| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
D2 Receptor ( Ki = 1.8 nM ); D3 Receptor ( Ki = 3.5 nM ); D4 Receptor ( Ki = 2400 nM )); D1 Receptor ( Ki = 18000 nM )
Dopamine D₂ receptor: Raclopride binds with a dissociation constant (Ki) of 1.8 nM (Table 2).[1] Dopamine D₃ receptor: Raclopride binds with a dissociation constant (Ki) of 3.5 nM (Table 2).[1] Dopamine D₄ receptor: Raclopride has very low affinity, with a dissociation constant (Ki) of 2400 nM (Table 2).[1] |
|---|---|
| 体外研究 (In Vitro) |
[³H]雷氯必利可以轻易标记脑组织中的多巴胺D₂和D₃受体,但由于其对D₄受体亲和力低,不能标记D₄受体。[1]
在人纹状体中,通过[¹¹C]雷氯必利结合测得的D₂受体密度,是使用[¹¹C]甲基螺环哌隆测得的密度的1.7-1.8倍,这表明其与受体单体/二聚体的结合存在差异。[1] 在治疗条件下,通过[¹¹C]雷氯必利PET成像测量,雷氯必利占据了人类60%至75%的D₂受体。[1] 雷氯必利被归类为典型的神经阻滞剂(抗精神病药),可阻断D₂受体。[1] |
| 体内研究 (In Vivo) |
酒石酸雷氯必利(0.1、0.3 或 0.6 mg/kg;腹腔注射;30 分钟;OF1 品系的白化雄性小鼠)显着减少分配给攻击行为的时间[2]。
Raclopride是一种替代苯甲酰胺,作为中枢多巴胺能D2受体的拮抗剂,具有高选择性和潜在的抗精神病作用。与经典的DA受体阻滞剂如氟哌啶醇相比,雷氯pride在锥体外系副作用的临床前试验中表现出非典型特征。raclopride对拮抗行为的抗攻击特性尚未得到充分的研究。本实验研究了0.1、0.3、0.6 mg/kg盐酸雷氯pride对雄性小鼠攻击行为和运动行为的影响。注射后30分钟进行攻击试验。会面被录下来,行为被评估,用11大类行为来衡量所花的时间。结果显示,雷氯pride有明显的抗攻击作用,几乎没有运动障碍,但探索性行为有所增加。这种行为特征与观察到的其他非典型抗精神病药非常相似,与经典化合物中的发现有所不同。[2] 急性腹腔注射雷氯必利(0.1、0.3、0.6 mg/kg)在隔离饲养的雄性小鼠中产生了明确的剂量依赖性抗攻击作用,所有三个剂量均显著减少了攻击行为(与生理盐水对照组相比,p < 0.02)。[2] 雷氯必利处理未引起显著的运动功能损害。即使在最高剂量(0.6 mg/kg)下,不动时间也极少且未显著增加。[2] 给予雷氯必利后,非社会性探索行为表现出不显著的、剂量依赖性的增加。[2] 雷氯必利的行为特征(抗攻击作用且运动损害最小)与其他非典型神经阻滞剂(如舒必利和氯氮平)相似,而与产生明显运动损害的经典神经阻滞剂(如氟哌啶醇)不同。[2] |
| 动物实验 |
动物:本研究使用了90只雄性OF1品系白化小鼠。其中一半单独饲养30天,以诱导隔离诱导的攻击行为。另一半小鼠群养,并用4%硫酸锌溶液进行鼻内灌洗使其暂时丧失嗅觉,作为非攻击性的“标准对手”。[2]
药物配制和给药:酒石酸雷克洛必利溶于生理盐水中。小鼠腹腔注射(IP)0.1、0.3或0.6 mg/kg剂量的雷克洛必利,注射体积为0.01 ml/g体重。对照组小鼠仅注射生理盐水。[2] 实验步骤:注射后30分钟进行攻击行为测试。实验小鼠(经处理)和标准对照小鼠(嗅觉缺失)被置于一个中性玻璃笼(60×33×30厘米)中,进行10分钟的接触。接触前,两组小鼠之间有1分钟的适应期,适应期之间用屏障隔开。接触过程在光暗周期的黑暗期,于白光下进行录像。[2] 行为分析:录像由一位不知晓实验分组的观察者使用定制程序进行分析。仅记录实验组和对照组小鼠在11个主要行为类别中花费的时间。[2] |
| 药代性质 (ADME/PK) |
雷克洛必利 能轻易穿过血脑屏障,注射后不久即可在大脑中达到高浓度。[2]
|
| 毒性/毒理 (Toxicokinetics/TK) |
雷氯必利可诱发动物僵直,这是典型D₂受体拮抗剂的常见副作用。[1]
利坦色林(一种5-HT₂受体拮抗剂)不能拮抗雷氯必利诱发的僵直,表明僵直主要由D₂受体阻滞介导。[1] |
| 参考文献 | |
| 其他信息 |
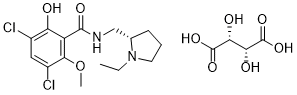
3,5-二氯-N-[[(2S)-1-乙基-2-吡咯烷基]甲基]-2-羟基-6-甲氧基苯甲酰胺属于水杨酰胺类化合物。
雷克洛必利已用于帕金森病的研究试验。 它是一种具有抗精神病特性的取代苯甲酰胺。它是多巴胺D2受体(参见“受体,多巴胺D2”)拮抗剂。 多巴胺受体是治疗精神分裂症、帕金森病和亨廷顿舞蹈症的主要靶点,Philip Seeman和Hubert Van Tol在这篇综述中对此进行了讨论。选择性靶向特定亚型多巴胺受体的药物可以改善治疗效果。大多数抗精神病药物阻断D2受体的程度与临床疗效直接相关,但氯氮平除外,它更倾向于D4受体。 D1 和 D2 受体可以相互增强作用,这可能通过 G 蛋白亚基实现。在精神分裂症中,D2 和 D3 受体密度升高 10%,而 D4 受体密度升高 600%。因此,D4 受体可能成为未来抗精神病药物的靶点。虽然抗精神病药物最初帮助发现了多巴胺受体,但目前克隆的五种多巴胺受体正在促进选择性抗精神病药物和抗帕金森病药物的发现。 抗精神病药物 这类药物可以控制躁动性精神病行为,缓解急性精神病状态,减轻精神病症状,并具有镇静作用。它们用于治疗精神分裂症、老年痴呆症、术后短暂性精神病或心肌梗死等。这些药物通常被称为神经阻滞剂,暗示其可能产生神经系统副作用,但并非所有抗精神病药物都会产生此类副作用。许多此类药物也可能对恶心、呕吐和瘙痒有效。(参见所有归类为抗精神病药物的化合物。) 多巴胺拮抗剂 这类药物与多巴胺受体结合但不激活受体,从而阻断多巴胺或外源性激动剂的作用。许多用于治疗精神病性疾病的药物(抗精神病药物)都是多巴胺拮抗剂,尽管它们的治疗效果可能归因于大脑的长期调节,而非阻断多巴胺受体的急性效应。多巴胺拮抗剂还被用于其他一些临床用途,包括作为止吐药、治疗图雷特综合征和治疗呃逆。多巴胺受体阻滞与神经阻滞剂恶性综合征相关。 雷氯必利是一种苯甲酰胺类抗精神病药物,也是一种选择性多巴胺D₂/D₃受体拮抗剂。[1] 它可用作放射性配体([³H]雷氯必利和[¹¹C]雷氯必利),用于标记和定量体外和体内(PET成像)的D₂和D₃受体。[1] [³H]奈莫那必利(标记D₂、D₃和D₄)和[³H]雷氯必利(标记D₂和D₃)结合密度的差异可用于估计组织中D₄受体的密度。[1] 大多数典型的抗精神病药物,包括雷氯必利,主要作用于D₂和D₃受体,这与靶向D₄受体的氯氮平不同。 D₄受体。[1] |
| 分子式 |
C19H26CL2N2O9
|
|---|---|
| 分子量 |
497.32374
|
| 精确质量 |
496.102
|
| 元素分析 |
C, 45.89; H, 5.27; Cl, 14.26; N, 5.63; O, 28.95
|
| CAS号 |
98185-20-7
|
| 相关CAS号 |
Raclopride; 84225-95-6
|
| PubChem CID |
16219926
|
| 外观&性状 |
Solid powder
|
| LogP |
1.127
|
| tPSA |
176.86
|
| 氢键供体(HBD)数目 |
6
|
| 氢键受体(HBA)数目 |
10
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
32
|
| 分子复杂度/Complexity |
520
|
| 定义原子立体中心数目 |
3
|
| SMILES |
CC[NH+]1CCC[C@H]1CNC(=O)c2c(c(cc(c2OC)Cl)Cl)O.[C@@H]([C@H](C(=O)[O-])O)(C(=O)O)O
|
| InChi Key |
QULBVRZTKPQGCR-NDAAPVSOSA-N
|
| InChi Code |
InChI=1S/C15H20Cl2N2O3.C4H6O6/c1-3-19-6-4-5-9(19)8-18-15(21)12-13(20)10(16)7-11(17)14(12)22-2;5-1(3(7)8)2(6)4(9)10/h7,9,20H,3-6,8H2,1-2H3,(H,18,21);1-2,5-6H,(H,7,8)(H,9,10)/t9-;1-,2-/m01/s1
|
| 化学名 |
3,5-dichloro-N-[[(2S)-1-ethylpyrrolidin-2-yl]methyl]-2-hydroxy-6-methoxybenzamide;(2R,3R)-2,3-dihydroxybutanedioic acid
|
| 别名 |
Raclopride; FLA-870; FLA870; Raclopride tartrate; 98185-20-7; EXZ5FGZ55J; Raclopride (tartrate); UNII-EXZ5FGZ55J; 98185-20-7 (tartrate); (S)-3,5-dichloro-N-((1-ethylpyrrolidin-2-yl)methyl)-2-hydroxy-6-methoxybenzamide (2R,3R)-2,3-dihydroxysuccinate; (S)-(-)-3,5-Dichloro-N-((1-ethyl-2-pyrrolidinyl)methyl)-6-methoxysalicylamide L-(+)-tartrate; FLA 870; Raclopride tartrate
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~100 mg/mL (~288 mM)
H2O: ~0.1 mg/mL (~0.3 mM) |
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0108 mL | 10.0539 mL | 20.1078 mL | |
| 5 mM | 0.4022 mL | 2.0108 mL | 4.0216 mL | |
| 10 mM | 0.2011 mL | 1.0054 mL | 2.0108 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05282277 | Recruiting | Drug: Transdermal Estradiol Drug: Raclopride C11 |
Depression Psychosis Anhedonia |
University of North Carolina, Chapel Hill |
April 20, 2022 | Phase 4 |
| NCT02169310 | Recruiting | Device: tDCS Drug: [11C] Raclopride |
Traumatic Brain Injury | National Institute of Neurological Disorders and Stroke (NINDS) |
November 18, 2014 | Phase 1 |
| NCT03190954 | Recruiting | Drug: [11C]raclopride plus placebo Drug: [11C]raclopride plus drug |
Normal Physiology Opioid Use Disorders |
National Institute on Alcohol Abuse and Alcoholism (NIAAA) |
August 17, 2017 | Early Phase 1 |
| NCT03648892 | Completed | Drug: [c11] raclopride Drug: [18F]fallypride |
Obesity Overweight Healthy Volunteers |
National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) |
September 21, 2018 | Early Phase 1 |
| NCT02020408 | Completed | Drug: [11C]raclopride Drug: [11C]DASB Drug: amphetamine |
Eating Disorder | University of California, San Diego |
May 2011 | Phase 4 |