| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 药代性质 (ADME/PK) |
Metabolism / Metabolites
Carbamates are enzymatically hydrolyzed in the liver; degradation products are excreted via the kidneys and liver. (L793) |
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
Toxicity Summary
Methoxycarb is a cholinesterase, or acetylcholinesterase (AChE) inhibitor. Carbamate compounds carbamate esters form unstable complexes with cholinesterase by carbamylation of the enzyme's active site. This inhibition is reversible. Cholinesterase inhibitors suppress the activity of acetylcholinesterase. Because acetylcholinesterase plays an important physiological role, chemicals that interfere with its activity are potent neurotoxins, causing excessive salivation and lacrimation even at low doses. High-dose exposure typically results in symptoms such as headache, salivation, nausea, vomiting, abdominal pain, and diarrhea. Acetylcholinesterase breaks down the neurotransmitter acetylcholine, which is released at the neuromuscular junction, causing muscle or organ relaxation. Inhibition of acetylcholinesterase results in the accumulation and sustained action of acetylcholine, leading to continuous nerve impulse transmission and an inability to stop muscle contractions. Toxicity Data LC50 (Rat) = 475 mg/m3Interactions This study investigated the combined toxicity of malathion, 2-sec-butylphenyl methylcarbamate, m-toluenemethylcarbamate, and 3,4-xylmethylcarbamate in mice and rats. ICR mice and male Fischer 344 rats were orally exposed to five different concentrations of insecticide suspensions to determine the median lethal dose (LD50); animal mortality was observed for at least 7 days post-administration. … Mice were sacrificed, brain tissue was homogenized, and brain acetylcholinesterase activity was measured. Of all the combinations tested, only the combination of malathion and 2-sec-butylphenyl methylcarbamate showed a significant synergistic effect in acute toxicity in male mice; other combinations with m-toluenemethylcarbamate or 3,4-xylmethylcarbamate did not show a significant enhancing effect on toxicity in male mice. Female mice showed similar synergistic responses to the insecticides. In rats, the strength of this synergistic effect was relatively lower than in mice. Symptoms such as muscle fasciculations, increased salivation, increased urination, convulsions, and dyspnea were similar in mice and rats. Significant differences existed in the time to death after administration between different insecticides and between rats and mice. ... Succinylcholine, other cholinergic drugs, and aminophylline are contraindicated. /Carbamates and Related Compounds/ Non-human Toxicity Values LD50 Male rat, oral: 580 mg/kg LD50 Female rat, oral: 498 mg/kg LD50 Mouse, oral: 109 mg/kg LD50 Rat, dermal: >2,000 mg/kg For more complete non-human toxicity data on METOLCARB (6 in total), please visit the HSDB record page. |
| 参考文献 |
[1]. Jingwei Sun, et al. Development of enzyme linked immunoassay for the simultaneous detection of carbaryl and metolcarb in different agricultural products. Anal Chim Acta. 2010 May 7;666(1-2):76-82.
|
| 其他信息 |
Metolcarb is a colorless crystalline solid. It is an insecticide used to control rice planthoppers, codling moths, citrus mealybugs, onion thrips, fruit flies, cotton bollworms, and aphids. It is not registered as an insecticide in the United States (EPA, 1998). Metolcarb is a carbamate compound. It has multiple functions, including as an EC 3.1.1.7 (acetylcholinesterase) inhibitor, carbamate insecticide, acaricide, and agrochemical. Its structure is similar to methylcarbamate and m-cresol. Metolcarb is a synthetic carbamate compound and acetylcholinesterase inhibitor used as an insecticide. It is a colorless crystalline solid that can be exposed through inhalation, ingestion, or contact. Metolcarb is a carbamate insecticide. Carbamate insecticides are derived from carbamate and their insecticidal action is similar to that of organophosphate insecticides. They are widely used in homes, gardens, and agriculture. The first carbamate insecticide, Sevin, was introduced in 1956, and its global usage exceeds that of all other carbamate insecticides combined. Due to its relatively low oral and dermal toxicity to mammals and its broad spectrum of application, Sevin is widely used in lawns and gardens. Most carbamate insecticides are highly toxic to hymenopteran insects, therefore precautions must be taken to prevent contact with them by insects such as bees or parasitic wasps. Some carbamate insecticides can be transported within plants, making them effective systemic treatments. (L795)
Mechanism of Action Cholinesterase inhibitor. |
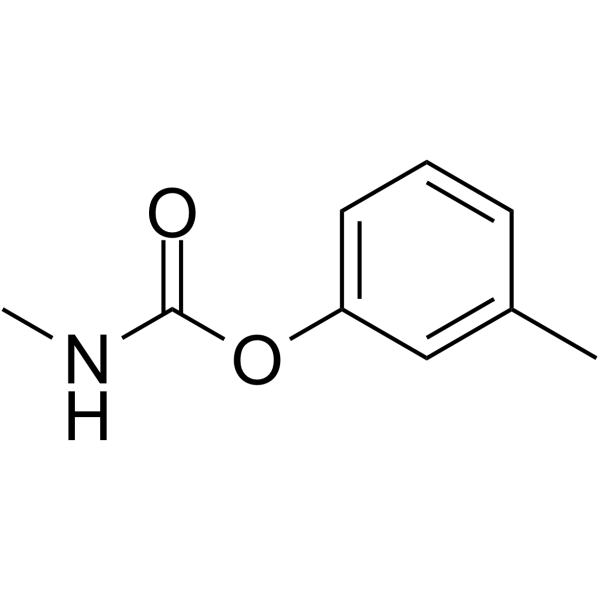
| 分子式 |
C9H11NO2
|
|---|---|
| 分子量 |
165.19
|
| 精确质量 |
165.078
|
| CAS号 |
1129-41-5
|
| 相关CAS号 |
Metolcarb-d3;1777782-68-9
|
| PubChem CID |
14322
|
| 外观&性状 |
Colorless crystalline solid
|
| 密度 |
1.1±0.1 g/cm3
|
| 沸点 |
241.6±23.0 °C at 760 mmHg
|
| 熔点 |
74-77 °C
|
| 闪点 |
99.9±22.6 °C
|
| 蒸汽压 |
0.0±0.5 mmHg at 25°C
|
| 折射率 |
1.517
|
| LogP |
1.63
|
| tPSA |
38.33
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
12
|
| 分子复杂度/Complexity |
159
|
| 定义原子立体中心数目 |
0
|
| SMILES |
CC1=CC(OC(NC)=O)=CC=C1
|
| InChi Key |
VOEYXMAFNDNNED-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C9H11NO2/c1-7-4-3-5-8(6-7)12-9(11)10-2/h3-6H,1-2H3,(H,10,11)
|
| 化学名 |
(3-methylphenyl) N-methylcarbamate
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.0536 mL | 30.2682 mL | 60.5364 mL | |
| 5 mM | 1.2107 mL | 6.0536 mL | 12.1073 mL | |
| 10 mM | 0.6054 mL | 3.0268 mL | 6.0536 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。