| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 靶点 |
Melanocortin receptors: MC1R, MC4R, MC3R, MC5R, and MC2R
|
|---|---|
| 体外研究 (In Vitro) |
布美拉肽是一种多种黑皮质素受体的激动剂,按其效力强弱依次为MC1R、MC4R、MC3R、MC5R和MC2R。目前尚不清楚激动这些受体如何改善性欲减退障碍,但MC4R受体广泛存在于中枢神经系统的多个区域。MC3R和MC4R分布于下丘脑,参与食物摄入和能量稳态调节。一种理论认为,布美拉肽刺激内侧视前区的多巴胺释放,该区域与多种生物的性行为相关。
布美拉肽是一种黑皮质素受体激动剂,需在预期性行为前45分钟注射使用。激动黑皮质素受体MC1R也会导致黑色素表达增加。
|
| 体内研究 (In Vivo) |
布美拉肽 是一种由7个氨基酸组成的多肽,用于治疗绝经前女性的性欲减退障碍。布美拉肽与酒精无相互作用。目前,其作用于受体的机制如何转化为临床疗效尚不明确。该药物首次在文献中被描述是在2003年,当时的研发代号为PT-141。此后,针对其治疗男女性功能障碍的潜力进行了研究,但目前仅获批用于女性。其他用于治疗女性性功能障碍的药物包括[氟班色林]、[雌激素]、[奥培米芬] 和 [普拉睾酮]。布美拉肽于2019年6月21日获得美国食品药品监督管理局批准。
布美拉肽是一种黑皮质素受体激动剂,其作用机制即基于此特性。
作为一种通过注射给药的黑皮质素受体激动剂,布美拉肽用于治疗女性性欲减退障碍。据报道,布美拉肽在治疗期间可能导致轻微的血清转氨酶升高,并与少数临床明显的急性肝损伤病例有关。
布美拉肽 是一种蛋白质类药物,其最高临床试验阶段(在所有适应症中)为IV期,于2019年首次获批,目前拥有3个已获批适应症和3个研究中的适应症。
|
| 药代性质 (ADME/PK) |
吸收、分布和排泄
Bremelanotide 的 Tmax 为 1.0 小时(0.5-1.0 小时),生物利用度为 100%。 Cmax 为 72.8 ng/mL,AUC 为 276 小时ng/mL。 64.8% 的放射性标记剂量经尿液排泄,22.8% 的剂量经粪便回收。 布美拉诺肽的平均分布容积为 25.0±5.8 升。 布美拉诺肽的平均清除率为 6.5±1.0 升/小时。 代谢/代谢物 布美拉诺肽由 7 个氨基酸组成,因此其代谢涉及多种水解反应。 生物半衰期 布美拉诺肽的半衰期为 2.7 小时(1.9-4.0 小时)。 |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
在预注册临床试验中,少数接受布美拉诺肽治疗的患者报告出现血清酶升高,而安慰剂组患者中出现类似比例的肝酶升高。一名患者在一年内接受了10次布美拉诺肽注射,出现一例急性肝炎,表现为血清转氨酶水平显著升高、碱性磷酸酶轻度升高和轻度黄疸,停药后症状消退。目前尚未有布美拉诺肽导致急性肝衰竭或慢性肝损伤的病例报告,但该药物的临床应用经验总体有限。因此,布美拉诺肽可能导致急性肝损伤,但这种情况很少见。 可能性评分:D(可能是临床上明显的肝损伤的罕见原因)。 妊娠和哺乳期影响 ◉ 哺乳期用药概述 目前尚无布美拉诺肽在哺乳期临床应用的信息。由于布美拉诺肽是一种分子量为1025的环状肽分子,其在乳汁中的含量可能非常低,且由于可能在婴儿的胃肠道中被破坏,因此不太可能被吸收。在获得更多数据之前,哺乳期妇女应谨慎使用布美拉诺肽,尤其是在哺乳新生儿或早产儿时。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 蛋白结合 布美拉诺肽在血清中的蛋白结合率为21%。 |
| 参考文献 | |
| 其他信息 |
布美拉诺肽是一种寡肽。
布美拉诺肽是一种由7个氨基酸组成的肽,用于治疗绝经前女性的性欲低下症。布美拉诺肽不与酒精发生相互作用。布美拉诺肽作用于受体并产生临床疗效的机制尚不明确。布美拉诺肽最早于2003年在文献中被报道,当时的研究代号为PT-141。此后,人们曾研究其在治疗男性和女性性功能障碍方面的应用,但目前仅批准用于女性。其他用于治疗女性性功能障碍的药物包括氟班色林、雌激素、奥培米芬和普拉睾酮。布美拉诺肽于2019年6月21日获得美国食品药品监督管理局(FDA)的批准。 布美拉诺肽是一种黑皮质素受体激动剂。布美拉诺肽的作用机制是作为黑皮质素受体激动剂。 布美拉诺肽是一种肠外给药的黑皮质素受体激动剂,用于治疗女性性欲低下症。据报道,布美拉诺肽在治疗期间会导致轻度血清酶升高,并且与罕见的临床表现明显的急性肝损伤病例有关。 另见:醋酸布美拉诺肽(活性成分)。 药物适应症 布美拉诺肽适用于治疗绝经前女性的性欲低下症,该性欲低下症并非由内科或精神疾病、伴侣关系问题或药物副作用引起。 作用机制 布美拉诺肽是多种黑皮质素受体的激动剂,按效力顺序排列为MC1R、MC4R、MC3R、MC5R和MC2R。目前尚不清楚这些受体激动剂如何改善性欲低下症,但MC4R受体存在于中枢神经系统的许多区域。MC3R和MC4R存在于下丘脑,参与食物摄入和能量稳态。一种理论认为,布美拉诺肽刺激内侧视前区多巴胺的释放,而内侧视前区参与多种生物的性行为。 药效学 布美拉诺肽是一种黑皮质素受体激动剂,在预期性活动前45分钟注射。黑皮质素受体MC1R的激动作用也会导致黑色素表达增加。服用布美拉诺肽的患者也可能出现恶心、头痛和呕吐。 |
| 分子式 |
C50H68N14O10
|
|---|---|
| 分子量 |
1025.18
|
| 精确质量 |
1024.524
|
| 元素分析 |
C, 58.58; H, 6.69; N, 19.13; O, 15.61
|
| CAS号 |
189691-06-3
|
| 相关CAS号 |
Bremelanotide Acetate; 1607799-13-2
|
| PubChem CID |
9941379
|
| 序列 |
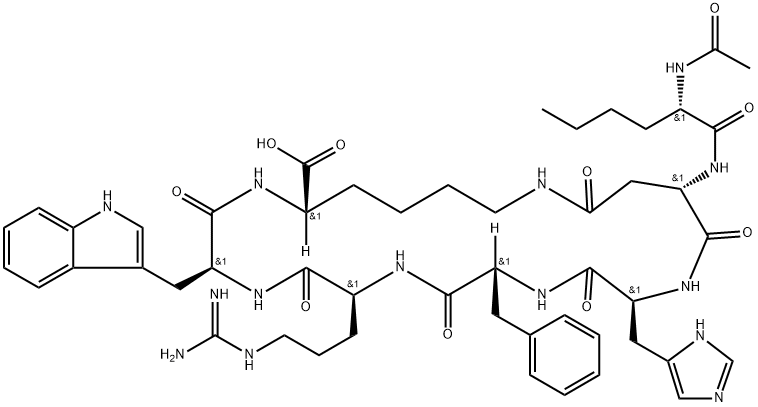
Nle-D(1)HFRWK(1); Ac-Nle-Asp(1)-His-D-Phe-Arg-Trp-Lys(1)-OH
|
| 短序列 |
XDHFRWK
|
| 外观&性状 |
Solid powder
|
| 密度 |
1.4±0.1 g/cm3
|
| 折射率 |
1.679
|
| LogP |
-1.21
|
| tPSA |
376.47
|
| 氢键供体(HBD)数目 |
13
|
| 氢键受体(HBA)数目 |
12
|
| 可旋转键数目(RBC) |
17
|
| 重原子数目 |
74
|
| 分子复杂度/Complexity |
1950
|
| 定义原子立体中心数目 |
7
|
| SMILES |
CCCC[C@H](NC(C)=O)C(N[C@H]1CC(NCCCC[C@H](NC([C@@H](NC([C@@H](NC([C@H](NC([C@@H](NC1=O)CC2=CN=CN2)=O)CC3=CC=CC=C3)=O)CCCNC(N)=N)=O)CC4=CNC5=CC=CC=C45)=O)C(O)=O)=O)=O
|
| InChi Key |
FFHBJDQSGDNCIV-MFVUMRCOSA-N
|
| InChi Code |
InChI=1S/C50H68N14O10/c1-3-4-16-35(58-29(2)65)43(67)64-41-25-42(66)54-20-11-10-18-37(49(73)74)60-46(70)39(23-31-26-56-34-17-9-8-15-33(31)34)62-44(68)36(19-12-21-55-50(51)52)59-45(69)38(22-30-13-6-5-7-14-30)61-47(71)40(63-48(41)72)24-32-27-53-28-57-32/h5-9,13-15,17,26-28,35-41,56H,3-4,10-12,16,18-25H2,1-2H3,(H,53,57)(H,54,66)(H,58,65)(H,59,69)(H,60,70)(H,61,71)(H,62,68)(H,63,72)(H,64,67)(H,73,74)(H4,51,52,55)/t35-,36-,37-,38+,39-,40-,41-/m0/s1
|
| 化学名 |
3S,6S,9R,12S,15S,23S)-15-[[(2S)-2-acetamidohexanoyl]amino]-9-benzyl-6-[3-(diaminomethylideneamino)propyl]-12-(1H-imidazol-5-ylmethyl)-3-(1H-indol-3-ylmethyl)-2,5,8,11,14,17-hexaoxo-1,4,7,10,13,18-hexazacyclotricosane-23-carboxylic acid
|
| 别名 |
PT-141; PT141; 189691-06-3; Bremelanotida; 6Y24O4F92S; PT-141 FREE BASE; Bremelanotide; PT 141
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.9754 mL | 4.8772 mL | 9.7544 mL | |
| 5 mM | 0.1951 mL | 0.9754 mL | 1.9509 mL | |
| 10 mM | 0.0975 mL | 0.4877 mL | 0.9754 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05709444 | Recruiting | Drug: Bremelanotide Drug: RAAS inhibition therapy |
Kidney Disease | Palatin Technologies, Inc | December 26, 2022 | Phase 2 |
| NCT04943068 | Recruiting | Drug: Bremelanotide Drug: Placebo |
Hypoactive Sexual Desire Disorder | Kwang Dong Pharmaceutical co., ltd. |
May 10, 2021 | Phase 3 |
| NCT04179734 | Recruiting | Drug: Bremelanotide Drug: Placebo |
Hypoactive Sexual Desire Disorder | Imperial College Healthcare NHS Trust |
October 7, 2019 | Phase 4 |
| NCT03973047 | Completed | Drug: Bremelanotide Drug: Zofran |
Nausea | AMAG Pharmaceuticals, Inc. | June 17, 2019 | Phase 1 |
| NCT00425256 | Recruiting | Drug: Bremelanotide | Sexual Arousal Disorder | Palatin Technologies, Inc | February 2006 | Phase 2 |