| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
醋酸戊二酰胺(25–100 mg/kg;Sc;5 天)可提高 BDNF 水平[3]。用于亨廷顿病 (HD) 动物模型的 N171-82Q 转基因小鼠系显示出更快的病程。从8周龄开始一直持续到20周龄(接近该疾病导致死亡的年龄),小鼠给予1毫克/只; SC; 5×周。多项运动功能评估显示,使用醋酸戊二醛可改善运动功能。对 15 周大的 N171-82Q 转基因小鼠进行为期 4 天的转棒测试,结果显示,给予醋酸戊二酰胺后,小鼠的表现有显着改善[3]。
|
|---|---|
| 参考文献 |
[1]. McKeage K. Glatiramer Acetate 40 mg/mL in Relapsing-Remitting Multiple Sclerosis: A Review. CNS Drugs. 2015;29(5):425-432.
[2]. Arnon R, et al. Mechanism of action of glatiramer acetate in multiple sclerosis and its potential for the development of new applications. Proc Natl Acad Sci U S A. 2004;101 Suppl 2(Suppl 2):14593-14598. [3]. Corey-Bloom J, et al. Beneficial effects of glatiramer acetate in Huntington's disease mouse models: Evidence for BDNF-elevating and immunomodulatory mechanisms. Brain Res. 2017;1673:102-110. [4]. Aharoni R, et al. Glatiramer acetate-specific T cells in the brain express T helper 2/3 cytokines and brain-derived neurotrophic factor in situ [published correction appears in Proc Natl Acad Sci U S A. 2005 Aug 23;102(34):12288]. Proc Natl Acad Sci U S A |
| 其他信息 |
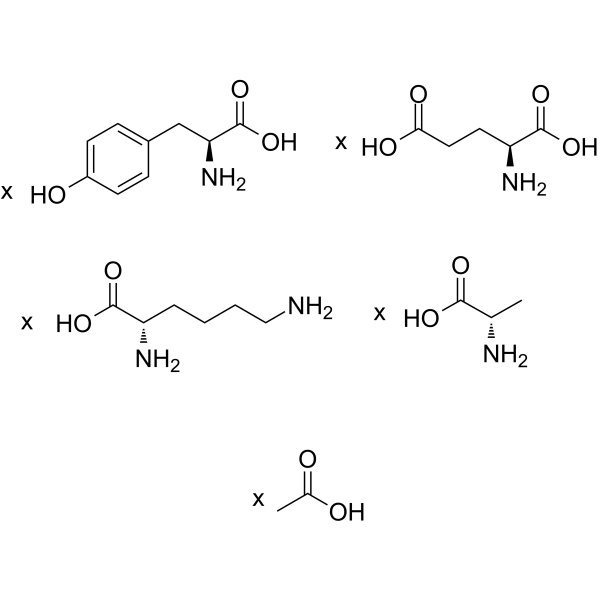
一种由L-丙氨酸、L-谷氨酸、L-赖氨酸和L-酪氨酸组成的随机聚合物,其结构与髓鞘碱性蛋白相似。它用于治疗复发缓解型多发性硬化症。
另见:醋酸格拉替雷(注释已移至)。 |
| 分子式 |
(C9H11NO3.C6H14N2O2.C5H9NO4.C3H7NO2)X.XC2H4O2
|
|---|---|
| 分子量 |
623.65
|
| 精确质量 |
623.301
|
| CAS号 |
147245-92-9
|
| PubChem CID |
3081884
|
| 外观&性状 |
Off-white to light yellow solid powder
|
| 沸点 |
385.2ºC at 760mmHg
|
| 熔点 |
>239°C (dec.) (lit.)
|
| 闪点 |
186.7ºC
|
| 蒸汽压 |
1.27E-06mmHg at 25°C
|
| LogP |
2.147
|
| tPSA |
374.13
|
| 氢键供体(HBD)数目 |
12
|
| 氢键受体(HBA)数目 |
18
|
| 可旋转键数目(RBC) |
13
|
| 重原子数目 |
43
|
| 分子复杂度/Complexity |
519
|
| 定义原子立体中心数目 |
4
|
| SMILES |
CC(=O)O.NCCCCC(C(=O)O)N.OC(CCC(C(=O)O)N)=O.CC(C(=O)O)N.OC(C(CC1=CC=C(O)C=C1)N)=O
|
| InChi Key |
FHEAIOHRHQGZPC-KIWGSFCNSA-N
|
| InChi Code |
InChI=1S/C9H11NO3.C6H14N2O2.C5H9NO4.C3H7NO2.C2H4O2/c10-8(9(12)13)5-6-1-3-7(11)4-2-6;7-4-2-1-3-5(8)6(9)10;6-3(5(9)10)1-2-4(7)8;1-2(4)3(5)6;1-2(3)4/h1-4,8,11H,5,10H2,(H,12,13);5H,1-4,7-8H2,(H,9,10);3H,1-2,6H2,(H,7,8)(H,9,10);2H,4H2,1H3,(H,5,6);1H3,(H,3,4)/t8-;5-;3-;2-;/m0000./s1
|
| 化学名 |
acetic acid;(2S)-2-amino-3-(4-hydroxyphenyl)propanoic acid;(2S)-2-aminopentanedioic acid;(2S)-2-aminopropanoic acid;(2S)-2,6-diaminohexanoic acid
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O: 50 mg/mL
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6035 mL | 8.0173 mL | 16.0346 mL | |
| 5 mM | 0.3207 mL | 1.6035 mL | 3.2069 mL | |
| 10 mM | 0.1603 mL | 0.8017 mL | 1.6035 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
A Study in Subjects With Relapsing-Remitting Multiple Sclerosis (RRMS) to Assess the Efficacy, Safety and Tolerability of Glatiramer Acetate (GA) Injection 40 mg Administered Three Times a Week Compared to Placebo
CTID: NCT01067521
Phase: Phase 3 Status: Completed
Date: 2021-12-09