methezolam, concentrations of 2.7–22.1 pg/mg were detected in hair (1–2 cm long fragments), and concentrations of 0.5–3.2 ng/mL were detected in blood. [2] |
| 毒性/毒理 (Toxicokinetics/TK) |
Acute toxicity: The oral LD50 in mice was reported to be approximately 700 mg/kg (from an external source, undetermined). [1] No hepatotoxicity was observed in case studies at recreational doses (0.5–2 mg). Plasma protein binding was not quantified. [2]
|
| 参考文献 |
[1]. Detection of the designer benzodiazepine metizolam in urine and preliminary data on its metabolism. Drug Test Anal. 2017 Jul;9(7):1026-1033.
[2]. Characterization of metizolam, a designer benzodiazepine, in alternative biological specimens. Toxicologie Analytique et Clinique. 2017 Feb; 29(1): 57-63. |
| 其他信息 |
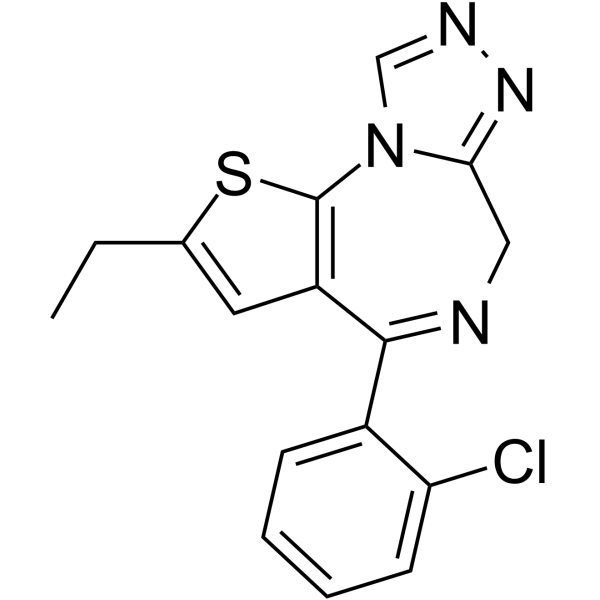
Metizolam is a novel benzodiazepine drug of the thienodiazepine class, with a structure similar to etizolam. It is not approved for medical use and is distributed as an "investigation chemical." [1]
Forensic identification: The main metabolites in urine (α-hydroxymetizolam glucuronide and dihydrodiol-metizolam) can serve as markers of ingestion. Hair analysis has a longer detection window than urine analysis. [2] |
| 分子式 |
C16H13CLN4S
|
|---|---|
| 分子量 |
328.8192
|
| 精确质量 |
328.054
|
| CAS号 |
40054-68-0
|
| PubChem CID |
12434325
|
| 外观&性状 |
Typically exists as Light yellow to yellow solid at room temperature
|
| 密度 |
1.5±0.1 g/cm3
|
| 沸点 |
527.2±60.0 °C at 760 mmHg
|
| 闪点 |
272.6±32.9 °C
|
| 蒸汽压 |
0.0±1.4 mmHg at 25°C
|
| 折射率 |
1.752
|
| LogP |
3.39
|
| tPSA |
71.3
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
22
|
| 分子复杂度/Complexity |
446
|
| 定义原子立体中心数目 |
0
|
| SMILES |
ClC1=C([H])C([H])=C([H])C([H])=C1C1C2C([H])=C(C([H])([H])C([H])([H])[H])SC=2N2C([H])=NN=C2C([H])([H])N=1
|
| InChi Key |
NQSSWDKQLVBUQN-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H13ClN4S/c1-2-10-7-12-15(11-5-3-4-6-13(11)17)18-8-14-20-19-9-21(14)16(12)22-10/h3-7,9H,2,8H2,1H3
|
| 化学名 |
7-(2-chlorophenyl)-4-ethyl-3-thia-1,8,11,12-tetrazatricyclo[8.3.0.02,6]trideca-2(6),4,7,10,12-pentaene
|
| 别名 |
Metizolam; Desmethyletizolam; 40054-68-0; K1C6XI9LLX; Metizolam [NFLIS-DRUG]; 4-(2-Chlorophenyl)-2-ethyl-6H-thieno(3,2-f)(1,2,4)triazolo(4,3-a)(1,4)diazepine; DTXSID101032847; 6H-Thieno(3,2-f)(1,2,4)triazolo(4,3-a)(1,4)diazepine, 4-(2-chlorophenyl)-2-ethyl-;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0412 mL | 15.2059 mL | 30.4118 mL | |
| 5 mM | 0.6082 mL | 3.0412 mL | 6.0824 mL | |
| 10 mM | 0.3041 mL | 1.5206 mL | 3.0412 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。