| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
|
| 靶点 |
Ferroptosis; system Xc-
System xc- (cystine-glutamate antiporter) inhibitor (induces ferroptosis via cysteine deprivation and subsequent glutathione depletion) [1] |
|---|---|
| 体外研究 (In Vitro) |
Imidazole ketone erastin(IKE)能有效减少DLBCL细胞数量[1]
18个DLBCL细胞系对IKE抑制表现出不同的敏感性,IC50<100 nM的细胞系被归类为敏感细胞系,IC50>10μM的细胞系归类为耐药细胞系,而IC50值在100 nM至10μM之间的细胞系则被归类为中度耐药细胞系(图1B)。我们进一步测试了与铁下垂抑制剂fer-1共同治疗后,IKE诱导的致死程度,fer-1是一种自由基捕获抗氧化剂,可抑制铁下垂过程中的致命脂质过氧化(Skouta等人,2014,Zilka等人,2017)。与fer-1的联合治疗挽救了DLBCL细胞系中由IKE诱导的细胞死亡,表明IKE在这些细胞系中诱导的致死性是由脂质过氧化和铁中毒引起的。[1] 先前的研究发现,IKE抑制谷氨酸释放,而IKE亲本类似物erastin抑制胱氨酸摄取。因此,我们测试了细胞水平的还原型谷胱甘肽(GSH),其生物合成需要半胱氨酸,作为IKE效力的读数。荧光法显示IKE对GSH的剂量依赖性耗竭(图1C);这种效应被10μMβ-ME的共同处理所逆转,它将胱氨酸还原为半胱氨酸,允许其通过系统A、ASC和L进入细胞,从而绕过系统xc−的抑制。在SUDHL-6细胞中,IKE对GSH耗竭的IC50为34 nM(图S1B),而柳氮磺胺吡啶对GSH耗尽的IC50在毫摩尔范围内。[1] 虽然与DFO的联合处理抑制了培养物中IKE诱导的细胞死亡(图S1E),但在IKE处理后,它仅部分消除了脂质代谢变化,这可能是由于DFO抑制了铁介导的脂质过氧化,而不是酶介导的脂质过氧化,这表明诱导细胞死亡只需要一部分脂质代谢变化。[1] Imidazole ketone erastin (IKE) 能有效降低18种弥漫性大B细胞淋巴瘤细胞系的细胞活性,敏感性各异。IC50值范围从<100 nM(敏感)到>10 µM(耐药)。在敏感的SUDHL-6细胞系中,IKE诱导的细胞死亡可被铁死亡抑制剂ferrostatin-1共处理所挽救,证实了铁死亡是其细胞死亡机制。[1] 在SUDHL-6细胞中,IKE处理(500 nM)导致还原型谷胱甘肽剂量依赖性耗竭,其GSH耗竭的IC50为34 nM。该效应可被β-巯基乙醇共处理逆转。[1] IKE处理在SUDHL-6细胞中诱导了脂质过氧化,通过流式细胞术检测C11-BODIPY荧光增强证实。该增强可被ferrostatin-1共处理抑制。[1] 使用抗二氢吡啶-MDA-赖氨酸加合物抗体进行的免疫荧光染色显示,IKE处理的SUDHL-6细胞中该脂质过氧化加合物水平增加。[1] SUDHL-6细胞的RT-qPCR分析显示,IKE处理(500 nM)显著上调了与铁死亡和系统 xc- 抑制相关的基因表达,包括 SLC7A11、PTGS2 和 CHAC1。PTGS2 的上调可被ferrostatin-1共处理抑制,而 CHAC1 的上调则不受影响。β-巯基乙醇共处理可阻止所有三个基因的上调。[1] 对IKE处理的SUDHL-6细胞进行的非靶向脂质组学分析显示,62种脂质物种发生显著改变,包括含多不饱和脂肪酸的磷脂酰胆碱、磷脂酰乙醇胺和三酰甘油的减少。这些变化可被β-巯基乙醇共处理逆转。Ferrostatin-1共处理增加了TAG水平并降低了单酰甘油水平。[1] IKE处理上调了参与脂质从头合成、磷脂重塑和酶介导的脂质过氧化的基因的mRNA水平。这种上调可被ferrostatin-1或β-巯基乙醇共处理部分逆转。[1] |
| 体内研究 (In Vivo) |
IKE体内药代动力学(PK)和药效学(PD)[1]
为了确定IKE在体内研究中的适用性,我们首先通过在NOD/SCID小鼠中使用腹膜内(IP)、静脉内(IV)和口服(PO)途径给药单剂量IKE(50mg/kg,5%DMSO,pH 4的HBSS)来评估多种给药途径。在8小时内测定IKE浓度表明IP是IKE给药的最有效和最实用的方法(表S1)。接下来,在携带SUDHL6异种移植物的NCG小鼠中,在24小时内单剂量服用IKE(50mg/kg,5%DMSO在pH 4的HBSS中,IP)后,测定血浆和肿瘤样本中的IKE浓度。IKE在1.35小时达到最高血浆浓度5.2μg/mL,在3.30小时达到最高肿瘤积聚2.5μg/mL(图3A,表S2)。[1] IKE体内非靶向脂质组学研究[1] 我们试图研究体内IKE治疗引起的脂质代谢变化。我们在不同时间点用单剂量IKE对肿瘤组织进行了非靶向脂质组学研究。我们发现IKE治疗后游离脂肪酸、磷脂和二酰基甘油(DAG)的相对丰度显著增加(单因素方差分析p<0.05)(图3F和图3G)。与细胞培养实验的差异可能源于体内不同的肿瘤微环境。鉴定的脂质富含亚油酸和花生四烯酸代谢(图S3A)。DAGs和游离脂肪酸水平的显著增加可能是由ATGL介导的TAG水解引起的(图S3B)。增加的脂肪酸可能反过来促进磷脂重塑以合成特定的磷脂,包括PC和PE。为了探索游离脂肪酸对细胞和铁下垂的影响,我们在有或没有IKE的情况下进行了游离脂肪酸的细胞存活测试。[1] IKE PEG-PLGA NP具有适合在体内应用的特性[1] IKE在酸性水条件下可溶,但在中性水条件下溶解度不同(图1A)。为了改善化合物的递送,我们试图使用纳米粒子制剂。我们选择了基于生物相容性和可生物降解的PEG-PLGA二嵌段共聚物的纳米粒子作为IKE载体系统(图4A)。PEG块用于通过与水分子的紧密结合来创建可变形的水合层,这可以防止单核吞噬细胞系统(MPS)的清除,从而延长循环寿命。PLGA块用于形成疏水性核心,以结合IKE,IKE通过扩散和表面及本体侵蚀提供持续释放。[1] IKE抑制体内肿瘤生长,PEG-PLGA-NP制剂提高其治疗指数[1] 我们研究了IKE在携带SUDHL6皮下异种移植物的雄性NCG小鼠体内的疗效。一旦肿瘤体积达到100 mm3,将小鼠随机分为五组,每天通过IP注射一次,分别用载体(pH 4的HBSS中5%的DMSO)、水中未官能化的PEG-PLGA NP、40 mg/kg游离IKE(pH 4时HBSS中5%DMSO)、23 mg/kg游离IKE。在实验期间,每天测量小鼠体重和肿瘤体积,以确定IKE的抗肿瘤作用和可能的毒性。肿瘤生长计算为第一次给药前第0天原始肿瘤体积的倍数变化(图4C)。从治疗的第9天开始,施用40mg/kg IKE、23mg/kg IKE和23mg/kg IKE NP导致肿瘤生长显著减少。23mg/kg游离IKE和23mg/kg IKE NP的肿瘤生长抑制作用没有显著差异;然而,如重量减轻所示,IKE NP的毒性较小(图4D)。与生理盐水载体相比,游离IKE-(pH 4的HBSS中5%的DMSO)处理的小鼠从第9天开始减肥,这可能是由于在pH范围为7.5-8.0的腹膜环境中给药后IKE沉淀造成的,对腹部器官造成损伤,或可能对全身系统xc-抑制产生毒性,或IKE的脱靶毒性。然而,经IKE NP处理的小鼠体重与生理盐水载体组和NP载体组相似;IKE NP制剂的较低毒性可能是由于NP能够防止疏水性药物的聚集(Sun等人,2014),或NP EPR效应,这降低了与常规疏水性药物相关的非特异性分布和全身毒性(Yue等人,2013)。通过使用LC-MS分析IKE肿瘤积聚,我们发现与23mg/kg的游离IKE相比,23mg/kg的IKE NP略微增强了肿瘤积聚,与40mg/kg的游离IKE治疗相当(图S5A)。总体而言,PEG-PLGA-NP制剂增加了IKE的治疗窗口。 在NCG小鼠的SUDHL-6皮下移植瘤模型中,每日一次腹腔注射Imidazole ketone erastin (IKE)(23 mg/kg 或 40 mg/kg)与溶媒对照组相比,在14天的治疗期内显著抑制了肿瘤生长。[1] 在移植瘤模型中,单次给药后,IKE处理从4小时开始导致肿瘤组织中GSH显著耗竭。[1] 在肿瘤组织中,IKE处理上调了 PTGS2、SLC7A11 和 CHAC1 mRNA的表达,从给药后3小时开始。[1] 肿瘤组织的免疫荧光分析显示,与溶媒对照组相比,IKE处理组中二氢吡啶-MDA-赖氨酸加合物和8-羟基-2'-脱氧鸟苷的水平增加,表明体内诱导了脂质过氧化和氧化应激。[1] 对来自IKE处理小鼠的肿瘤组织进行的非靶向脂质组学显示,游离脂肪酸、磷脂和二酰甘油的相对丰度显著增加。[1] 将IKE配制在可生物降解的聚乙二醇-聚(乳酸-共-乙醇酸)纳米粒中,所得制剂(IKE NP,含23 mg/kg IKE)抑制肿瘤生长的效果与游离IKE(23 mg/kg)相似,但毒性显著降低(小鼠体重减轻减少)。与同等剂量的游离IKE相比,IKE NP显示出轻微的肿瘤蓄积增强。[1] 疗效研究中的免疫荧光分析证实,在游离IKE或IKE NP处理的小鼠肿瘤中,COX-2蛋白、二氢吡啶-MDA-赖氨酸加合物和8-OH-dG的水平增加,而cleaved caspase-3未增加,表明细胞死亡是铁死亡性而非凋亡性。[1] |
| 酶活实验 |
Glutamate-‐release assay/谷氨酸释放测定。[2]
人星形细胞瘤细胞(CCF-STTG1)被用作胱氨酸-谷氨酸逆向转运蛋白(xc-)的来源。细胞在96孔板中生长。在>95%融合时,取出培养基,用Earle平衡盐溶液(EBSS)洗涤细胞,以去除培养基中含有的谷氨酸。然后将细胞在37°C下与EBSS(空白)或含有胱氨酸80μM(总计)±erastin(30 nM至100μM)的EBSS一起孵育2小时。已知的靶标抑制剂柳氮磺胺吡啶(SAS)和(S)-4-羧基苯甘氨酸(S-4CPG)用作试验中的阳性对照。在培养期后,用荧光法检测释放到培养基中的谷氨酸。将含有谷氨酸氧化酶(0.04 U/mL)、辣根过氧化物酶(0.125 U/mL)和Amplex UltraRed(50μM)的Tris缓冲液(100 mM,pH 7.4)加入板中,并跟踪荧光变化率(ex 530,em 590)。将数据标准化为总量和空白((1-(未知-空白)/(总量-空白))100),并根据标准化荧光强度值确定SAS、S-4CPG、erastin、erastin代谢物和erastin类似物的半最大抑制常数(IC50)[2]。 |
| 细胞实验 |
DLBCL Lines Sensitivity Measurement [1]
将DLBCL细胞以每孔10000个细胞的速度铺在白色384孔板(每孔32μL)上,形成技术复制品,并孵育过夜。然后用8μL培养基处理细胞,该培养基含有两倍稀释的载体系列(DMSO),IKE(从100μM开始),有或没有Fer-1(从200μM开始。孵育24小时后,向每个孔中加入40μL 50%CellTiter-Glo 50%细胞培养基,在室温下摇动孵育15分钟。使用Victor X5平板读数器测量发光。[1] Flow Cytometry Assay [1] 将20万个SUDHL-6细胞接种在六孔板中,并用DMSO、特定浓度的IKE或fer-1处理。最终细胞密度为0.05百万个细胞/mL。24小时后,通过300×g离心5分钟收集细胞。将细胞重新悬浮在含有2μM C11-BODIPY(BODIPY 581/591 C11)的500μL HBSS中,并在37°C下孵育15分钟。将细胞沉淀并重新悬浮在HBSS中。用门控在FL1通道上测量荧光强度,仅记录活细胞(由DMSO处理组构建的门控)。每种情况下至少分析10000个细胞。 DLBCL细胞敏感性测定: 将DLBCL细胞接种于384孔板中过夜培养。然后用Imidazole ketone erastin (IKE)的两倍稀释系列(从100 µM开始)处理细胞,可联合或不联合ferrostatin-1。孵育24小时后,使用发光法细胞活力检测试剂评估细胞活力。测量发光值,数据归一化至溶媒对照。计算剂量反应曲线和IC50值。[1] 脂质ROS流式细胞术检测: 用DMSO、IKE或IKE联合ferrostatin-1处理SUDHL-6细胞24小时。收集细胞,重悬于含有脂质过氧化探针C11-BODIPY的缓冲液中并孵育。通过流式细胞术在FL1通道测量荧光强度,圈定活细胞。[1] 还原型谷胱甘肽测量: 用IKE处理细胞24小时,可联合或不联合β-巯基乙醇。收集细胞,洗涤并计数。裂解细胞,上清液去蛋白。使用商业检测试剂盒,按照制造商说明书,通过荧光法测定GSH水平。[1] 定量PCR: 用IKE、ferrostatin-1或β-巯基乙醇处理细胞指定时间。提取RNA,反转录为cDNA,使用基因特异性引物和SYBR Green预混液进行定量PCR反应。mRNA水平以 HPRT1 为内参归一化,并使用ΔΔCt法计算。[1] 细胞免疫荧光: 处理后的细胞固定、透化并封闭。与一抗孵育过夜,然后与荧光标记的二抗孵育。染色细胞核。通过共聚焦显微镜捕获图像,并量化荧光强度。[1] IKE PEG-PLGA NP的细胞活性测定: 接种细胞,并用游离IKE或IKE负载纳米粒的稀释系列处理。24小时后,使用发光法检测细胞活力。[1] |
| 动物实验 |
采用三种不同给药途径对小鼠进行药代动力学分析[1]
注射前称量NOD/SCID小鼠(12周龄,体重约28 g),并按每笼3只小鼠的密度分组。将IKE溶解于pH 4的5% DMSO/95% Hank's平衡盐溶液(HBSS)中,配制成5 mg/mL的溶液。另取不含IKE的pH 4的5% DMSO/95% HBSS溶液(赋形剂1)作为赋形剂。所有溶液均使用0.22 μm Steriflip过滤器进行除菌。小鼠分别采用三种不同的给药途径:腹腔注射(IP)和口服(PO)50 mg/kg IKE,以及静脉注射(IV)17 mg/kg IKE。分别于0、1、3、4和8小时采集各时间点3只小鼠的样本。此外,每组另设三只小鼠作为对照组,分别通过腹腔注射(IP)、口服(PO)和静脉注射(IV)给予等量的载体1,并在8小时后采集样本。在适当时间点,用二氧化碳窒息法处死小鼠3分钟,并通过心脏穿刺采集约0.5 mL血液。血液立即转移至K3 EDTA微量离心管(SARSTEDT 41.1504.105)中,并置于冰上。样本在4℃下以2100 × g离心10分钟,然后将血浆转移至干净的离心管中。血浆样本经液氮速冻后储存于-80℃。IKE的提取方法为:取100 μL血浆,加入900 μL乙腈。样本在室温下旋转混合至少5分钟,并在4℃下以4000 × g超声处理10分钟后进行浓缩。移除上清液,并在 GeneVac 蒸发器上以 HPLC 设置过夜干燥。干燥后,将样品重悬于 100 μL 甲醇中,并进行液相色谱-质谱联用 (LC-MS) 分析,每个样品分析两次。质量控制标准样品通过将 IKE 溶解于 100 μL 水中,并采用相同步骤进行提取制备,以确保提取效率。[1] 在携带 SUDHL6 异种移植瘤的 NCG 小鼠中进行药代动力学和药效学分析[1] 将 IKE 溶解于 pH 4 的 5% DMSO/95% HBSS 溶液中,配制成 5 mg/mL 或 3 mg/mL 的溶液。以pH值为4的5% DMSO/95% HBSS溶液作为载体1。使用NanoAssemblr制备的IKE-PEG-PLGA纳米颗粒和未功能化的PEG-PLGA纳米颗粒(不含IKE)(载体2)在去离子水中透析过夜,并至少更换两次透析液。透析后的IKE-PEG-PLGA纳米颗粒和未功能化的PEG-PLGA纳米颗粒经Amicon Ultra-15超滤离心管浓缩,配制成浓度为80 mg/mL的PEG-PLGA纳米颗粒溶液。[1] IKE功效研究[1] 将IKE溶解于pH值为4的5% DMSO/95% HBSS溶液中,配制成浓度为4 mg/mL的溶液。以pH值为4的5% DMSO/95% HBSS溶液作为载体1。使用NanoAssemblr制备的IKE修饰的PEG-PLGA纳米颗粒和未功能化的PEG-PLGA纳米颗粒(未负载IKE)(载体2)在去离子水中透析过夜;透析液至少更换两次。透析后的IKE修饰的PEG-PLGA纳米颗粒和未功能化的PEG-PLGA纳米颗粒经Amicon Ultra-15超滤离心管浓缩,制备浓度为80 mg/mL的PEG-PLGA纳米颗粒溶液。 NOD/SCID小鼠的药代动力学:将咪唑酮类药物erastin (IKE) 溶解于pH 4的5% DMSO/Hank's平衡盐溶液(HBSS)中。小鼠单次给予IKE(腹腔注射和口服途径为50 mg/kg;静脉注射途径为17 mg/kg)。在不同时间点采集血样。分离血浆,并用乙腈提取IKE。采用液相色谱-质谱联用(LC-MS)分析样品,以测定IKE浓度随时间的变化。[1] 异种移植模型的药代动力学/药效学:将皮下移植SUDHL-6异种移植瘤的NCG小鼠单次给予IKE(50 mg/kg,腹腔注射,溶于pH 4的5% DMSO/HBSS)。在不同时间点对小鼠实施安乐死。收集血浆和肿瘤样本。将肿瘤样本匀浆化,提取 IKE 并用液相色谱-质谱联用(LC-MS)进行定量。同时,另取肿瘤样本进行谷胱甘肽(GSH)测定、qPCR、免疫荧光和脂质组学分析。[1] 异种移植模型疗效研究:将携带 SUDHL-6 异种移植瘤(肿瘤体积约 100 mm³)的 NCG 小鼠随机分组。连续 14 天,每天腹腔注射一次以下药物:载体(5% DMSO/HBSS pH4)、空白 PEG-PLGA 纳米颗粒、游离 IKE(23 或 40 mg/kg,溶于 5% DMSO/HBSS pH4)或载 IKE 的 PEG-PLGA 纳米颗粒(相当于 23 mg/kg IKE 溶于水)。每日测量肿瘤大小和小鼠体重。小鼠在末次给药后3小时处死,并收集肿瘤进行分析(IKE浓度、生物标志物、脂质组学)。[1] |
| 药代性质 (ADME/PK) |
在NOD/SCID小鼠中,单次腹腔注射咪唑酮类药物erastin (IKE) (50 mg/kg)后,血浆浓度(Cmax)在1.35小时达到峰值(Tmax),为5.2 µg/mL。与静脉注射和口服途径相比,腹腔注射被认为是最有效且最便捷的给药途径。[1] 在携带SUDHL-6异种移植瘤的NCG小鼠中,单次腹腔注射IKE (50 mg/kg)后,血浆浓度在1.35小时达到峰值(Cmax),为5.2 µg/mL,肿瘤浓度在3.30小时达到峰值(Cmax),为2.5 µg/mL。血浆半衰期(T1/2)为1.83小时,肿瘤半衰期为3.50小时。 [1]
与血浆相比,PEG-PLGA纳米颗粒包裹的IKE在肿瘤组织中的蓄积量增加,且与等剂量游离IKE相比,肿瘤蓄积量略有提高。[1] IKE具有中等水溶性,在酸性条件下可溶,但在中性pH条件下溶解度较低。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
在疗效研究中,每日腹腔注射游离的咪唑酮类药物erastin (IKE)(23 mg/kg 和 40 mg/kg,溶于 5% DMSO/HBSS pH4 溶液)导致小鼠体重显著下降,该现象从治疗第 9 天左右开始出现。[1]
观察到的游离 IKE 的毒性(体重下降)归因于药物在腹腔(pH 值约为 7.5-8.0)中的潜在沉淀和/或系统性抑制或脱靶效应。[1] 将 IKE 制成 PEG-PLGA 纳米颗粒(IKE NP,相当于 23 mg/kg IKE)可显著降低这种毒性,与载体对照组相比,小鼠体重未出现显著下降。这种毒性的降低归因于纳米颗粒能够防止药物聚集,并通过增强渗透性和滞留性 (EPR) 效应增强肿瘤靶向性,从而降低全身暴露量。 [1] 给予空白PEG-PLGA纳米颗粒(每日750 mg/kg,持续两周)未引起可检测到的体重减轻或可观察到的毒性迹象。[1] |
| 参考文献 | |
| 其他信息 |
铁死亡是一种受调控的细胞死亡形式,可通过抑制胱氨酸-谷氨酸逆向转运蛋白系统xc-诱导。在现有的xc-抑制剂中,咪唑酮类化合物erastin (IKE) 是一种强效且代谢稳定的xc-抑制剂,同时也是一种铁死亡诱导剂,具有潜在的体内应用价值。我们利用弥漫性大B细胞淋巴瘤(DLBCL)异种移植模型研究了IKE的药代动力学和药效学特征,结果表明IKE通过抑制xc-发挥抗肿瘤作用,导致谷胱甘肽耗竭、脂质过氧化,并在体外和体内诱导铁死亡生物标志物的表达。我们利用非靶向脂质组学和qPCR技术,鉴定了IKE诱导的铁死亡过程中脂质代谢的独特特征。此外,可生物降解的聚乙二醇-聚乳酸-羟基乙酸共聚物纳米颗粒被用于辅助IKE递送,并在弥漫性大B细胞淋巴瘤(DLBCL)异种移植模型中显示出比游离IKE更低的毒性。[1]
在原本不具有亲电官能团的骨架上引入反应性羰基,从而构建可逆共价抑制剂,是一种潜在的提高化合物效力的有效策略。然而,醛类化合物代谢不稳定,这使得该策略无法应用于动物模型或人体临床研究中。为了克服这一限制,我们设计了能够形成可逆共价加合物的酮基官能团,同时该官能团具有较高的代谢稳定性,并能提高其侧链骨架的水溶性。我们用铁死亡诱导剂和实验性治疗药物erastin测试了该策略,并观察到化合物效力显著提高。特别是,一种名为 IKE 的新型羰基 erastin 类似物展现出更高的效力、溶解度和代谢稳定性,因此是未来体内癌症治疗应用的理想候选药物。[2] 咪唑酮 erastin (IKE) 是 erastin 的一种类似物,其设计包含酮基、异丙氧基取代基和咪唑部分。与 erastin 相比,这些修饰赋予了 IKE 纳摩尔级的效力、更高的代谢稳定性以及在酸性条件下更高的水溶性。[1] IKE 可诱导铁死亡,这是一种由脂质过氧化驱动的铁依赖性细胞死亡,其主要机制是通过抑制胱氨酸-谷氨酸反向转运系统 xc-,导致半胱氨酸缺乏、谷胱甘肽耗竭和谷胱甘肽过氧化物酶 4 (GPX4) 失活。 [1] 该研究强调了诱导铁死亡作为治疗依赖胱氨酸输入的癌症(例如弥漫性大B细胞淋巴瘤(DLBCL))的潜在策略。[1] 研究采用可生物降解的PEG-PLGA纳米颗粒作为递送系统,通过增强肿瘤蓄积和降低全身毒性来提高IKE的治疗指数。[1] 研究鉴定了铁死亡的药效学生物标志物,包括PTGS2、SLC7A11和CHAC1 mRNA的上调,以及体内脂质过氧化加合物(MDA-赖氨酸)和氧化性DNA损伤(8-OH-dG)水平的升高。[1] 脂质组学分析揭示了IKE诱导的铁死亡过程中脂质代谢的显著变化,包括含PUFA的磷脂和TAG的减少,以及脂质生物合成和重塑途径的激活。[1] |
| 分子式 |
C35H35CLN6O5
|
|---|---|
| 分子量 |
655.1426
|
| 精确质量 |
654.235
|
| 元素分析 |
C, 64.17; H, 5.39; Cl, 5.41; N, 12.83; O, 12.21
|
| CAS号 |
1801530-11-9
|
| PubChem CID |
91824786
|
| 外观&性状 |
White to yellow solid powder
|
| LogP |
4.5
|
| tPSA |
110Ų
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
8
|
| 可旋转键数目(RBC) |
11
|
| 重原子数目 |
47
|
| 分子复杂度/Complexity |
1120
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
PSPXJPWGVFNGQI-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C35H35ClN6O5/c1-24(2)47-32-12-7-25(31(43)20-40-14-13-37-23-40)19-30(32)42-33(38-29-6-4-3-5-28(29)35(42)45)21-39-15-17-41(18-16-39)34(44)22-46-27-10-8-26(36)9-11-27/h3-14,19,23-24H,15-18,20-22H2,1-2H3
|
| 化学名 |
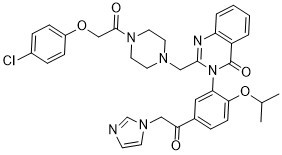
3-(5-(2-(1H-imidazol-1-yl)acetyl)-2-isopropoxyphenyl)-2-((4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)methyl)quinazolin-4(3H)-one
|
| 别名 |
Imidazole ketone erastin; IKE; Imidazole ketone erastin; 1801530-11-9; IKE; PUN30119; PUN-301193-(5-(2-(1H-imidazol-1-yl)acetyl)-2-isopropoxyphenyl)-2-((4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)methyl)quinazolin-4(3H)-one; CHEMBL3629671; 2-({4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl}methyl)-3-{5-[2-(imidazol-1-yl)acetyl]-2-isopropoxyphenyl}quinazolin-4-one; Imidazole ketone erastinIKE; Ferroptosis inducer IKE;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5264 mL | 7.6320 mL | 15.2639 mL | |
| 5 mM | 0.3053 mL | 1.5264 mL | 3.0528 mL | |
| 10 mM | 0.1526 mL | 0.7632 mL | 1.5264 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。